KS 4 Particles and energy Boardworks Ltd 2003

KS 4 Particles and energy © Boardworks Ltd 2003
Teacher’s Notes A slide contains teacher’s notes wherever this icon is displayed To access these notes go to ‘Notes Page View’ (Power. Point 97) or ‘Normal View’ (Power. Point 2000). Notes Page View Normal View Flash Files A flash file has been embedded into the Power. Point slide wherever this icon is displayed – These files are not editable. © Boardworks Ltd 2003
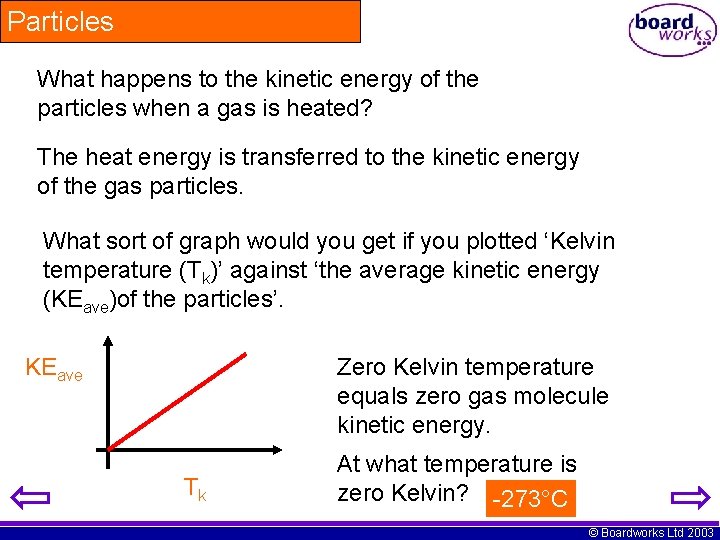
Particles What happens to the kinetic energy of the particles when a gas is heated? The heat energy is transferred to the kinetic energy of the gas particles. What sort of graph would you get if you plotted ‘Kelvin temperature (Tk)’ against ‘the average kinetic energy (KEave)of the particles’. KEave Zero Kelvin temperature equals zero gas molecule kinetic energy. Tk At what temperature is zero Kelvin? -273°C © Boardworks Ltd 2003
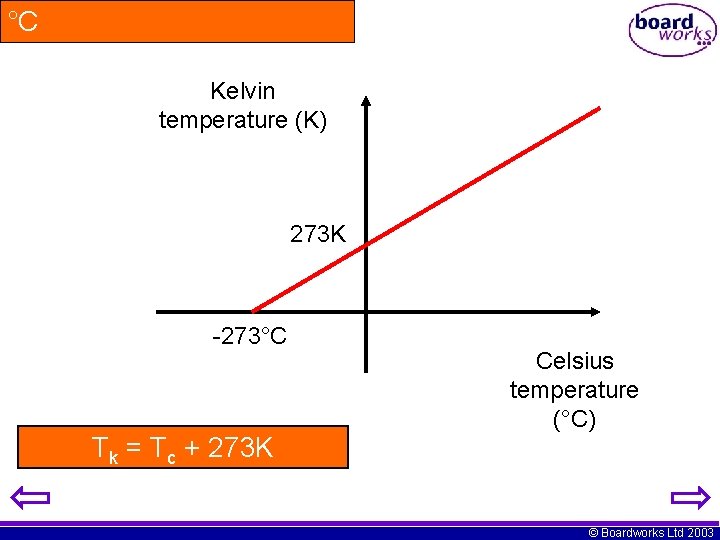
°C Kelvin temperature (K) 273 K -273°C Tk = Tc + 273 K Celsius temperature (°C) © Boardworks Ltd 2003
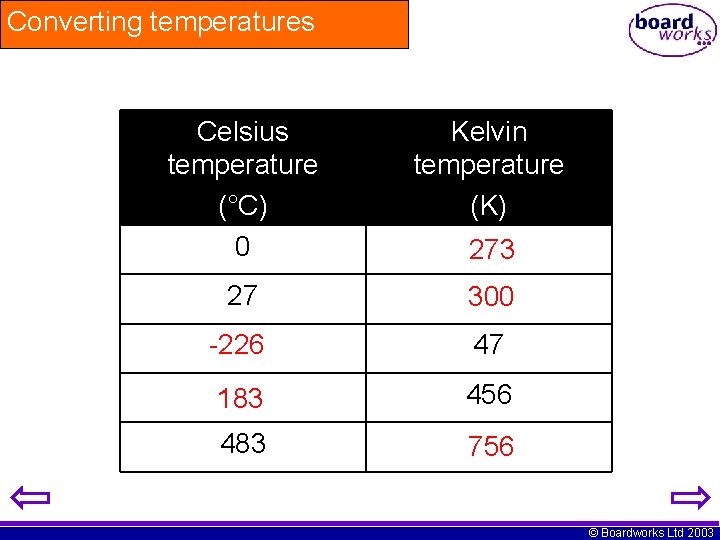
Converting temperatures Celsius temperature (°C) 0 Kelvin temperature (K) 27 300 -226 47 183 456 483 756 273 © Boardworks Ltd 2003

Pressure and particles What causes pressure in gases? In a sealed container, the particles repeatedly strike the walls of the container. This causes pressure. What happens if you increase the temperature? more pressure gas gets hotter more kinetic energy more collisions at greater speed © Boardworks Ltd 2003
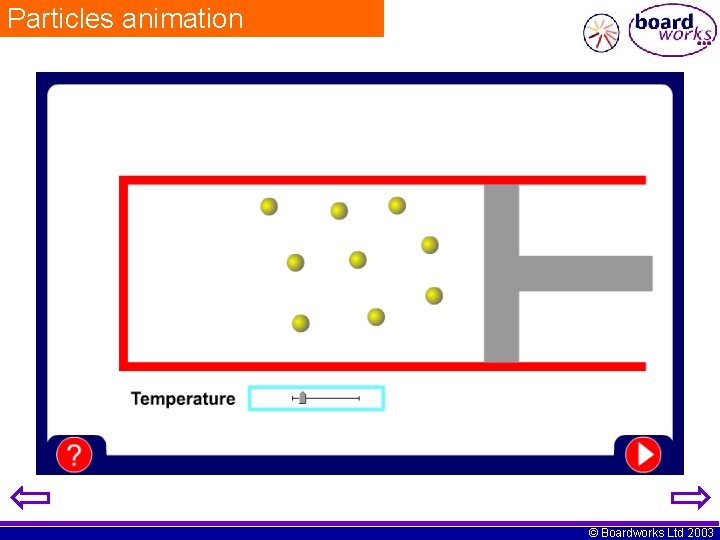
Particles animation © Boardworks Ltd 2003
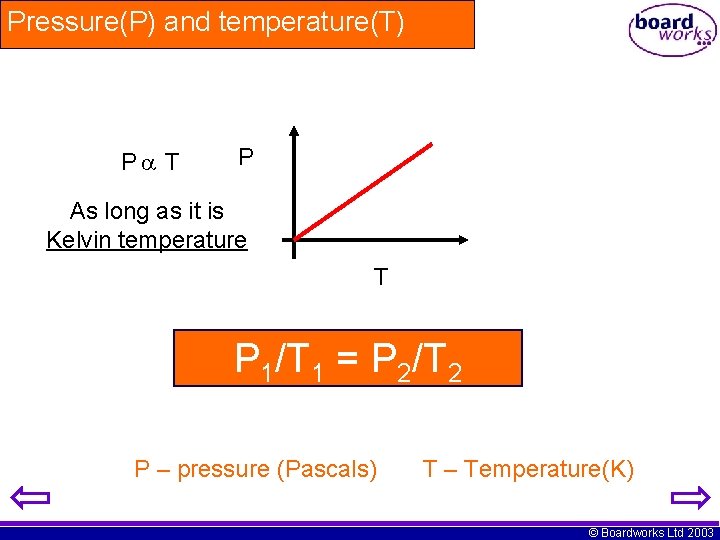
Pressure(P) and temperature(T) P T P As long as it is Kelvin temperature T P 1/T 1 = P 2/T 2 P – pressure (Pascals) T – Temperature(K) © Boardworks Ltd 2003

A gas at a pressure of 50 Pa and a temperature of 40 K is heated to a temperature of 120 K in a sealed container. What is the new pressure of the gas? P 1/T 1 = P 2/T 2 P 2= T 2(P 1/T 1) P 2= 120 x(50/40) P 2= 150 Pa © Boardworks Ltd 2003

A gas at a pressure of 200 Pa and a temperature of 140 K is heated to an unknown temperature in a sealed container. The new pressure is 6 000 Pa. What is the new temperature of the gas? P 1/T 1 = P 2/T 2 T 2= P 2(T 1/P 1) T 2= 6 000 x(140/200) T 2= 4 200 Pa © Boardworks Ltd 2003

Einstein During nuclear reactions mass can be changed into energy. This happens in stars and nuclear reactors, and nuclear fission bombs. The amount of energy produced as mass is changed into energy is given by the formula: Energy = mass change x (speed of light)2 E = mc 2 Energy – J Mass – Kg Speed of light – ms -2 © Boardworks Ltd 2003

A mass of 2 Kg is completely converted into energy. The speed of light is 3 x 108 ms-1. How much energy is released? E = mc 2 E = 2 x(3 x 108)2 E = 1. 8 x 1017 J © Boardworks Ltd 2003
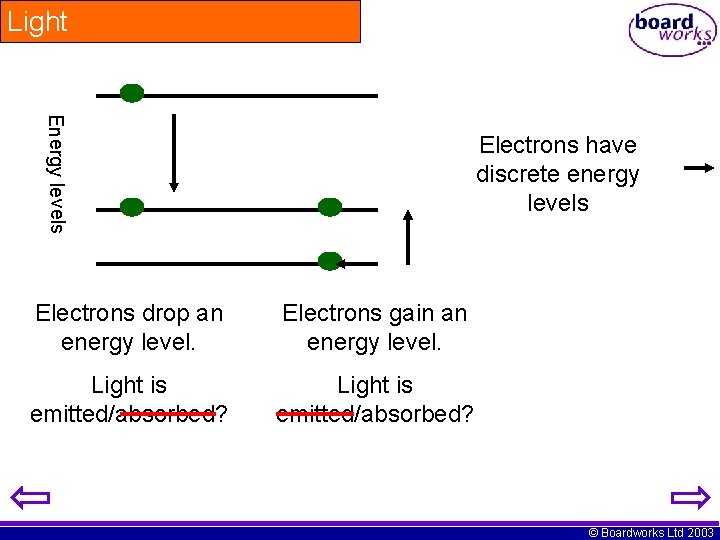
Light Energy levels Electrons have discrete energy levels Electrons drop an energy level. Electrons gain an energy level. Light is emitted/absorbed? © Boardworks Ltd 2003
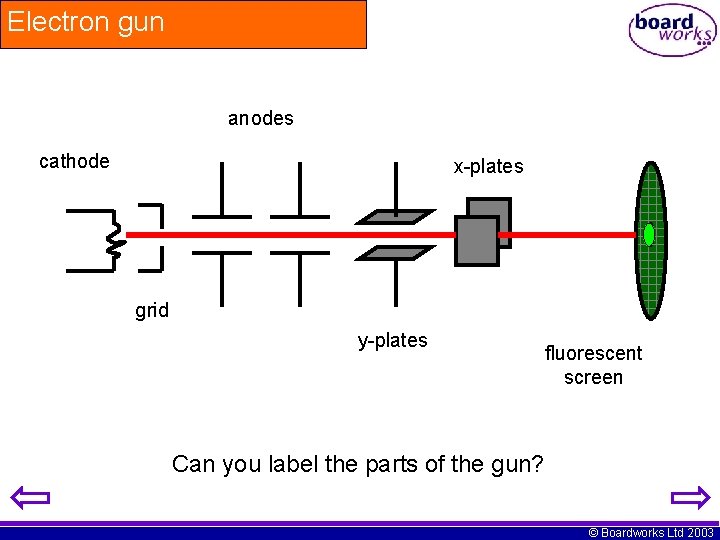
Electron gun anodes cathode x-plates grid y-plates fluorescent screen Can you label the parts of the gun? © Boardworks Ltd 2003
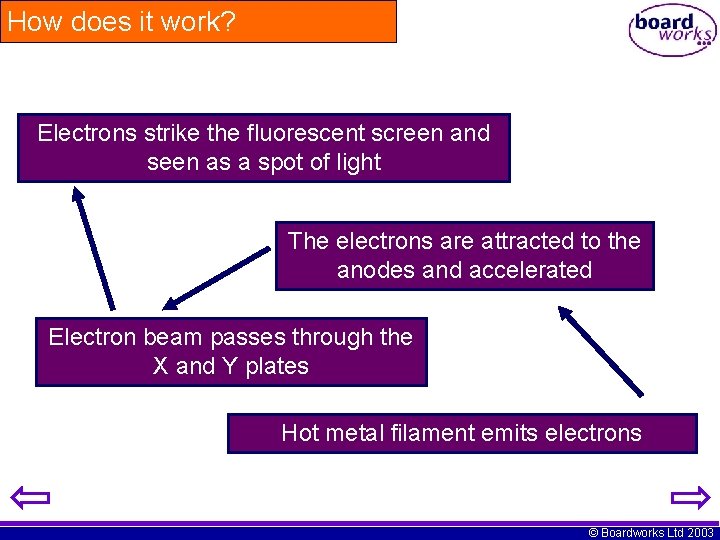
How does it work? Electrons strike the fluorescent screen and seen as a spot of light The electrons are attracted to the anodes and accelerated Electron beam passes through the X and Y plates Hot metal filament emits electrons © Boardworks Ltd 2003

KE=QV You can treat the electron beam as a flow of electrons: Kinetic energy = Charge x Voltage KE=QV Kinetic energy measured in Joules (J) Charge measured in Coulombs (C) Voltage measured in volts (V) © Boardworks Ltd 2003
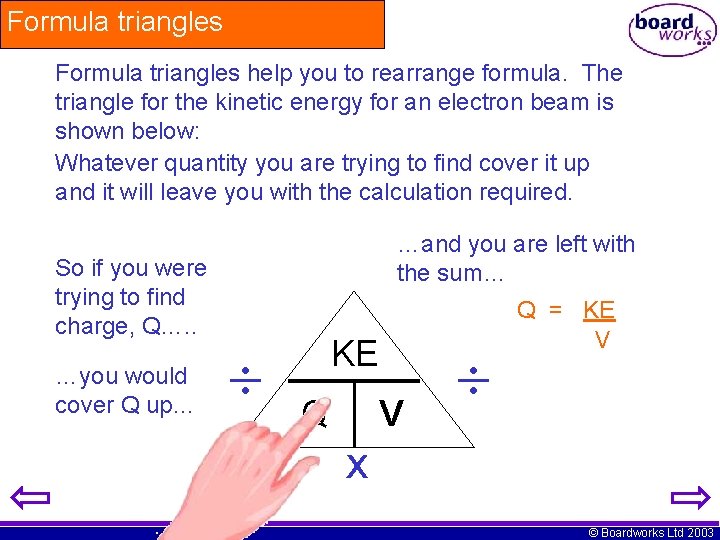
Formula triangles help you to rearrange formula. The triangle for the kinetic energy for an electron beam is shown below: Whatever quantity you are trying to find cover it up and it will leave you with the calculation required. So if you were trying to find charge, Q…. . …you would cover Q up… …and you are left with the sum… KE Q V Q = KE V x © Boardworks Ltd 2003
Formula triangles © Boardworks Ltd 2003

Uses of electron beams? Oscilloscopes TV tubes Electroplating Monitors X ray production Combustion engines © Boardworks Ltd 2003

What are the units of pressure? A. Metres B. Joules C. Newtons D. Pascals © Boardworks Ltd 2003

What is 225 K in Celsius temperature? A. -48°C B. 48°C C. 498°C D. -498°C © Boardworks Ltd 2003

What is 333°C in Kelvin temperature? A. 606 K B. -606 K C. 60 K D. -60 K © Boardworks Ltd 2003

A gas at a pressure of 60 Pa at a temperature of 50 K in a sealed container is compressed to a pressure of 480 Pa. What is the new temperature of the gas? A. 8 K B. 80 K C. 6. 2 K D. 400 K © Boardworks Ltd 2003

Which of the following is not a use of electron beams? A. Oscilloscopes B. Monitors C. Spray painting D. X ray production © Boardworks Ltd 2003
- Slides: 24