Lab 16 Date Chemical Reactions II Purpose Observe

Lab 16 Date: Chemical Reactions II Purpose Observe and classify different types of chemical reactions

Experiment Part 1 – Iron in aqueous copper (II) sulfate Place an iron nail in a beaker containing approx. 40 m. L of copper(II) sulfate solution. Record observations after a few minutes. Return nail & solution back to original place

Part 2 – Copper in aqueous iron (III) sulfate Place a copper wire in a beaker containing approx. 40 m. L of iron (III) sulfate solution. Record observations after a few minutes. Return wire & solution back to original place
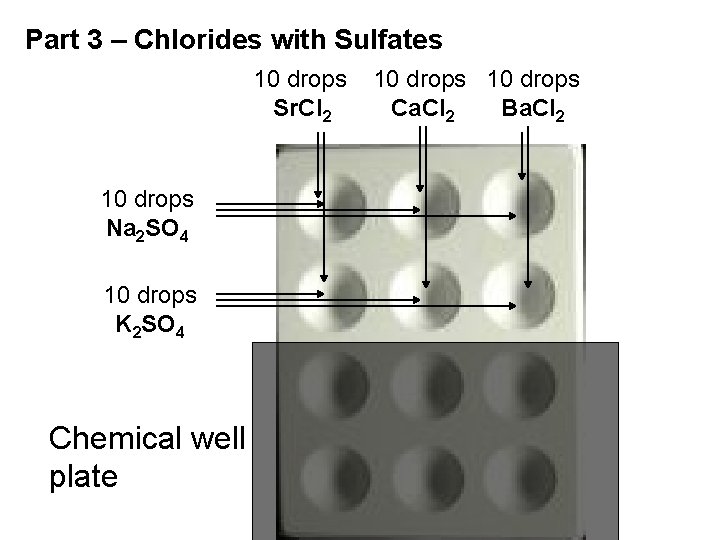
Part 3 – Chlorides with Sulfates 10 drops Sr. Cl 2 10 drops Na 2 SO 4 10 drops K 2 SO 4 Chemical well plate 10 drops Ca. Cl 2 Ba. Cl 2
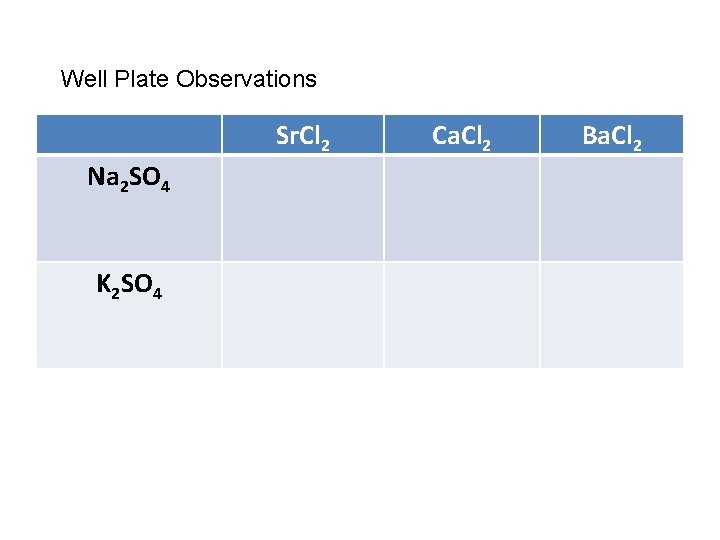
Well Plate Observations Sr. Cl 2 Na 2 SO 4 K 2 SO 4 Ca. Cl 2 Ba. Cl 2

Analysis – 1. Write a skeleton equation for part 1. Assume iron(III) ions were produced. 2. What was observed in part 2? Explain. 3. Write a skeleton equation for all 6 equations in part 3. 4. What was observed in part 3? Explain. 5. Balance the equations in #1 & #3 6. Classify equations 1 and 3 into one of the five reaction types we have discussed. 7. Write a balanced chemical equations for strontium chloride reacting with manganese (II) sulfate. Results –
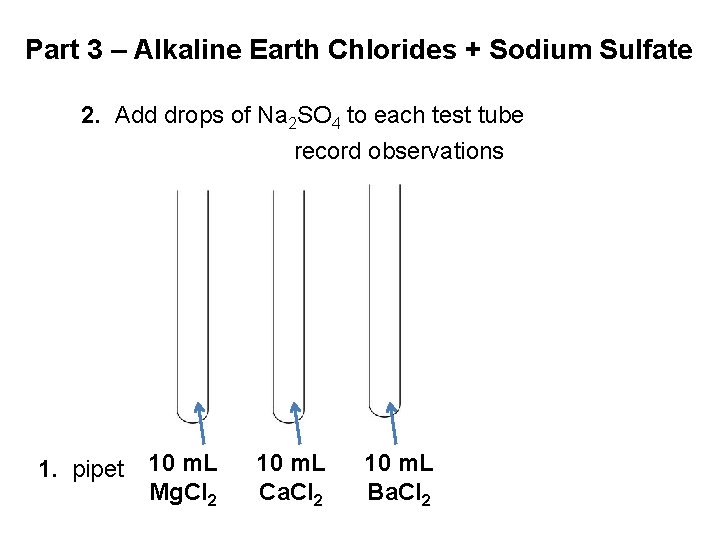
Part 3 – Alkaline Earth Chlorides + Sodium Sulfate 2. Add drops of Na 2 SO 4 to each test tube record observations 1. pipet 10 m. L Mg. Cl 2 10 m. L Ca. Cl 2 10 m. L Ba. Cl 2
- Slides: 7