LAA Closure Design Modifications Patient Populations and Reimbursement

LAA Closure: Design Modifications, Patient Populations and Reimbursement Status Ian T. Meredith AM MBBS, Ph. D, FRACP, FCSANZ, FACC, FSCAI, FAPSIC Global Chief Medical Officer Boston Scientific Corporation

Presenter Disclosure Information Ian T. Meredith AM Employee of Boston Scientific Corporation, Global Chief Medical Officer I will be discussing concept devices not available for sale

LAAC with WATCHMANTM Worldwide Experience • 2002 – First in Man Implant • 2005 – CE Mark • 2015 ‒ FDA approval of left atrial appendage occlusion (LAAC) with WATCHMAN device to reduce risk of stroke in NVAF • Clinical Trial Experience ‒ 2 RCTs with 5 year follow-up ‒ >5800 patients studied ‒ >10, 000 patient-years of follow-up • >40, 000 worldwide implants in over 75 countries
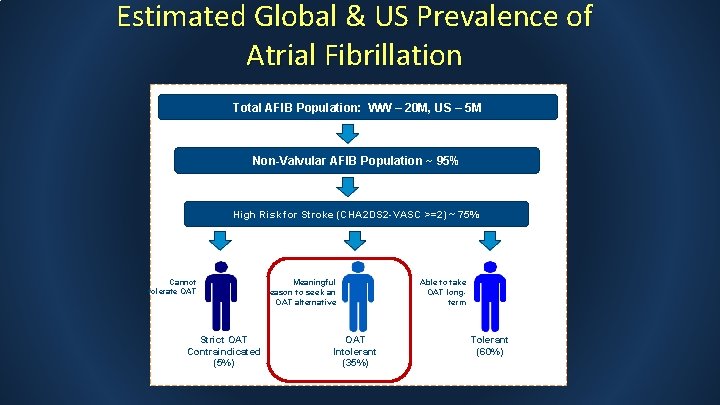
Estimated Global & US Prevalence of Atrial Fibrillation Total AFIB Population: WW – 20 M, US – 5 M Non-Valvular AFIB Population ~ 95% High Risk for Stroke (CHA 2 DS 2 -VASC >=2) ~ 75% Cannot tolerate OAT Strict OAT Contraindicated (5%) Meaningful reason to seek an OAT alternative OAT Intolerant (35%) Able to take OAT longterm Tolerant (60%)

Existing RCTs don’t adequately address majority of AF population OAT Risk >>Benefit Unable to take blood thinners (5%) OAT Risk << Benefit Reasons to seek an alternative to long term blood thinners (35%) PROTECT/PREVAIL WATCH TAVR ASAP Too Able to take long term blood thinners (60%)

Existing RCTs don’t adequately address majority of AF population OAT Risk >>Benefit Unable to take blood thinners (5%) OAT Risk << Benefit Reasons to seek an alternative to long term blood thinners (35%) Able to take long term blood thinners (60%) PROTECT/PREVAIL WATCH TAVR ASAP Too PROTECT/PREVAIL • Theoretically evaluated WM in intolerant and tolerant patients • In reality mainly evaluated intolerant patients • Mixed results in this population and use of warfarin control (now considered 2 nd line therapy) mean that overall WM adoption has been limited

WATCHMAN Indications for Use Worldwide Differences US Indication International Indication: The WATCHMAN Device is indicated to reduce the risk of thromboembolism from the left atrial appendage in patients with non-valvular atrial fibrillation who: The WATCHMAN LAA Closure Technology is intended to prevent thrombus embolization from the left atrial appendage and reduce the risk of life-threatening bleeding events in patients with non-valvular atrial fibrillation who are eligible for anticoagulation therapy Are at increased risk for stroke and systemic embolism based on CHADS 2 or CHA 2 DS 2 -VASc scores and are recommended for anticoagulation therapy; Are deemed by their physicians to be suitable for warfarin; and Have an appropriate rationale to seek a nonpharmacologic alternative to warfarin, taking into account the safety and effectiveness of the device compared to warfarin or who have a contraindication to anticoagulation therapy.

CE Mark Labeling Changes Old DFU New DFU Lifelong ASA for at least 12 months DAPT for 6 Months DAPT for at least 3 months No NOAC usage NOAC use specified Specific Dosages Dosage at physician’s discretion Distinction between “OAC eligible” and “OAC Contraindicated” patients Consideration of patient preference, risk of bleeding, and stroke risk Current US Directions for Use require a combination of short-term warfarin and DAPT in the 6 months following the implant.

Existing RCTs don’t adequately address majority of AF population OAT Risk >>Benefit Unable to take blood thinners (5%) OAT Risk << Benefit Reasons to seek an alternative to long term blood thinners (35%) PROTECT/PREVAIL WATCH TAVR ASAP Too Able to take long term blood thinners (60%)

ASAP-TOO (NCT 02928497): Overview Study Objective Evaluate LAA Closure with WATCHMAN in NVAF patients deemed not suitable for oral anticoagulation therapy Study Design Prospective, multi-center Randomized 2: 1 (Watchman vs Control) Considering Group Sequential Design Effectiveness Endpoint Time to first occurrence of ischemic stroke or systemic embolism Primary Endpoint Safety Endpoint 7 -day rate of all-cause death, ischemic stroke, systemic embolism, or device- or procedure- related events requiring open cardiac surgery or major endovascular intervention Patient Population 888 Number of Sites 100 global sites Follow-up* • • 45 Day with TEE 6, 18 month phone visit 12 month with TEE Bi-annually for years 2 -5 *Brain imaging required at baseline if prior stroke or TIA Holmes et al. AHJ 2017; 189: 68 -74.
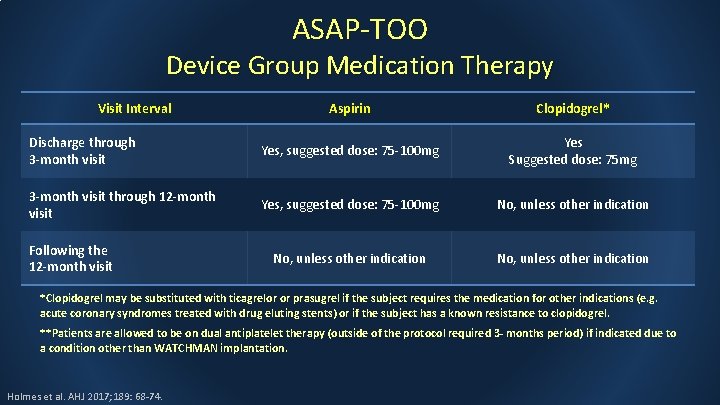
ASAP-TOO Device Group Medication Therapy Visit Interval Aspirin Clopidogrel* Discharge through 3 -month visit Yes, suggested dose: 75 -100 mg Yes Suggested dose: 75 mg 3 -month visit through 12 -month visit Yes, suggested dose: 75 -100 mg No, unless other indication Following the 12 -month visit *Clopidogrel may be substituted with ticagrelor or prasugrel if the subject requires the medication for other indications (e. g. acute coronary syndromes treated with drug eluting stents) or if the subject has a known resistance to clopidogrel. **Patients are allowed to be on dual antiplatelet therapy (outside of the protocol required 3 - months period) if indicated due to a condition other than WATCHMAN implantation. Holmes et al. AHJ 2017; 189: 68 -74.
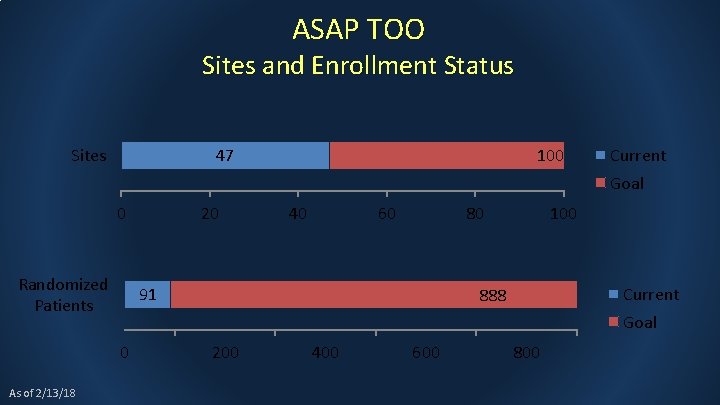
ASAP TOO Sites and Enrollment Status Sites 47 100 Current Goal 0 Randomized Patients 40 60 80 91 100 Current 888 Goal 0 As of 2/13/18 20 200 400 600 800

WATCHMAN Clinical Spectrum Expand Patient Population Concomitant Procedures ASAP-TOO AF Ablation + LAAC First Line in Specific Patient Populations GI Bleeds Renal Pts First Line Therapy WM vs NOAC ? ? ? Prior ICH TAVR + LAAC

Design Modifications

WATCHMAN FLX™ : Designed to Broaden Treatment Matrix and Improve Ease of Use • Designed to enhance stability, aid ease-of-use and enable greater apposition to appendage wall • Anticipate starting EU and U. S. clinical trials mid-year 2018 Caution: WATCHMAN FLX is an Investigational Device. Limited by Federal (or US) law to investigational use only. Not available for sale.

WATCHMAN™ Tru. Seal™ Access System Quick-Close Valve • Longer cap • More robust thread engagement • Fewer turns to close Reinforced Proximal Shaft • Enhanced kink resistance Ergonomic Hub • Larger gripping surface • Anticipate US and EU commercial availability late 2018 Caution: WATCHMAN Tru. Seal Access System is an Investigational Device. Limited by Federal (or US) law to investigational use only. Not available for sale.

Reimbursement
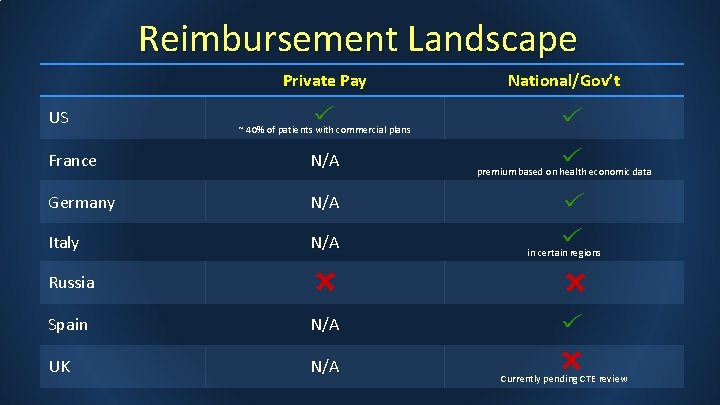
Reimbursement Landscape Private Pay US National/Gov’t ~ 40% of patients with commercial plans France N/A Germany N/A Italy N/A premium based on health economic data in certain regions Russia Spain N/A UK N/A Currently pending CTE review
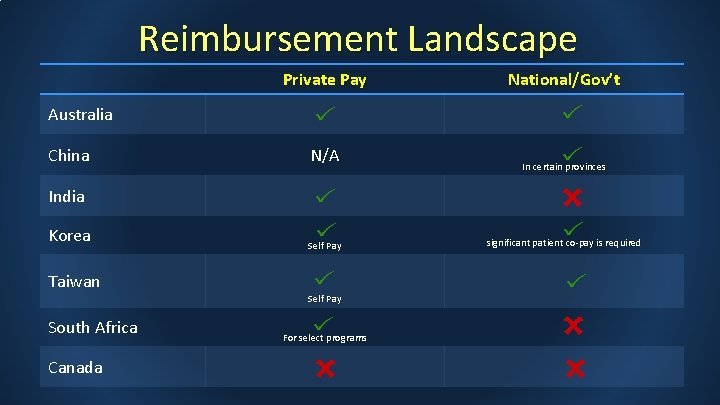
Reimbursement Landscape Private Pay National/Gov’t Australia China N/A In certain provinces India Korea Self Pay Taiwan Self Pay South Africa Canada For select programs significant patient co-pay is required

Summary • On-going trials are expected to characterize key patient populations • Next Generation device features are designed provide ‒ Control ‒ Sizing Flexibility ‒ LAA Seal ‒ Healing • Reimbursement has been secured in some countries, but access still limited in some regions
- Slides: 20