KYOTO LACC and beyond David Milliken Consultant In

KYOTO LACC and beyond David Milliken Consultant In Gynaecological Oncology Musgrove Park Hospital, Taunton, Somerset, UK

• Background to LACC • 2007 early cx cancer treatment was rad hyst • Potential Adverse events from this were significant ? avoidable • Min invasive surgery blossoming • Retrospective studies suggested these were lower in laparoscopic and robotic surgery and there was some data suggesting oncological outcomes were similar

• Reduction in blood loss periop comps hospital stay lower wound related complications • Retrospective data showing no difference in 5 yr disease free and overall survival • Both for robotic and laparoscopic

• At that time 2 large endometrials • LAP 2 in united states • LACE in Australia • suggesting that Min access was safe beneficial • Though survival outcomes were pending

• Extending in to cervix cancer was logical • Andreas Obermair and Pedro Ramirez developed trial together • LACC started 2008 • Difficulties in recruiting: Clinicians felt it was a retrograde step, patients were aware of minimal access and were demanding it

• Laparoscopic radical hysterectomy shows reduction in blood loss, postoperative complications, and hospital stay compared to open approach. No significant difference in 5 -year DFS and OS. (N=1, 539) • • Robotic radical hysterectomy is associated with less blood loss, lower transfusion rates, lower wound related complications, and shorter hospital stay compared to open radical hysterectomy. (N=4, 013) • • Wang Y, Deng L, Xu H, Zhang Y, Liang Z. BMC Cancer 2015 Shazly S, Murad M, Dowdy S, Gostout B, Famuyida A. Gyn Oncol 2016 Disease recurrence and survival not different between robotic radical hysterectomy and open radical hysterectomy. (N=491) • Sert BM, Boggess JF, Ahmad S, Jackson AL, Stavitzski NM, Dahl AA, Holloway RW EJSO 2016

NCCN guidelines 1. 2018 ESGO Guidelines Radical surgery by a gynaecological oncologist is the preferred treatment modality. Minimal invasive approach is favored.

• Phase III Randomized Trial of Laparoscopic or Robotic Radical Hysterectomy vs. Abdominal Radical Hysterectomy in Patients with Early. Stage Cervical Cancer: • LACC Trial

• Primary Objective • LACC Trial • Compare disease-free survival at 4. 5 years amongst patients who underwent a total laparoscopic or robotic radical hysterectomy (TLRH/TRRH) vs. a total abdominal radical hysterectomy (TARH) for early stage cervical cancer.

• Secondary Objectives • Compare patterns of recurrence between arms • Compare treatment-associated morbidity (6 months from surgery) • Compare the cost effectiveness of TLRH/TRRH vs. TARH • Assess pelvic floor function • Compare overall survival between arms • Determine the feasibility of sentinel lymph node mapping • Quality of Life (Qo. L) between arms

• Study Design • International, multicenter, randomized, phase III trial to test for non- inferiority of TLRH/TRRH vs. standard care (TARH) • Therefore, the primary intent to demonstrate that minimally invasive surgery was within 7. 2% of the DFS rate of the standard care (TARH) arm • Test for non-inferiority was based upon a 97. 5% one-sided confidence interval. Based on exponential survival times, for a 4. 5 -year follow-up, a total of 740 patients (370 per arm) was determined to have at least 90% power for non-inferiority.

• Inclusion Criteria • Confirmed primary squamous cell carcinoma, adenocarcinoma, or adenosquamous carcinoma of the uterine cervix • FIGO stage IA 1 (with LVSI), IA 2, or IB 1 • Type II or III radical hysterectomy (Piver-Rutledge Classification) • Performance status of ECOG 0 -1 • Age 18 years or older • Signed an approved Informed Consent

• June 2017 – safety signal from Data safety management committee. Recruiting paused • Further data accrual – safety signal worse • Trial halted.
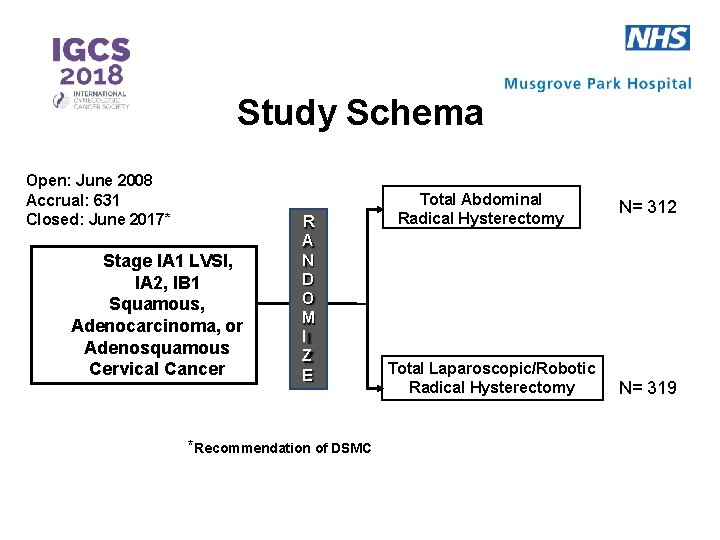
Study Schema Open: June 2008 Accrual: 631 Closed: June 2017* Stage IA 1 LVSI, IA 2, IB 1 Squamous, Adenocarcinoma, or Adenosquamous Cervical Cancer R A N D O M I Z E *Recommendation of DSMC Total Abdominal Radical Hysterectomy Total Laparoscopic/Robotic Radical Hysterectomy N= 312 N= 319
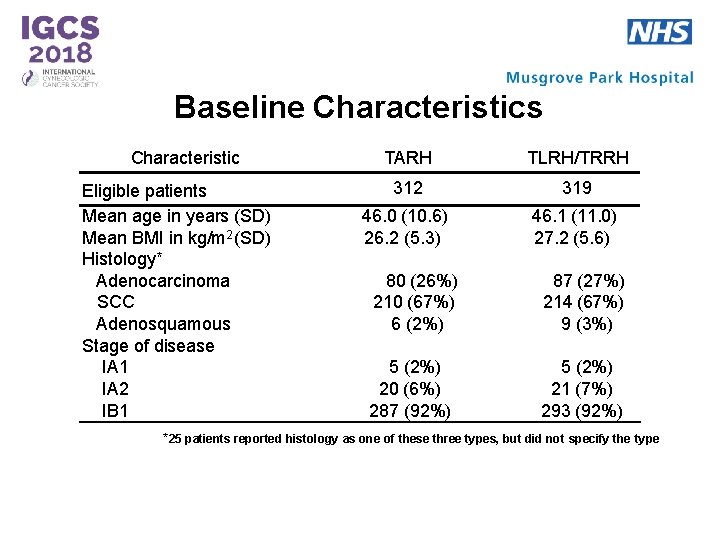
Baseline Characteristics Characteristic Eligible patients Mean age in years (SD) Mean BMI in kg/m 2 (SD) Histology* Adenocarcinoma SCC Adenosquamous Stage of disease IA 1 IA 2 IB 1 TARH TLRH/TRRH 312 319 46. 0 (10. 6) 26. 2 (5. 3) 46. 1 (11. 0) 27. 2 (5. 6) 80 (26%) 210 (67%) 6 (2%) 87 (27%) 214 (67%) 9 (3%) 5 (2%) 20 (6%) 287 (92%) 5 (2%) 21 (7%) 293 (92%) *25 patients reported histology as one of these three types, but did not specify the type
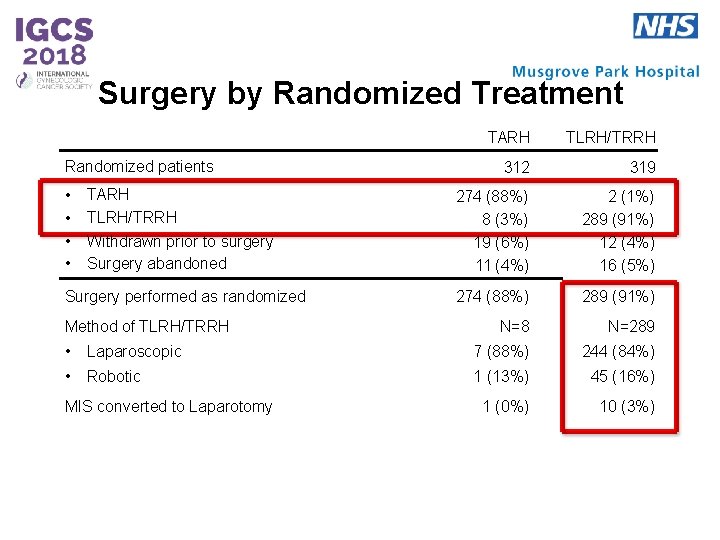
Surgery by Randomized Treatment Randomized patients • • TARH TLRH/TRRH Withdrawn prior to surgery Surgery abandoned Surgery performed as randomized Method of TLRH/TRRH TARH TLRH/TRRH 312 319 274 (88%) 8 (3%) 19 (6%) 11 (4%) 2 (1%) 289 (91%) 12 (4%) 16 (5%) 274 (88%) 289 (91%) N=8 N=289 • Laparoscopic 7 (88%) 244 (84%) • Robotic 1 (13%) 45 (16%) 1 (0%) 10 (3%) MIS converted to Laparotomy
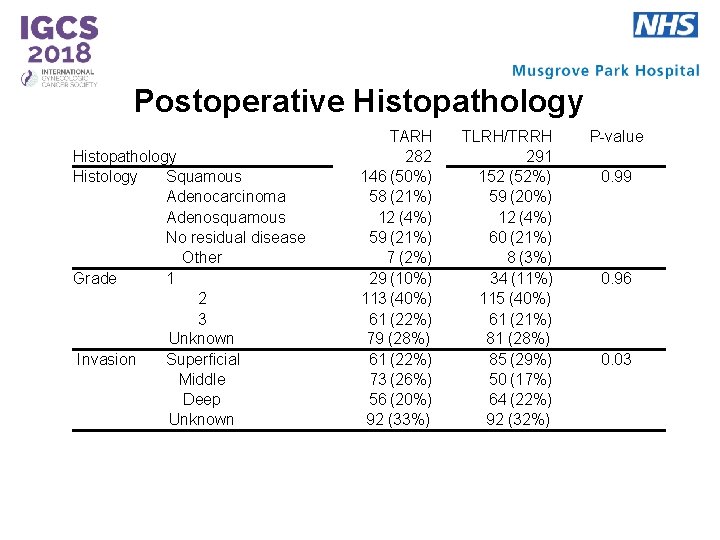
Postoperative Histopathology Histology Squamous Adenocarcinoma Adenosquamous No residual disease Other Grade 1 2 3 Unknown Invasion Superficial Middle Deep Unknown TARH 282 146 (50%) 58 (21%) 12 (4%) 59 (21%) 7 (2%) 29 (10%) 113 (40%) 61 (22%) 79 (28%) 61 (22%) 73 (26%) 56 (20%) 92 (33%) TLRH/TRRH 291 152 (52%) 59 (20%) 12 (4%) 60 (21%) 8 (3%) 34 (11%) 115 (40%) 61 (21%) 81 (28%) 85 (29%) 50 (17%) 64 (22%) 92 (32%) P-value 0. 99 0. 96 0. 03
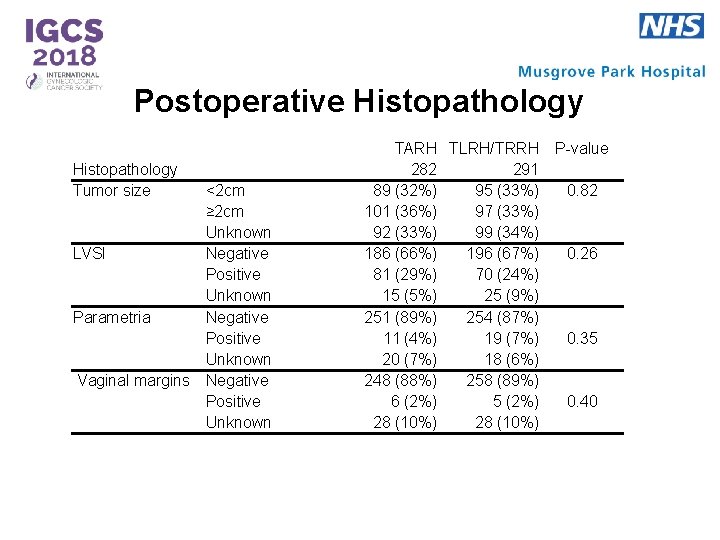
Postoperative Histopathology Tumor size <2 cm ≥ 2 cm Unknown LVSI Negative Positive Unknown Parametria Negative Positive Unknown Vaginal margins Negative Positive Unknown TARH TLRH/TRRH P-value 282 291 89 (32%) 95 (33%) 0. 82 101 (36%) 97 (33%) 92 (33%) 99 (34%) 186 (66%) 196 (67%) 0. 26 81 (29%) 70 (24%) 15 (5%) 25 (9%) 251 (89%) 254 (87%) 11 (4%) 19 (7%) 0. 35 20 (7%) 18 (6%) 248 (88%) 258 (89%) 6 (2%) 5 (2%) 0. 40 28 (10%)
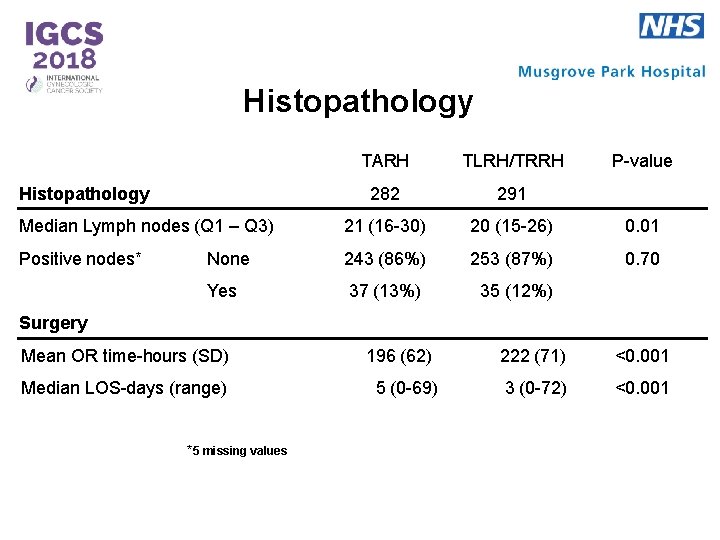
Histopathology TARH TLRH/TRRH 282 291 Median Lymph nodes (Q 1 – Q 3) 21 (16 -30) 20 (15 -26) 0. 01 Positive nodes* None 243 (86%) 253 (87%) 0. 70 Yes 37 (13%) 35 (12%) Histopathology P-value Surgery Mean OR time-hours (SD) Median LOS-days (range) *5 missing values 196 (62) 5 (0 -69) 222 (71) <0. 001 3 (0 -72) <0. 001
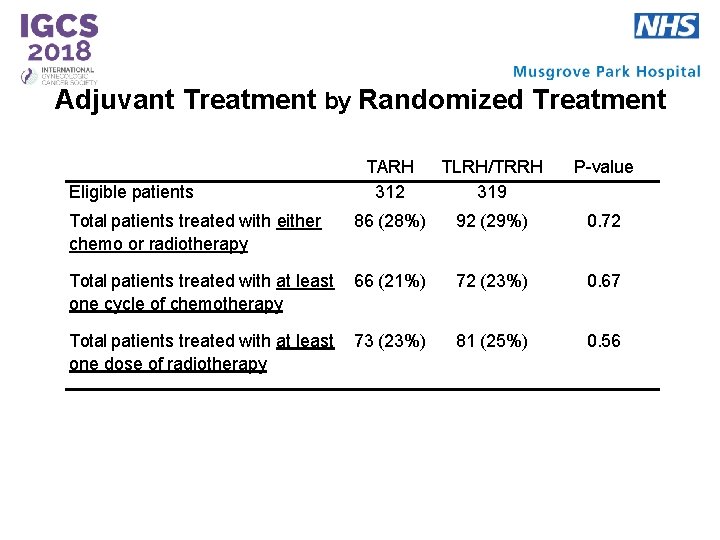
Adjuvant Treatment by Randomized Treatment TARH 312 TLRH/TRRH 319 P-value Total patients treated with either chemo or radiotherapy 86 (28%) 92 (29%) 0. 72 Total patients treated with at least one cycle of chemotherapy 66 (21%) 72 (23%) 0. 67 Total patients treated with at least one dose of radiotherapy 73 (23%) 81 (25%) 0. 56 Eligible patients

Data Completeness Primary outcome (DFS) Median Follow-up time (min- max) 2. 5 years (0. 0 - 6. 3) Completeness* at 4. 5 years (%) 219/558 (39. 2%) Information available at 4. 5 years (%) 59. 7% Overall survival Median Follow-up time (min- max) 2. 5 years (0. 0 - 6. 3) Completeness* at 4. 5 years (%) 208/558 (37. 3%) Information available at 4. 5 years (%) 54. 3% *Completeness is proportion of patients with the event of interest, or with follow-up to 4. 5 years, out of the total patients that we can achieve data at 4. 5 years (excluding withdrawals and LTFU)
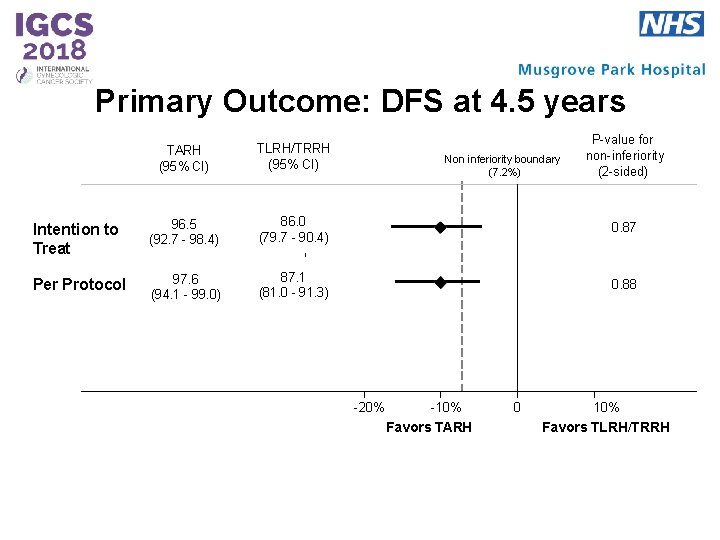
Primary Outcome: DFS at 4. 5 years P-value for non-inferiority (2 -sided) TARH (95% CI) TLRH/TRRH (95% CI) Intention to Treat 96. 5 (92. 7 - 98. 4) 86. 0 (79. 7 - 90. 4) 0. 87 Per Protocol 97. 6 (94. 1 - 99. 0) 87. 1 (81. 0 - 91. 3) 0. 88 Non inferiority boundary (7. 2%) -20% -10% Favors TARH 0 10% Favors TLRH/TRRH
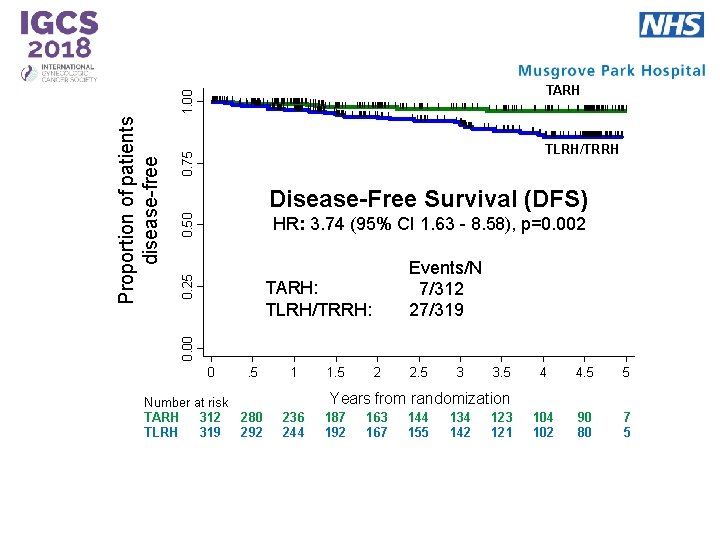
0. 75 TLRH/TRRH 0. 50 Disease-Free Survival (DFS) 0. 25 HR: 3. 74 (95% CI 1. 63 - 8. 58), p=0. 002 Events/N 7/312 27/319 TARH: TLRH/TRRH: 0. 00 Proportion of patients disease-free 1. 00 TARH 0 Number at risk TARH 312 TLRH 319 . 5 1 1. 5 2 2. 5 3 3. 5 4 4. 5 5 104 102 90 80 7 5 Years from randomization 280 292 236 244 187 192 163 167 144 155 134 142 123 121
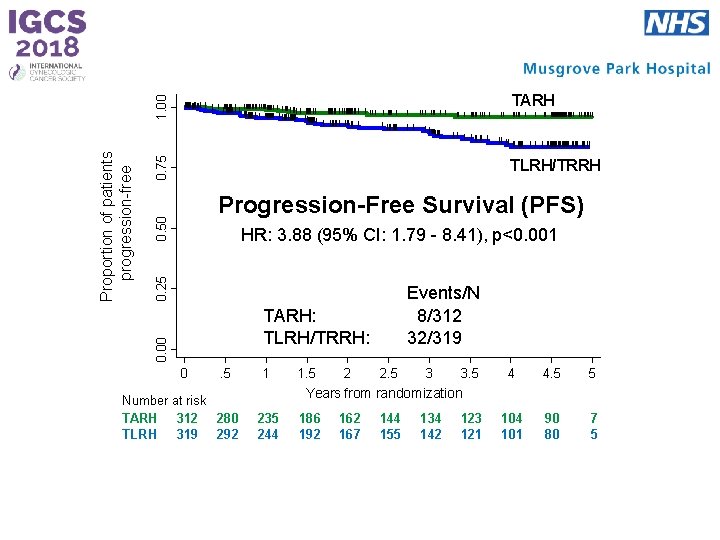
1. 00 0. 75 TLRH/TRRH 0. 50 Progression-Free Survival (PFS) 0. 25 HR: 3. 88 (95% CI: 1. 79 - 8. 41), p<0. 001 Events/N 8/312 32/319 TARH: TLRH/TRRH: 0. 00 Proportion of patients progression-free TARH 0 . 5 Number at risk TARH 312 280 TLRH 319 292 1 1. 5 2 2. 5 3 3. 5 4 4. 5 5 104 101 90 80 7 5 Years from randomization 235 244 186 192 167 144 155 134 142 123 121
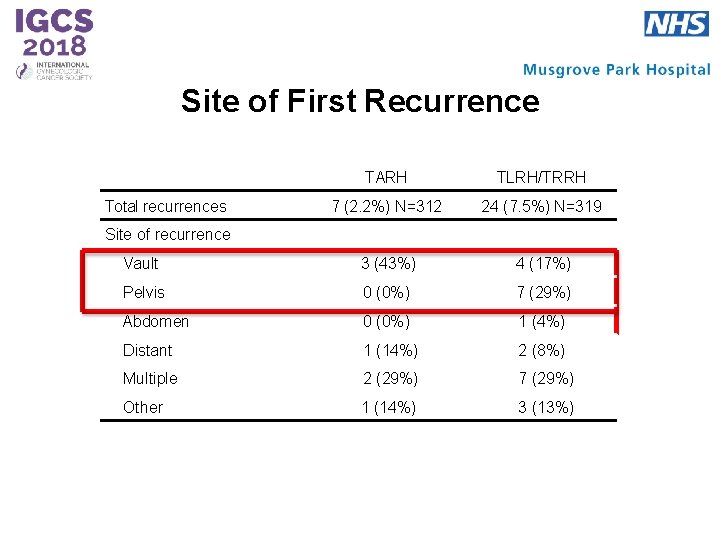
Site of First Recurrence TARH TLRH/TRRH 7 (2. 2%) N=312 24 (7. 5%) N=319 Vault 3 (43%) 4 (17%) Pelvis 0 (0%) 7 (29%) Abdomen 0 (0%) 1 (4%) Distant 1 (14%) 2 (8%) Multiple 2 (29%) 7 (29%) Other 1 (14%) 3 (13%) Total recurrences Site of recurrence
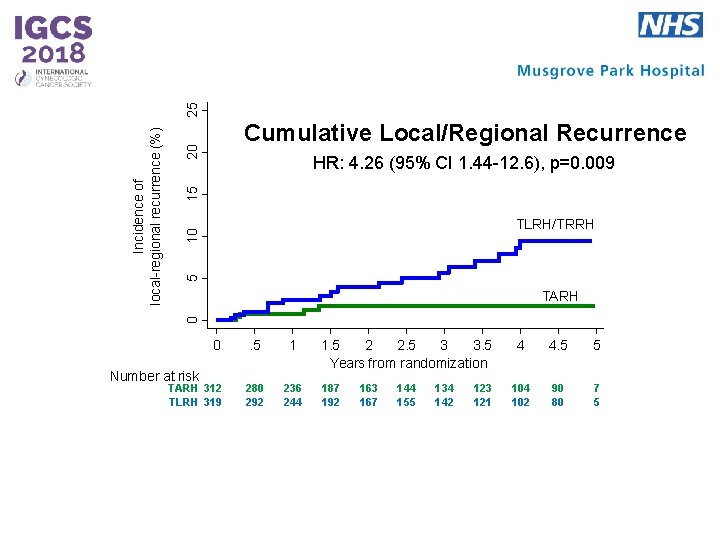
25 20 15 HR: 4. 26 (95% CI 1. 44 -12. 6), p=0. 009 5 10 TLRH/TRRH TARH 0 Incidence of local-regional recurrence (%) Cumulative Local/Regional Recurrence 0 Number at risk TARH 312 TLRH 319 . 5 1 280 292 236 244 1. 5 2 2. 5 3 3. 5 Years from randomization 187 192 163 167 144 155 134 142 123 121 4 4. 5 5 104 102 90 80 7 5
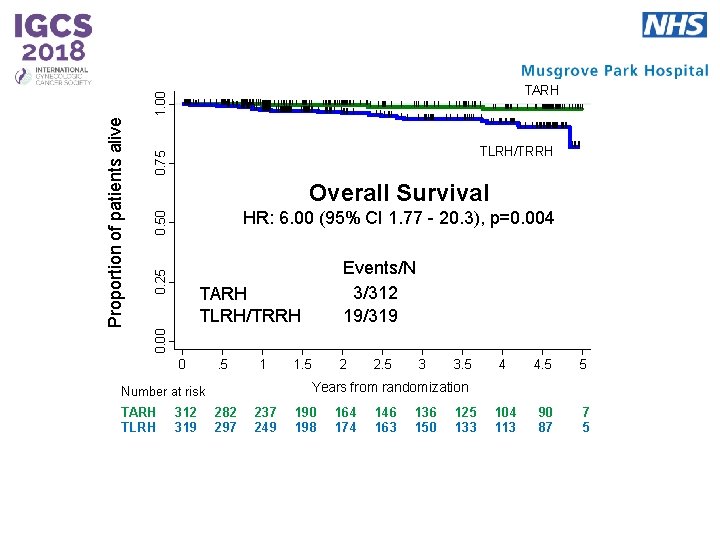
0. 75 TLRH/TRRH Overall Survival 0. 25 0. 50 HR: 6. 00 (95% CI 1. 77 - 20. 3), p=0. 004 Events/N 3/312 19/319 TARH TLRH/TRRH 0. 00 Proportion of patients alive 1. 00 TARH 0 . 5 1 312 319 2 2. 5 3 3. 5 4 4. 5 5 104 113 90 87 7 5 Years from randomization Number at risk TARH TLRH 1. 5 282 297 237 249 190 198 164 174 146 163 136 150 125 133
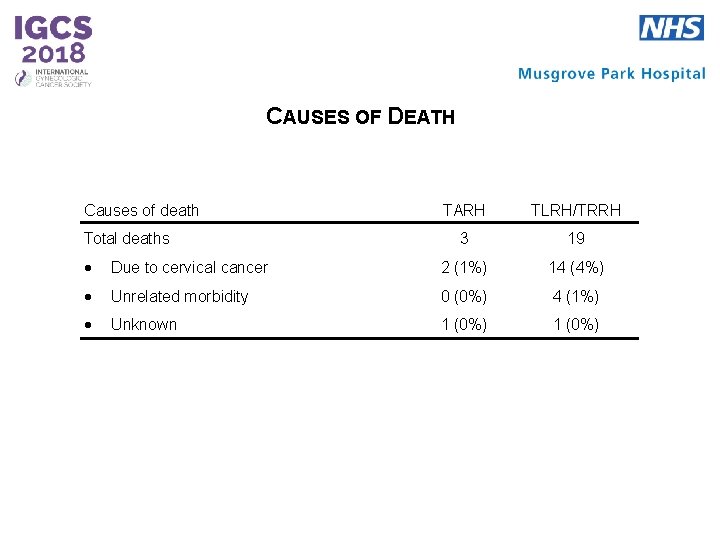
CAUSES OF DEATH Causes of death Total deaths TARH TLRH/TRRH 3 19 Due to cervical cancer 2 (1%) 14 (4%) Unrelated morbidity 0 (0%) 4 (1%) Unknown 1 (0%)
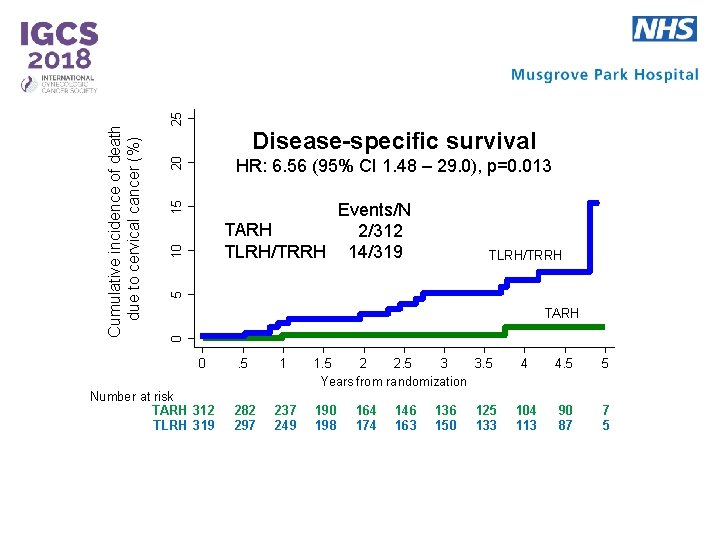
25 20 HR: 6. 56 (95% CI 1. 48 – 29. 0), p=0. 013 TLRH/TRRH 5 10 15 Events/N TARH 2/312 TLRH/TRRH 14/319 TARH 0 Cumulative incidence of death due to cervical cancer (%) Disease-specific survival 0 Number at risk TARH 312 TLRH 319 . 5 1 282 297 237 249 3. 5 1. 5 2 2. 5 3 Years from randomization 190 198 164 174 146 163 136 150 125 133 4 4. 5 5 104 113 90 87 7 5

Conclusions • Disease-free survival at 4. 5 years for minimally invasive radical hysterectomy was inferior compared to the open approach • Minimally invasive radical hysterectomy was associated with higher rates of loco/regional recurrences • Results of the LACC Trial should be discussed with patients scheduled to undergo radical hysterectomy

LACC Trial • Strengths -Largest prospective randomized trial -Multicenter & international collaboration -Surgeon proficiency requirements -Powered to evaluate oncologic outcomes -Recurrence Adjudication Committee • Limitations -Early termination -Lack of central pathology review -Data maturity

Rauh-Hain Retrospective study National cancer database Same population Ø 2000 patients Ø Minimal access vs open Ø Survival of 1 a 2 or 1 b 1 Ø Similar findings plus at the point where minimal access surgery introduced in USA there was a drop in survival

Why is LACC different? • Most retrospective data is focusing on intra operative or immediate post op comparisons, lack of focus on oncological outcomes. Unbalanced groups • Looked back at their data to 1990 • Concurrent vs sequential • Comparison is not in same timeframe • Early groups, selection criteria different, higher rate of 1 B 2 tumours, imaging inferior ie PET CT MRI modern ct etc not readily available therefore higher rate of positive margins higher rate of parametrial involvement. • If adjuvant treatment subsequently required – radiation only • The recent group much more sophisticated pre op assessment, better adjuvant therapies

Why is LACC different? Post operative follow up time of MIS is less than the open for obvious reasons The retrospective MIS data is very similar to the MIS arm of LACC Recent study comparing Robotic surgery to open radical hysterectomy No difference in recurrence rate, but recurrence rate was higher than LACC MIS data More bulky tumours less microscopic tumours LVSI and parametrial invasion more prevelant in open group.

Responses to editorials • MIS access arm recruited 2 patients per year ? All surgeons sufficiently experienced 319 divided by 33 divided by 9 yr period. Accrual over time increases • MIS worse in lacc not because its bad but open did so well that it made MIS look bad. Disease free survival is of open in lacc 96. 5 , only 4 studies in recent metanalysis mentioned survival other data all between 93. 3 93. 6 94. 4 for open. • Uterine manipulators, approach to and type of colpotomy not in data but is in op notes • Missing data • Ethnicity of surgeon…. ? ? !!!



- Slides: 38