LAB 1 1 HOW TO USE A MICROPIPETTE

LAB 1. 1: HOW TO USE A MICROPIPETTE

INTRODUCTION • Review Introduction and chapter 1 goals. – (A-17 in student manual). • Purpose of this lab. – Tools of genetic engineering. 2
OVERVIEW • Goals: – Become familiar with the small volumes of solutions used in molecular biology • And why it is important!! – Introduce proper use of the micropipette – Practice using the micropipette 3

INTRODUCTION • Working with extremely small quantities – Why is this necessary? • • Very high cost of purifying enzymes and DNA • Plasmids = $360 million per pound – 0. 45359237 kilogram • Hence, need to use instruments to handle small quantities Volumes measured in: – Microliters (μL) – 1 μL = 0. 001 m. L – 10 μL = 0. 01 m. L – 100 μL = 0. 1 m. L – 1000 μL = 1 m. L 4

THE DIGITAL MICROPIPETTE • Micropipettes – Variable small quantities of liquid – Use with proper tip • Firm connection! – Do not lay down or point tip upward with fluid inside • P-20 – Dispense liquid only between 2 and 20 μL – Too much quantity can contaminate the barrel https: //www. amgenbiotechexperience. com/curriculumresources/using-micropipette 5 Locked Unlocked
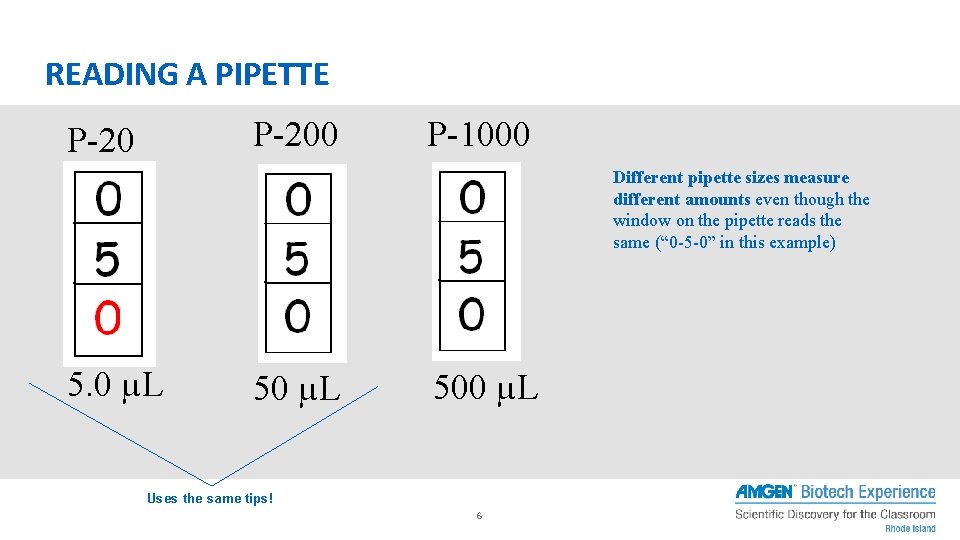
READING A PIPETTE P-200 P-20 P-1000 Different pipette sizes measure different amounts even though the window on the pipette reads the same (“ 0 -5 -0” in this example) 5. 0 µL 500 µL Uses the same tips! 6
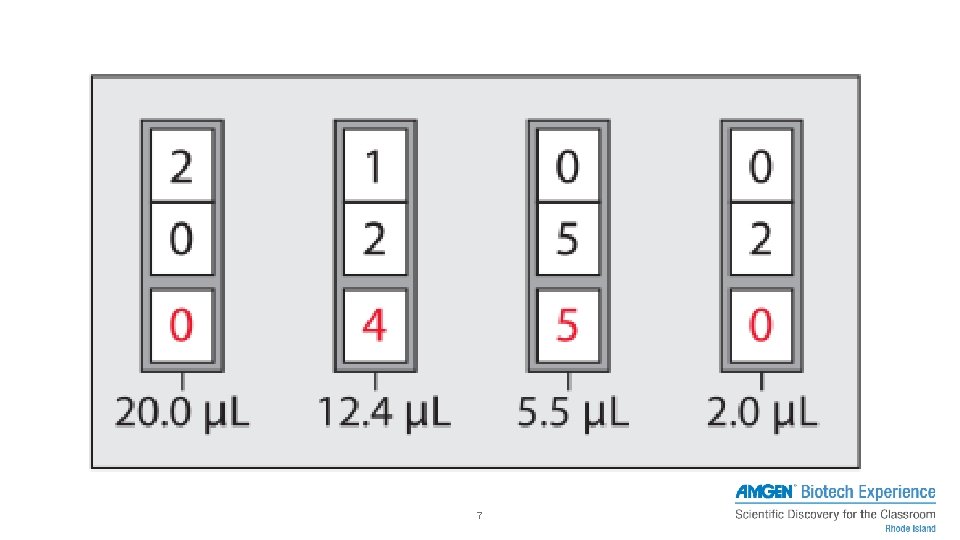
7
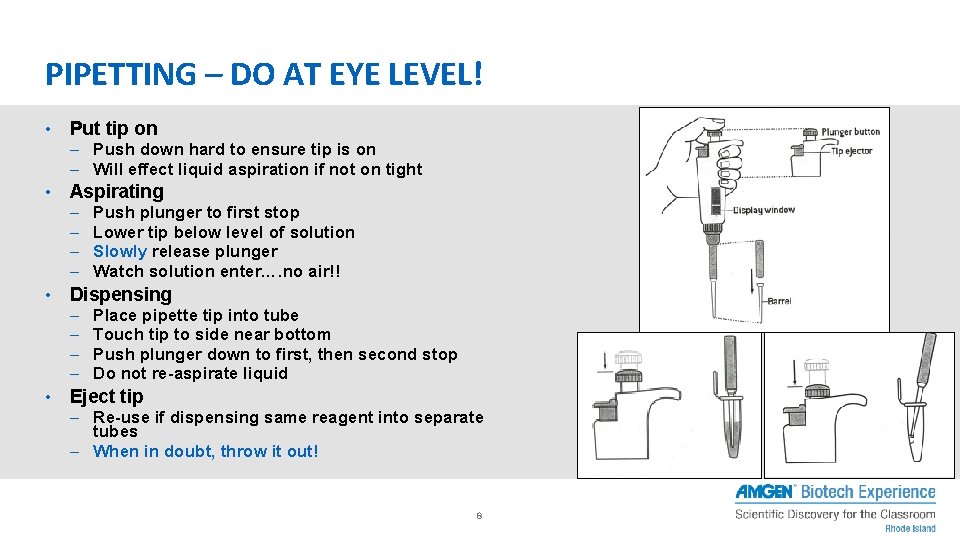
PIPETTING – DO AT EYE LEVEL! • Put tip on – Push down hard to ensure tip is on – Will effect liquid aspiration if not on tight • Aspirating – – • Dispensing – – • Push plunger to first stop Lower tip below level of solution Slowly release plunger Watch solution enter…. no air!! Place pipette tip into tube Touch tip to side near bottom Push plunger down to first, then second stop Do not re-aspirate liquid Eject tip – Re-use if dispensing same reagent into separate tubes – When in doubt, throw it out! 8

MICROPIPETTES “NEVERS” • Never use micropipette without a tip • Never lay micropipette down with sample in the tip • Never let the plunger button snap back • Never set the P-20 pipette below 2. 0μL or above 20. 0μL https: //www. amgenbiotechexperience. com/curriculum-resources/loading-micropipette 9
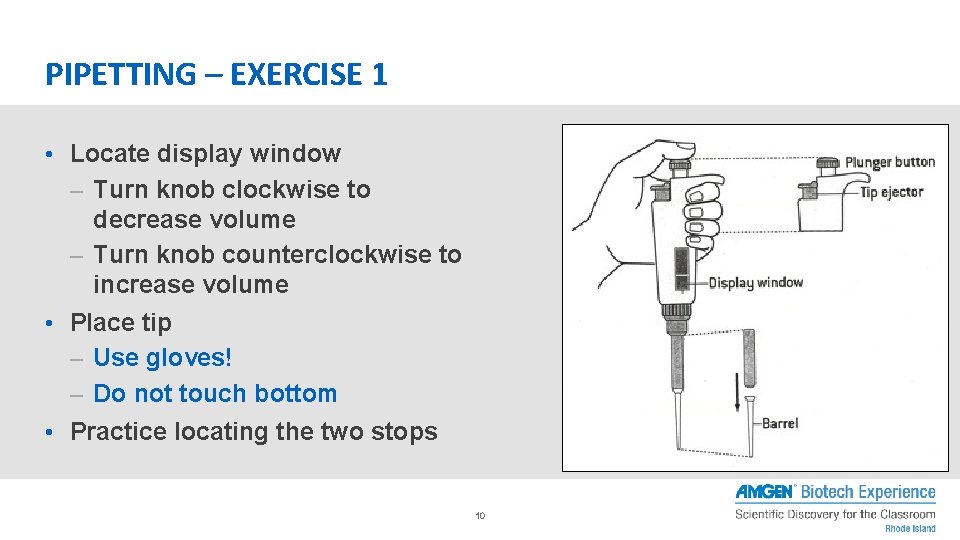
PIPETTING – EXERCISE 1 • Locate display window – Turn knob clockwise to decrease volume – Turn knob counterclockwise to increase volume • Place tip – Use gloves! – Do not touch bottom • Practice locating the two stops 10

PIPETTING TECHNIQUES Hold micropipette and microfuge tubes at eye level when loading or dispensing samples https: //www. amgenbiotechexperience. com/curric ulum/curriculum-resources/working-smallvolumes 11

PIPETTING TECHNIQUES Place both elbows on the table and use your other hand to stabilize the bottom of the pipette 12

TIPS Common mistake! If YOU are pipetting, YOU should be holding the tube 13

VARIOUS AMOUNTS The amount you push the plunger depends on the amount: At 20. 0μL the plunger is much higher than at 2. 0μL 14

MICROPIPETTE USE 1. Twist dial to desired volume 2. Add disposable pipette tip 3. Press plunger to first stop 4. Insert pipette tip into solution to be transferred 5. Slowly release plunger to retrieve liquid 6. Move pipette tip into desired tube 7. Press plunger past first stop to second stop to transfer liquid, keep the plunger down as you remove it from the tube 8. Eject tip 15

PIPETTING – EXERCISE 1 • Load 20μL of red dye (RD) and dispense onto paper towel / • • laminated sheet 15μL of RD 10μL of RD 5μL of RD 2μL of RD 16
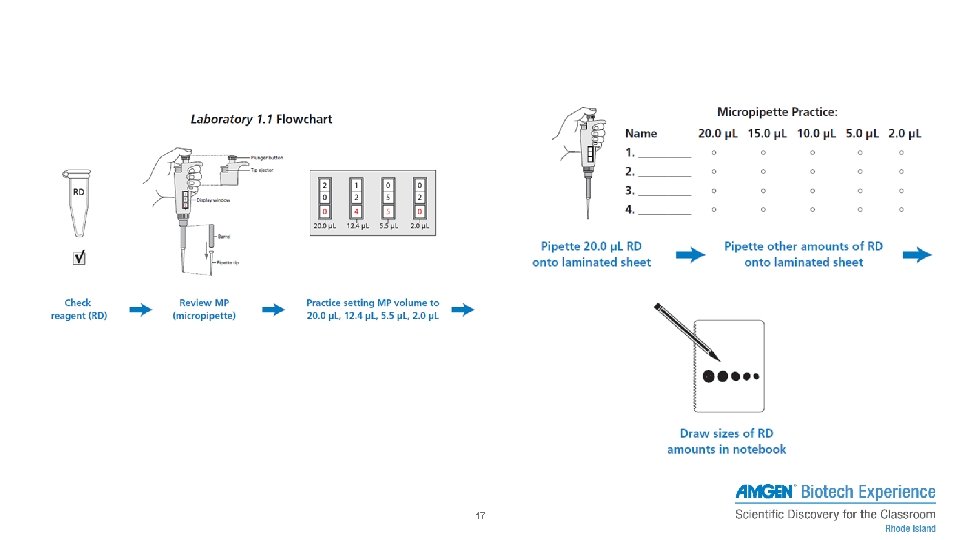
17

LAB 1. 2 Gel Electrophoresis

LAB 1. 2 – GEL ELECTROPHORESIS • Become familiar with gel electrophoresis • Practice using the micropipette to load wells in practice plates • Practice running an electrophoresis gel using three dyes https: //www. amgenbiotechexperience. com/curriculumresources/gel-electrophoresis-gel-analysis 19

WHAT WILL YOU NEED TO DO? • Prepare double-combed 0. 8% agarose gel – Do this several days prior to lab – Can store in plastic Ziplock bags with buffer – **Store “wells up” for orientation • Locate dyes – One rack per group – Each group given 1 tube of solutions 1, 2 and 3 • (S 1, S 2, S 3) • Get the 20 X SB (sodium borate) to 1 X SB 20

MAKING GELS • Estimate that each gel tray holds 30 m. L of agarose gel – 4 gels = 120 m. L ( let's estimate for 5 gels) –. 08 x 150 m. L = 1. 2 grams agarose – A 0. 8% gel for a volume of 150 m. L is : 1. 2 grams of agarose in 150 m. L of 1 X Buffer. • Alternatively; 1. 8 grams of agarose in 200 m. L of 1 X Buffer =6 gels ( 6 gels=180 m. L ) § Melt agarose in microwave ( or hot plate) until all “flecks” are dissolved § Let cool until you can “handle” it (~60°C) § Pour 4 gels, let cool, then store in one or more zip lock bags or plastic container § ***When returning gel to electrophoresis box, make sure wells are facing up https: //www. amgenbiotechexperience. com/curriculumresources/making-agarose-gel 21

SB BUFFER SOLUTION • 1 X SB buffer used in agarose gel solution • 1 X SB buffer used in electrophoresis box • Materials include 20 x SB buffer solution • Dilute 20 x SB buffer solution with d. H 20 to make the 1 x SB buffer solution: C 1 V 1 = C 2 V 2 22

MAKING GELS • Leave in water bath @ 60°C until your students are ready to pour them – This is after you melt it in an Erlenmeyer flask, in the microwave at a boil – A hot water bath is not hot enough to dissolve agarose. • Students can gently slide it into ziplock bag for lab the next day. https: //www. amgenbiotechexperience. com/curriculumresources/pouring-agarose-gel 23

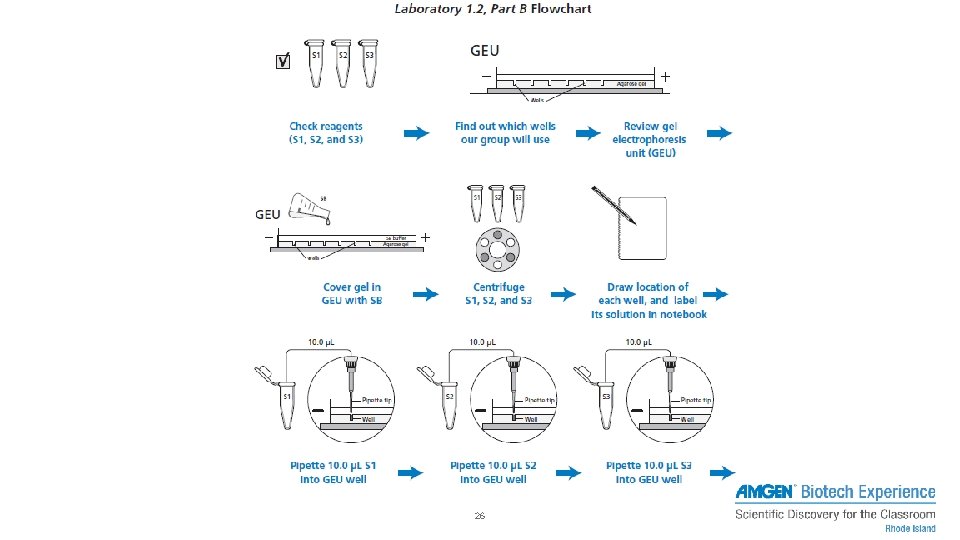
PART A – PIPETTING INTO WELLS 1. Load RD into pipettes 2. Dispense into practice gels/plates 24
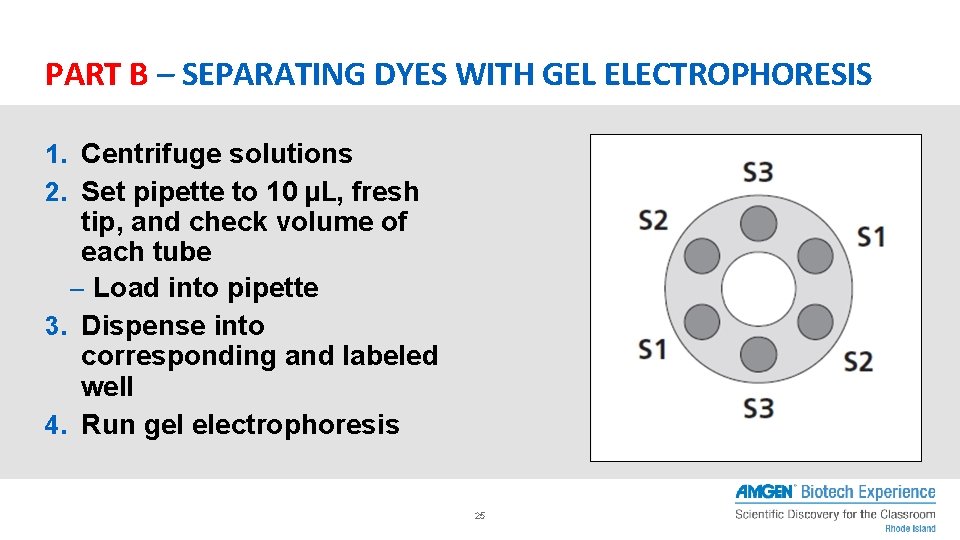
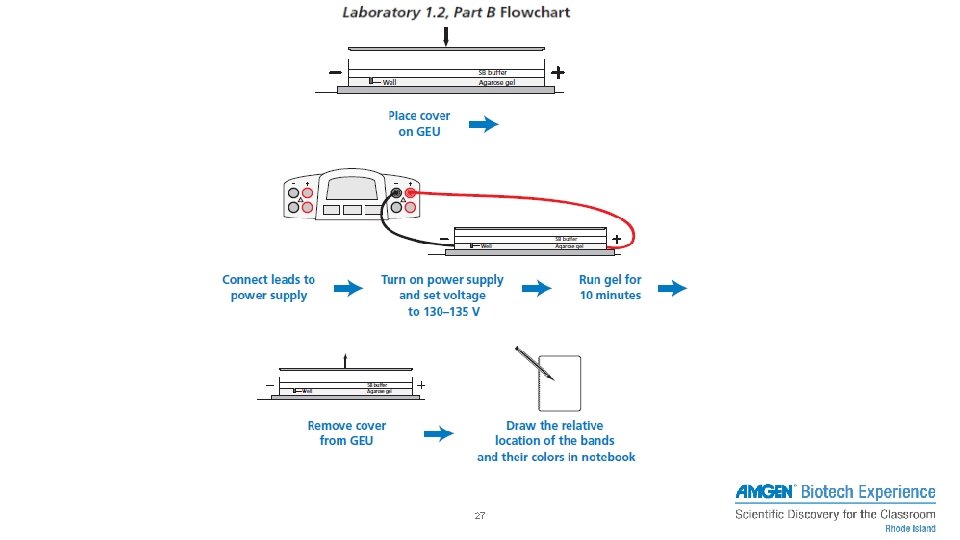
PART B – SEPARATING DYES WITH GEL ELECTROPHORESIS 1. Centrifuge solutions 2. Set pipette to 10 μL, fresh tip, and check volume of each tube – Load into pipette 3. Dispense into corresponding and labeled well 4. Run gel electrophoresis 25
26
27
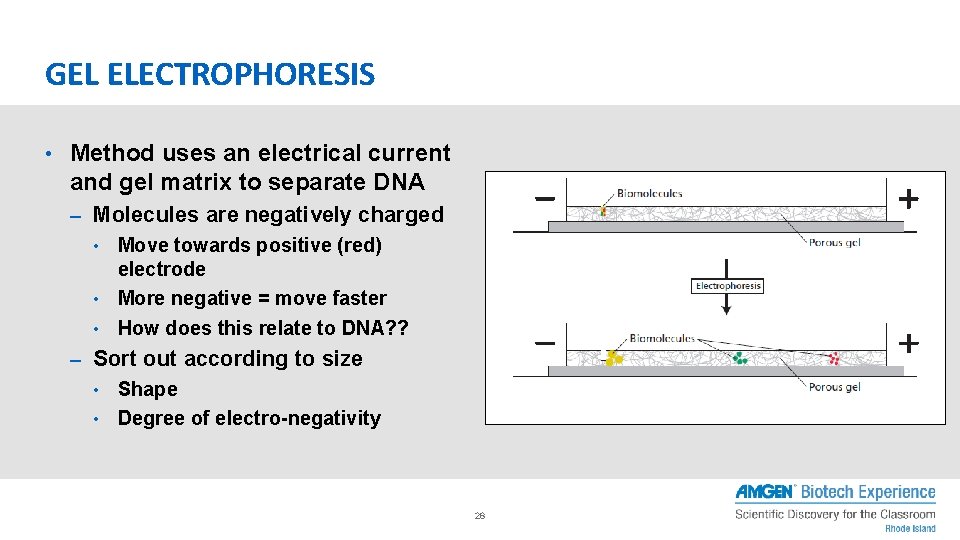
GEL ELECTROPHORESIS • Method uses an electrical current and gel matrix to separate DNA – Molecules are negatively charged • Move towards positive (red) electrode • More negative = move faster • How does this relate to DNA? ? – Sort out according to size • Shape • Degree of electro-negativity 28

SETTING UP THE GEL BOX • Remove tape before placing in the gel box (if needed) • Wells should be at the negative (black) end (run to red) • Buffer should just cover the gel – no dimples – Cover with 1 X SB buffer • Sodium borate • Put the gel box in position before loading the wells https: //www. amgenbiotechexperience. com/curriculumresources/using-electrophoresis-box 29

USING GEL ELECTROPHORESIS TO SEPARATE MOLECULES • Set micropipette to 10 μL and load each sample – Record the wells and what is put into each well: • Loading samples: – Center pipette – Depress plunger slowly – Use two hands! – Under buffer level, above gel well • May be able to skip lanes https: //www. amgenbiotechexperience. com/curriculumresources/loading-gel 30

IMPROPER LOADING TECHNIQUES • Tip is in the well • Tip punched through the gel • Dye spreading under the well 31

PROPER LOADING TECHNIQUE • Tip is above the well and in the buffer • Sample in well Courtesy of K. Schramm 32

USING GEL ELECTROPHORESIS TO SEPARATE MOLECULES • Close cover • Connect electrical leads – Negative – black – Positive - red • Turn on power and set voltage to 130 -135 v – Check for bubbles – After 3 minutes, check that dyes are moving – Turn off after roughly 10 -15 minutes • Should see all three dyes • Turn off power and record results https: //www. amgenbiotechexperience. com/curriculumresources/running-agarose-gel 33

SOLUTIONS • Dyes – Orange G = 408. 40 atomic units (au) – Bromophenol blue = 669. 98 au – Xylene cyanole = 538. 62 au • Heavier molecules move slower • Molecules with more negative electric charge will move faster • Solution 1: – bromophenol blue, xylene cyanole, glycerin, water • Solution 2: – bromophenol blue, xylene cyanole, orange G, glycerin, water • Solution 3: – xylene cyanole, glycerin, water 34 K. Schramm

LET’S PLAY Which dye do you predict has the largest molecular weight? A 3 2 A. Orange G 1 B. Bromophenol blue C. Xylene cyanole 35

SOLUTION Bromophenol blue (purple) is more negatively charged than xylene cyanole (blue) due to negatively charged bromine ions. Therefore, it travels farther than the smaller xylene cyanole. A 3 2 1 36

LAB!!
- Slides: 37