Ksrleti mdszerek Experimental techniques in reaction kinetics Techniques


- Slides: 24

Kísérleti módszerek Experimental techniques in reaction kinetics

Techniques classified Batch reactors vs. flow reactors Online methods: real-time in situ detection Offline methods: — aliquot from the batch — “quenched flow” Pseudo-first order (isolation or flooding) method (only if relative concentrations are available) Unbiased; higher precision Biased; lower precision

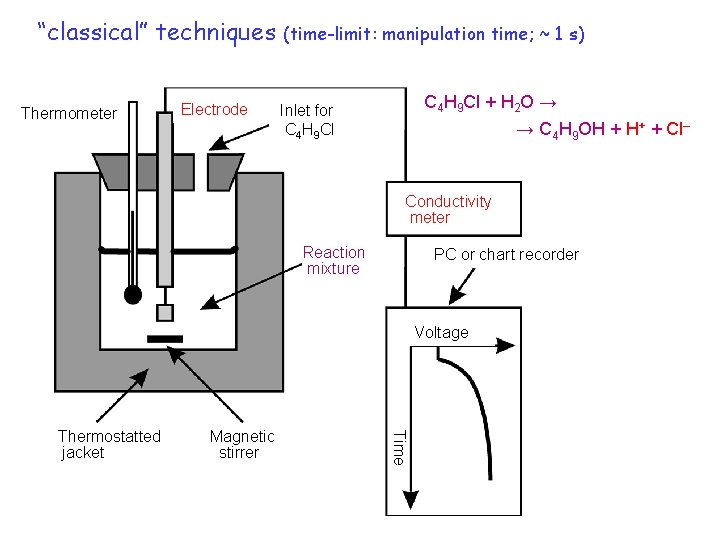
“classical” techniques Thermometer Electrode (time-limit: manipulation time; ~ 1 s) C 4 H 9 Cl + H 2 O → Inlet for C 4 H 9 Cl → C 4 H 9 OH + H+ + Cl– Conductivity meter Reaction mixture PC or chart recorder Voltage Magnetic stirrer Time Thermostatted jacket

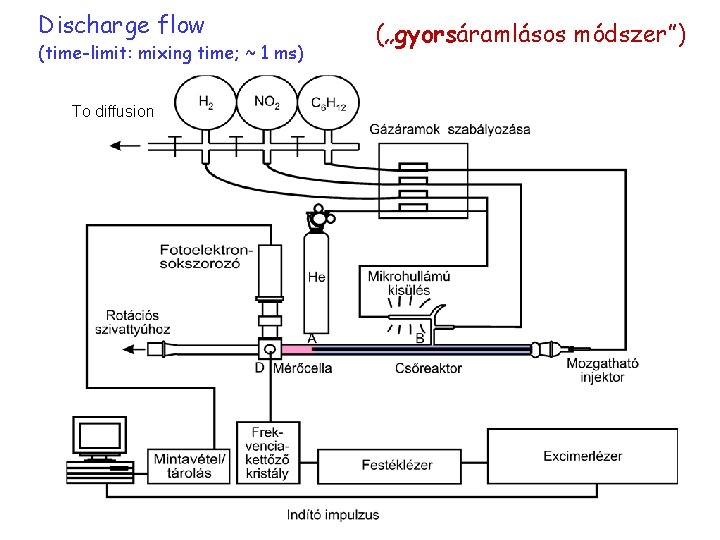
Discharge flow (time-limit: mixing time; ~ 1 ms) To diffusion („gyorsáramlásos módszer”)

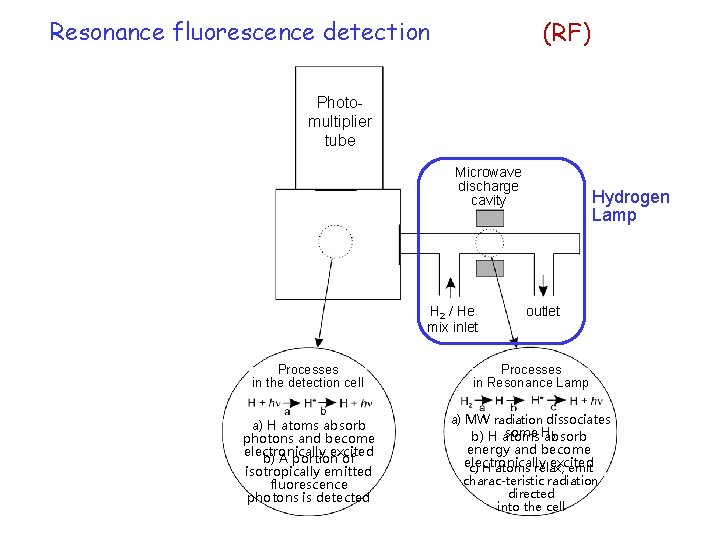
Resonance fluorescence detection (RF) Photomultiplier tube Microwave discharge cavity H 2 / He mix inlet Hydrogen Lamp outlet Processes in the detection cell Processes in Resonance Lamp a) H atoms absorb photons and become electronically excited b) A portion of isotropically emitted fluorescence photons is detected a) MW radiation dissociates some H 2 b) H atoms absorb energy and become electronically excited c) H atoms relax, emit charac-teristic radiation directed into the cell

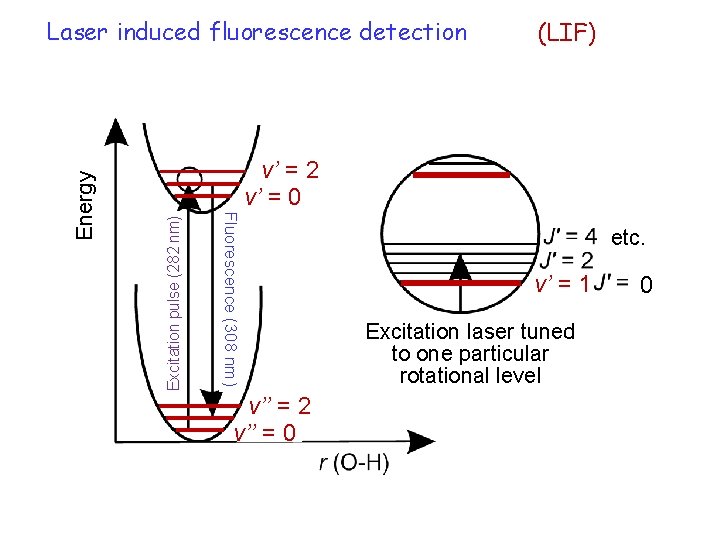
(LIF) Excitation pulse (282 nm) v’ = 2 v’ = 0 Fluorescence (308 nm) Energy Laser induced fluorescence detection v’’ = 2 v’’ = 0 etc. v’ = 1 Excitation laser tuned to one particular rotational level 0

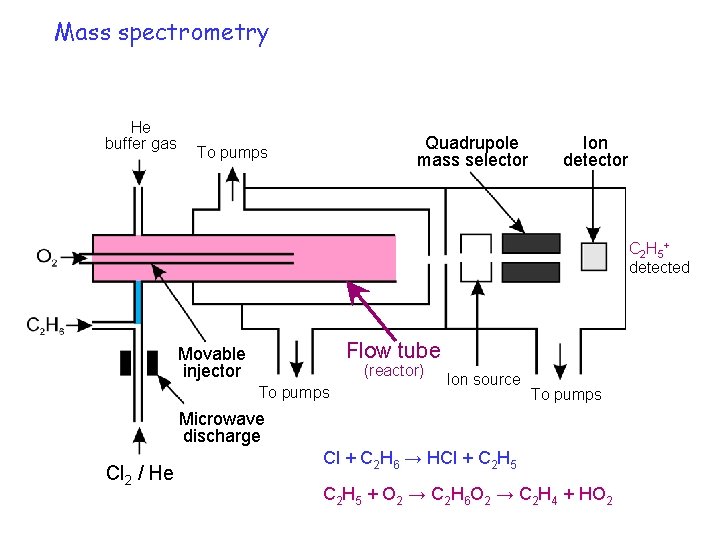
Mass spectrometry He buffer gas Quadrupole mass selector To pumps Ion detector C 2 H 5 + detected Flow tube Movable injector (reactor) To pumps Ion source To pumps Microwave discharge Cl 2 / He Cl + C 2 H 6 → HCl + C 2 H 5 + O 2 → C 2 H 6 O 2 → C 2 H 4 + HO 2

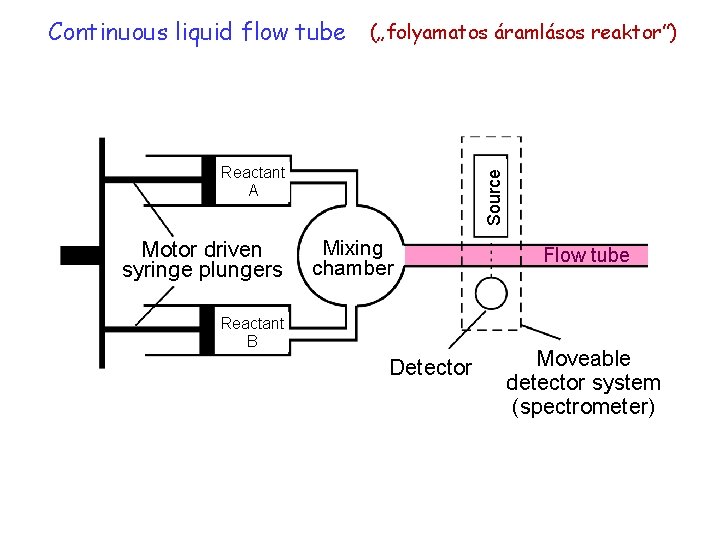
Continuous liquid flow tube („folyamatos áramlásos reaktor”) Motor driven syringe plungers Source Reactant A Mixing chamber Reactant B Detector Flow tube Moveable detector system (spectrometer)

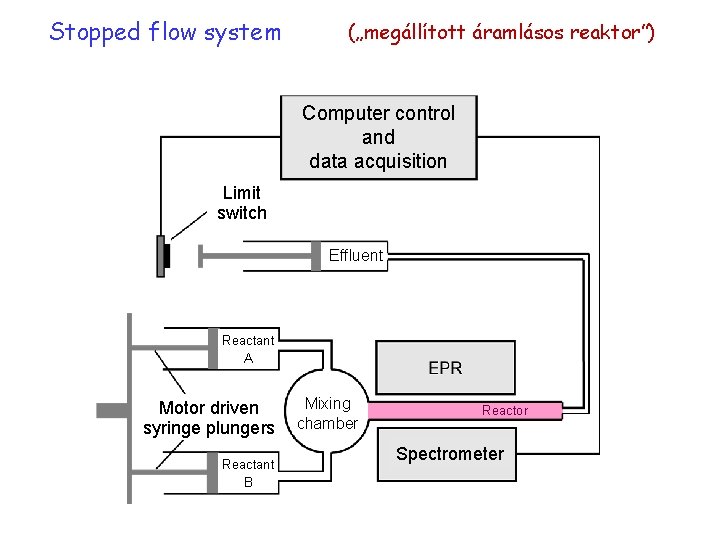
Stopped flow system („megállított áramlásos reaktor”) Computer control and data acquisition Limit switch Effluent Reactant A Motor driven syringe plungers Reactant B Mixing chamber Reactor Spectrometer

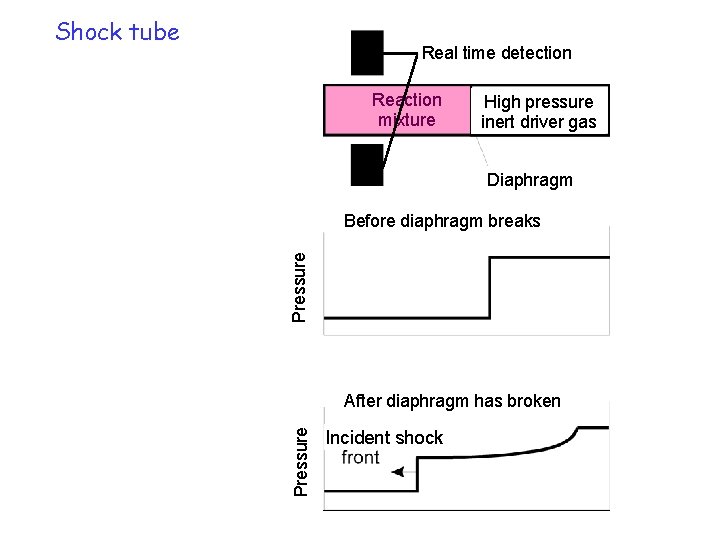
Shock tube Real time detection Reaction mixture High pressure inert driver gas Diaphragm Pressure Before diaphragm breaks Pressure After diaphragm has broken Incident shock

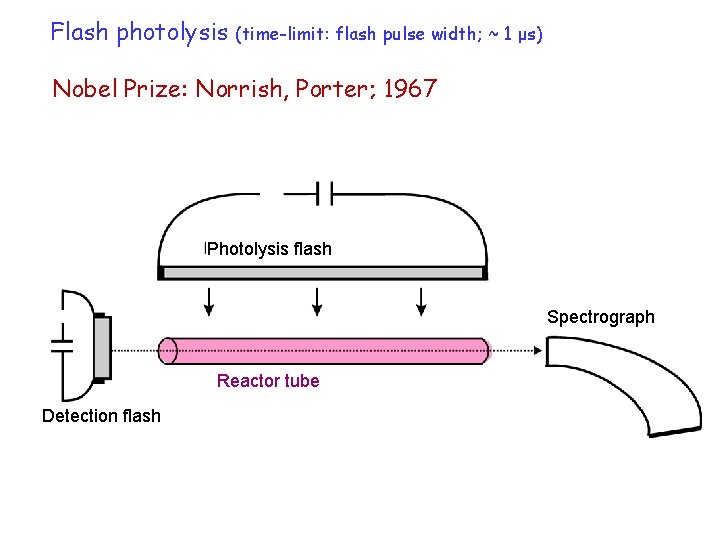
Flash photolysis (time-limit: flash pulse width; ~ 1 μs) Nobel Prize: Norrish, Porter; 1967 Photolysis flash Spectrograph Reactor tube Detection flash

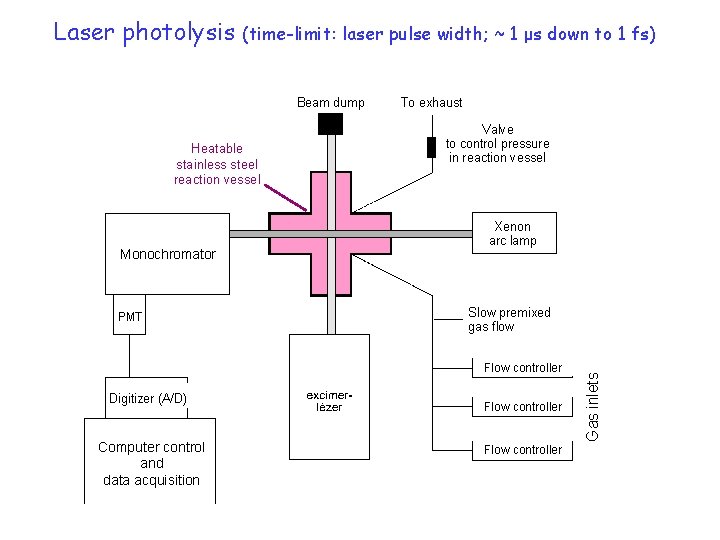
(time-limit: laser pulse width; ~ 1 μs down to 1 fs) Beam dump Heatable stainless steel reaction vessel Monochromator PMT To exhaust Valve to control pressure in reaction vessel Xenon arc lamp Slow premixed gas flow Flow controller Digitizer (A/D) Computer control and data acquisition Flow controller Gas inlets Laser photolysis

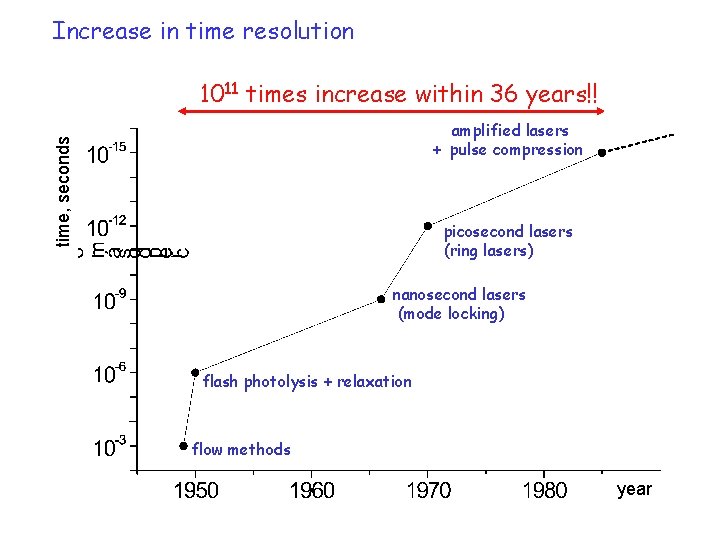
időfelbontás Increase in time resolution 1011 times increase within 36 years!! time, seconds amplified lasers + pulse compression picosecond lasers (ring lasers) nanosecond lasers (mode locking) flash photolysis + relaxation flow methods year

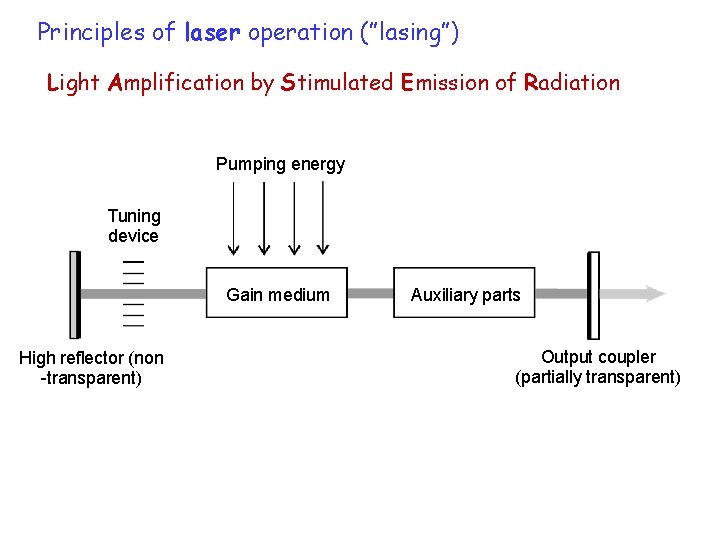
Principles of laser operation (”lasing”) Light Amplification by Stimulated Emission of Radiation Pumping energy Tuning device Gain medium High reflector (non -transparent) Auxiliary parts Output coupler (partially transparent)

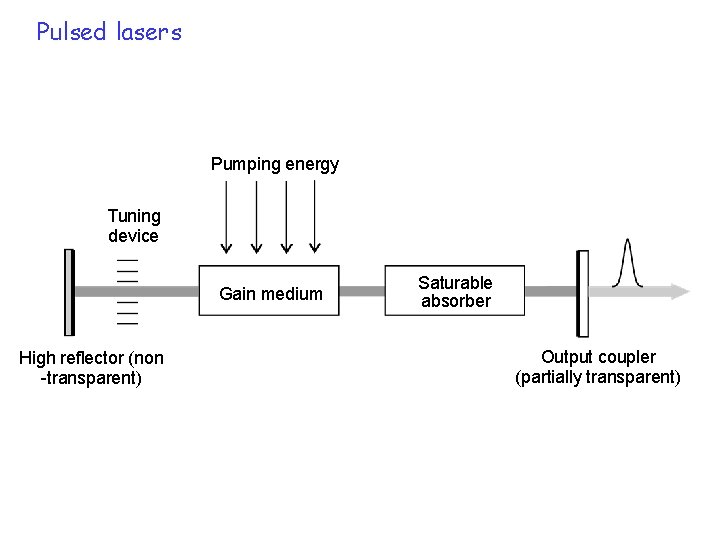
Pulsed lasers Pumping energy Tuning device Gain medium High reflector (non -transparent) Saturable absorber Output coupler (partially transparent)

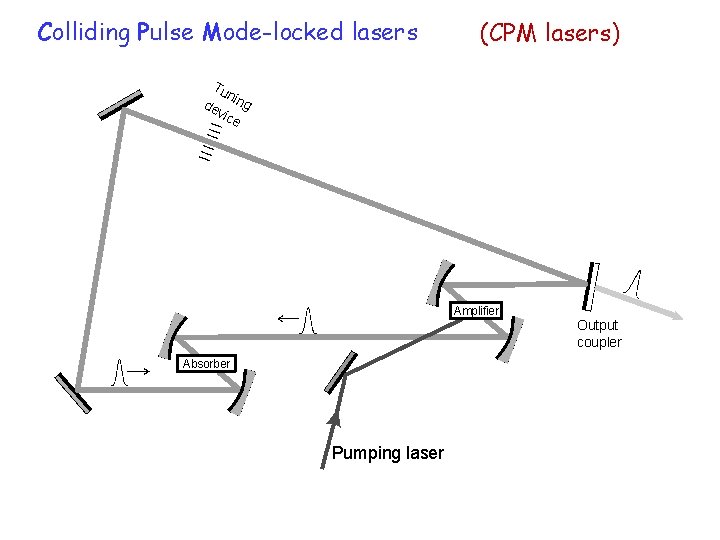
Colliding Pulse Mode-locked lasers (CPM lasers) Tu n de ing vic e Amplifier Output coupler Absorber Pumping laser

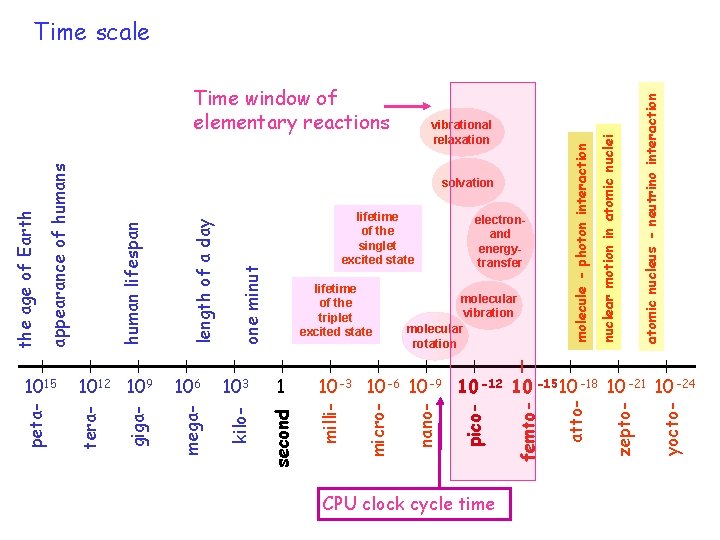
időskála 2 CPU clock cycle time atomic nucleus - neutrino interaction nuclear motion in atomic nuclei yocto- zepto- femto- pico- second 10 -3 10 -6 10 -9 10 -12 10 -15 10 -18 10 -21 10 -24 micro- 1 milli- 103 electronand energytransfer molecular vibration molecular rotation nano- one minut 106 kilo- length of a day 1012 109 giga- 1015 mega- lifetime of the triplet excited state tera- human lifespan lifetime of the singlet excited state atto- appearance of humans vibrational relaxation solvation peta- the age of Earth Time window of elementary reactions molecule - photon interaction Time scale

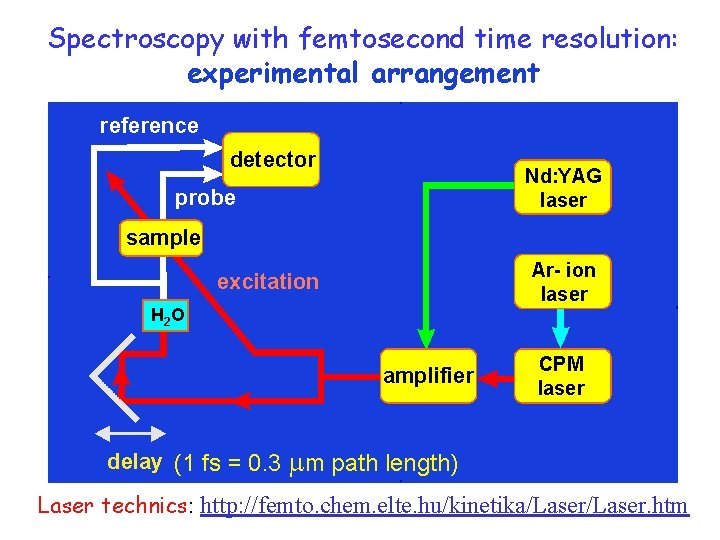
pump-probe Spectroscopy with femtosecond time resolution: experimental arrangement reference detector Nd: YAG laser probe sample Ar- ion laser excitation H 2 O amplifier delay (1 fs = 0. 3 CPM laser m path length) Laser technics: http: //femto. chem. elte. hu/kinetika/Laser. htm

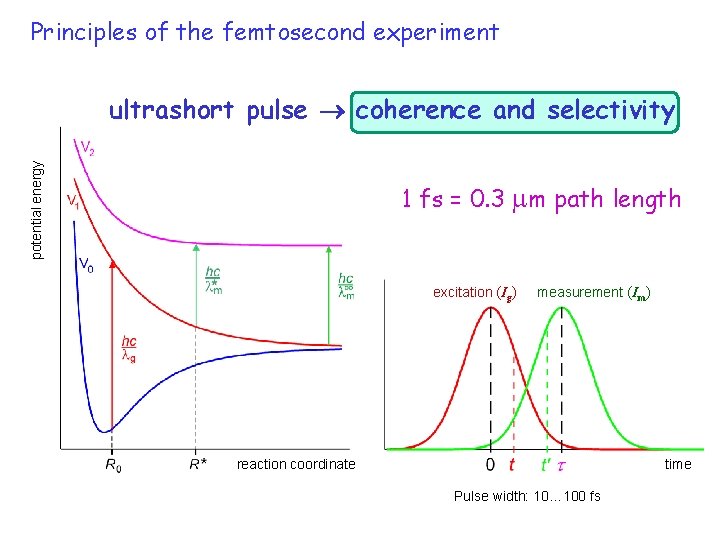
pump-probe 5 Principles of the femtosecond experiment potential energy ultrashort pulse coherence and selectivity 1 fs = 0. 3 m path length excitation (Ig) measurement (Im) reaction coordinate time Pulse width: 10… 100 fs

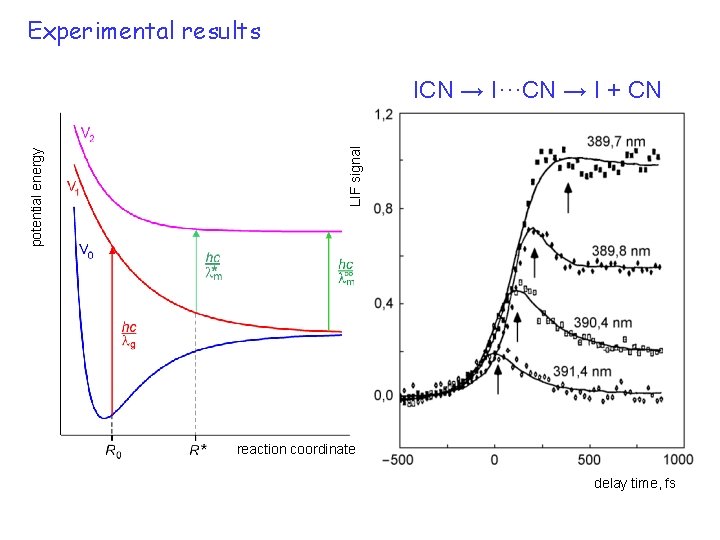
pump-probe 6 Experimental results LIF signal potential energy ICN → I···CN → I + CN reaction coordinate delay time, fs

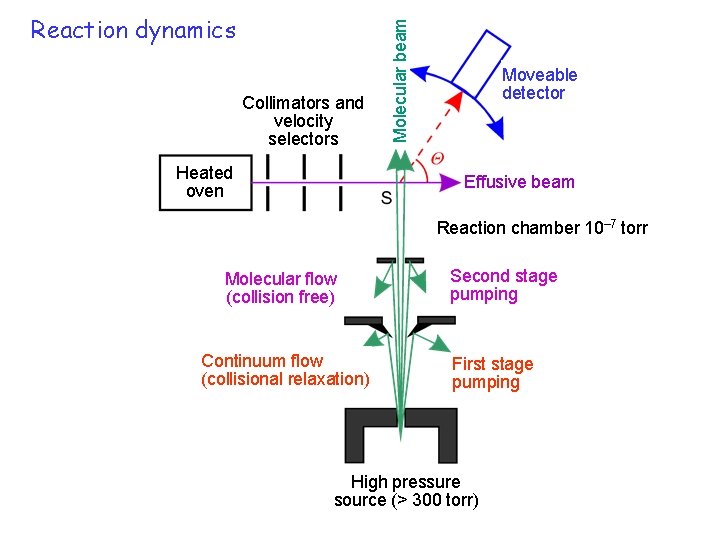
Collimators and velocity selectors Heated oven Molecular beam Reaction dynamics Moveable detector Effusive beam Reaction chamber 10– 7 torr Molecular flow (collision free) Continuum flow (collisional relaxation) Second stage pumping First stage pumping High pressure source (> 300 torr)

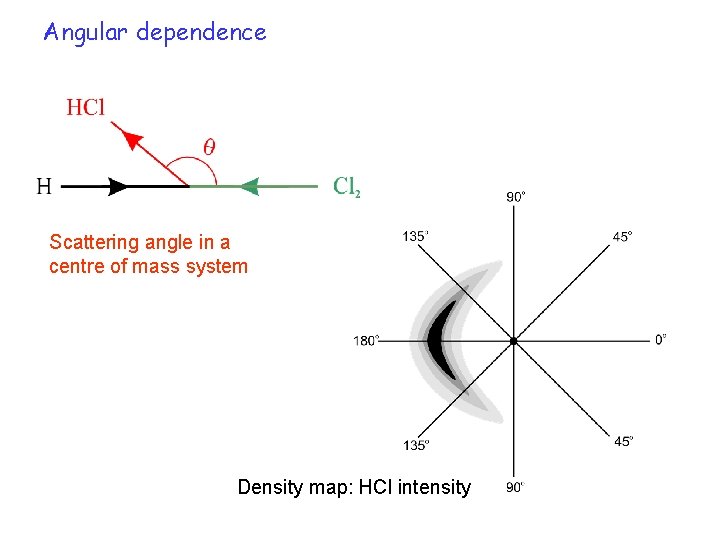
Angular dependence Scattering angle in a centre of mass system Density map: HCl intensity

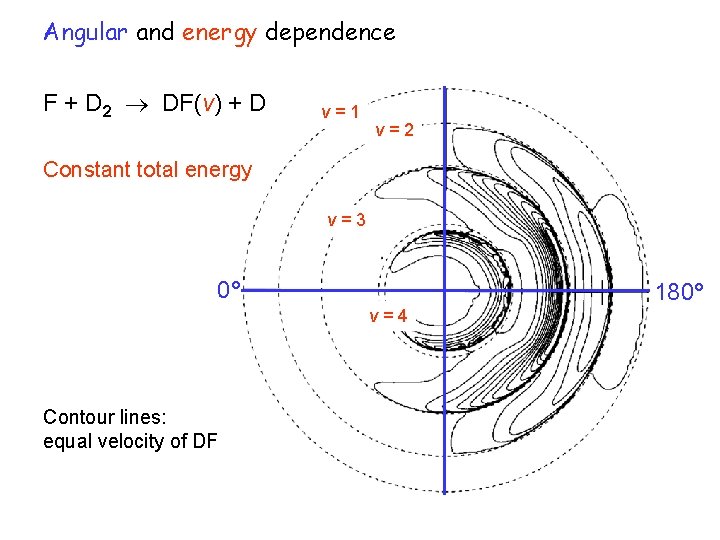
Angular and energy dependence F + D 2 DF(v) + D v=1 v=2 Constant total energy v=3 0° 180° v=4 Contour lines: equal velocity of DF

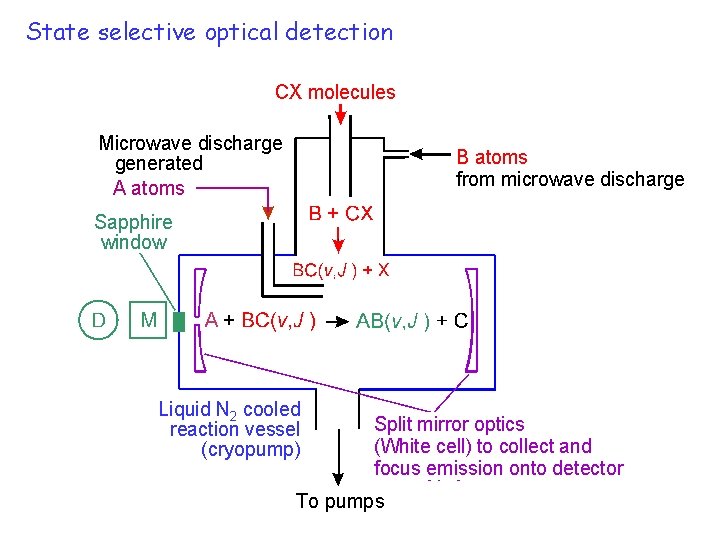
State selective optical detection CX molecules Microwave discharge generated A atoms B atoms from microwave discharge Sapphire window Liquid N 2 cooled reaction vessel (cryopump) Split mirror optics (White cell) to collect and focus emission onto detector To pumps