Lab 20 Sodium Carbonate or Sodium Bicarbonate Purpose

Lab 20 Sodium Carbonate or Sodium Bicarbonate Purpose Use mole ratios to determine a compound to be either Na 2 CO 3 or Na. HCO 3. Background Na. HCO 3 = sodium bicarbonate (baking soda) Na 2 CO 3 = sodium carbonate Both compounds react with HCl to produce Na. Cl, CO 2, and H 2 O HCl = hydrochloric acid
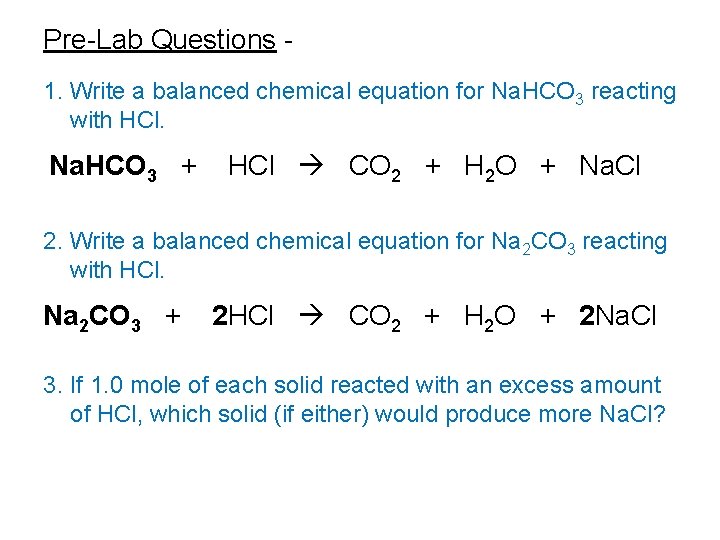
Pre-Lab Questions 1. Write a balanced chemical equation for Na. HCO 3 reacting with HCl. Na. HCO 3 + HCl CO 2 + H 2 O + Na. Cl 2. Write a balanced chemical equation for Na 2 CO 3 reacting with HCl. Na 2 CO 3 + 2 HCl CO 2 + H 2 O + 2 Na. Cl 3. If 1. 0 mole of each solid reacted with an excess amount of HCl, which solid (if either) would produce more Na. Cl?

Experiment (M 1) Mass of empty evap. dish + watch glass = ____ g (M 2) Mass of dish + watch glass + 1. 00 gram of solid = ____ g Put ~ 5 m. L of HCl into a beaker. Slowly add drops of HCl to the dish until all of the solid has reacted. Slowly evaporate the liquid in the dish with the watch glass on top of the dish. Avoid spattering. Let the evap. dish cool. (M 3) Mass of evap. dish + watch glass + product = ____ g Clean out dish and watch glass.
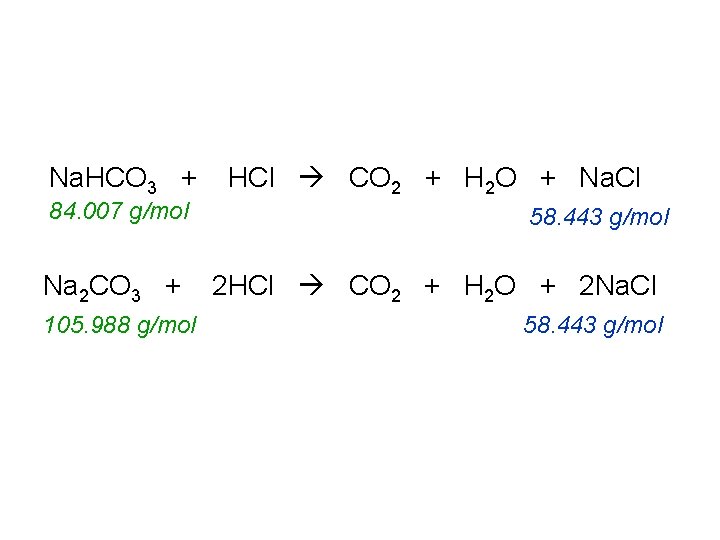
Na. HCO 3 + 84. 007 g/mol Na 2 CO 3 + 105. 988 g/mol HCl CO 2 + H 2 O + Na. Cl 58. 443 g/mol 2 HCl CO 2 + H 2 O + 2 Na. Cl 58. 443 g/mol

Analysis 1. Which reactant was used in excess in this lab? 2. Calculate the mass of the solid you began with. 3. Calculate the mass of the Na. Cl that was produced. 4. Convert the mass of the Na. Cl into moles of Na. Cl. 5. Convert the mass of the solid into moles of Na 2 CO 3. 6. Convert the mass of the solid into moles of Na. HCO 3. 7. Determine the substance to be Na 2 CO 3 or Na. HCO 3 by comparing the mole ratio from your experiment to the balanced chemical equation. Results -
- Slides: 5