KOMPOSISI DAN SIFAT BAHAN PANGAN Komponen Alami Komponen
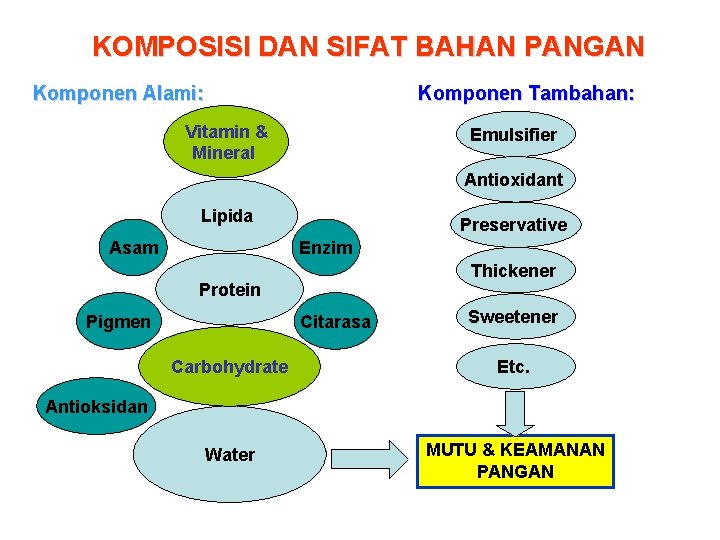
KOMPOSISI DAN SIFAT BAHAN PANGAN Komponen Alami: Komponen Tambahan: Vitamin & Mineral Emulsifier Antioxidant Lipida Asam Preservative Enzim Thickener Protein Pigmen Citarasa Sweetener Carbohydrate Etc. Water MUTU & KEAMANAN PANGAN Antioksidan

KARBOHIDRAT 1. 2. 3. 4. 5. PENDAHULUAN KARBOHIDRAT DALAM BAHAN PANGAN KLASIFIKASI DAN STRUKTUR KIMIA REAKSI KIMIA KARBOHIDRAT SIFAT FUNGSIONAL KARBOHIDRAT

1. PENDAHULUAN • 75 % of Biological World • 80 % of the Calorie Intake of humankind Ø 75 – 80 % starch • In USA : Composition of Calorie Intake Ø Carbohydrate : 46% • 47 % Starch • 52% Sucrose Ø Fats : 42% Ø Protein : 12% • Rumus Umum : Cx(H 2 O)y • Modifikasi struktur Sifat Fungsional

2. KARBOHIDRAT DALAM BAHAN PANGAN Karbohidrat utama dalam : • animal : glucose, glycogen • milk : lactose • plant : cellulose, starch • seaweed: alginate, carrageenan, agar
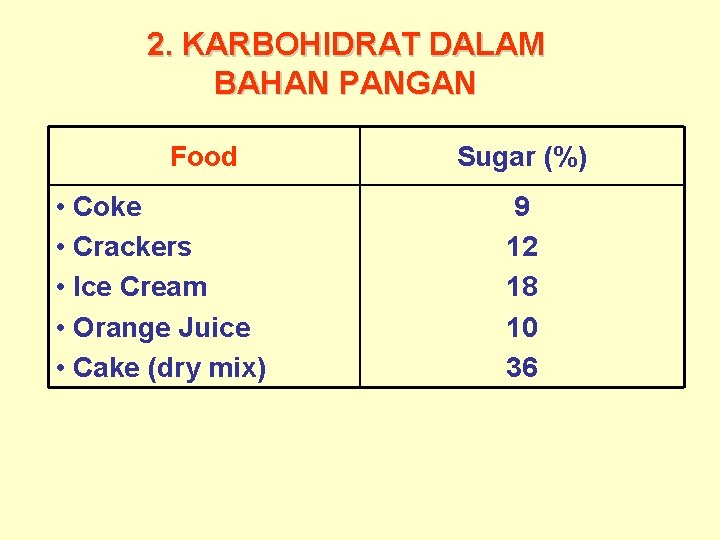
2. KARBOHIDRAT DALAM BAHAN PANGAN Food • Coke • Crackers • Ice Cream • Orange Juice • Cake (dry mix) Sugar (%) 9 12 18 10 36
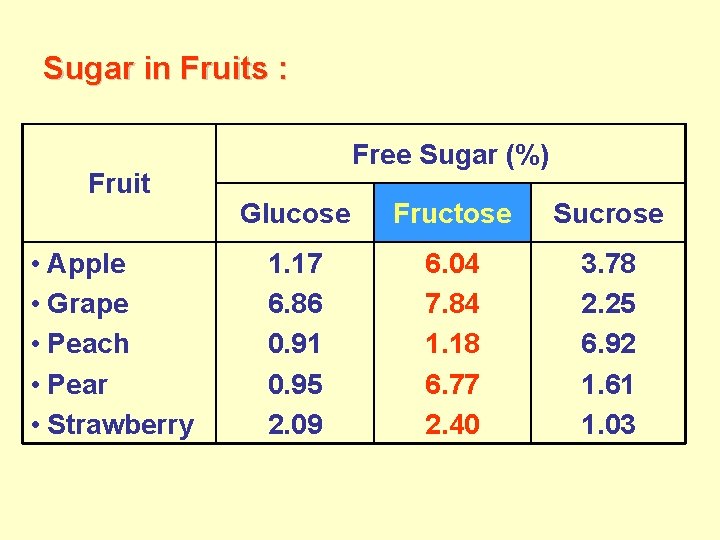
Sugar in Fruits : Fruit • Apple • Grape • Peach • Pear • Strawberry Free Sugar (%) Glucose Fructose Sucrose 1. 17 6. 86 0. 91 0. 95 2. 09 6. 04 7. 84 1. 18 6. 77 2. 40 3. 78 2. 25 6. 92 1. 61 1. 03
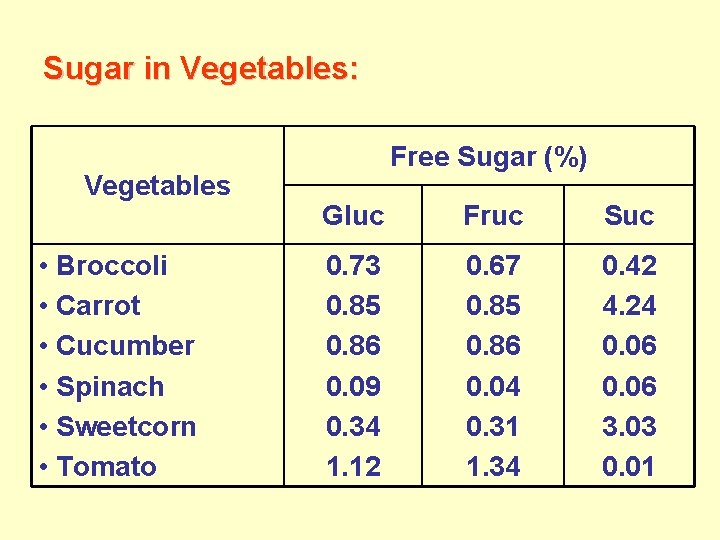
Sugar in Vegetables: Vegetables • Broccoli • Carrot • Cucumber • Spinach • Sweetcorn • Tomato Free Sugar (%) Gluc Fruc Suc 0. 73 0. 85 0. 86 0. 09 0. 34 1. 12 0. 67 0. 85 0. 86 0. 04 0. 31 1. 34 0. 42 4. 24 0. 06 3. 03 0. 01
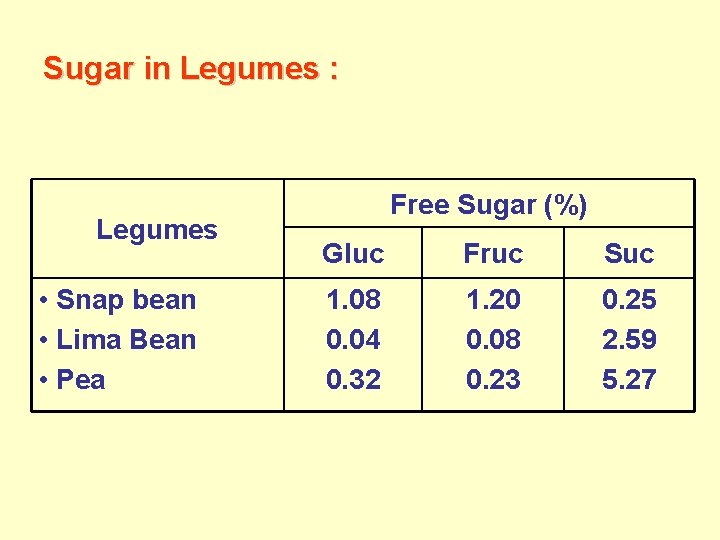
Sugar in Legumes : Legumes • Snap bean • Lima Bean • Pea Free Sugar (%) Gluc Fruc Suc 1. 08 0. 04 0. 32 1. 20 0. 08 0. 23 0. 25 2. 59 5. 27
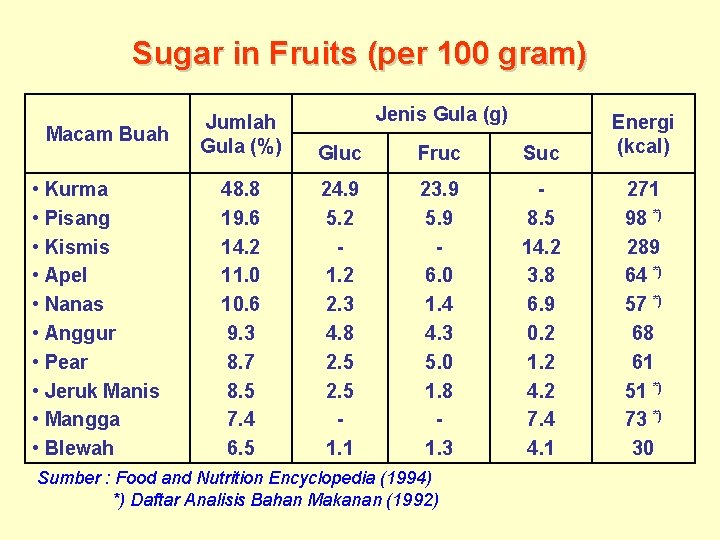
Sugar in Fruits (per 100 gram) Macam Buah • Kurma • Pisang • Kismis • Apel • Nanas • Anggur • Pear • Jeruk Manis • Mangga • Blewah Jenis Gula (g) Jumlah Gula (%) Gluc Fruc Suc Energi (kcal) 48. 8 19. 6 14. 2 11. 0 10. 6 9. 3 8. 7 8. 5 7. 4 6. 5 24. 9 5. 2 1. 2 2. 3 4. 8 2. 5 1. 1 23. 9 5. 9 6. 0 1. 4 4. 3 5. 0 1. 8 1. 3 8. 5 14. 2 3. 8 6. 9 0. 2 1. 2 4. 2 7. 4 4. 1 271 98 *) 289 64 *) 57 *) 68 61 51 *) 73 *) 30 Sumber : Food and Nutrition Encyclopedia (1994) *) Daftar Analisis Bahan Makanan (1992)
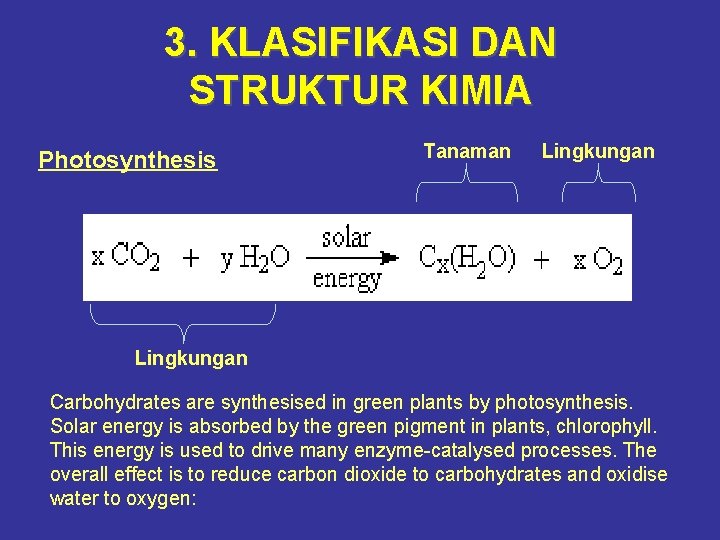
3. KLASIFIKASI DAN STRUKTUR KIMIA Photosynthesis Tanaman Lingkungan Carbohydrates are synthesised in green plants by photosynthesis. Solar energy is absorbed by the green pigment in plants, chlorophyll. This energy is used to drive many enzyme-catalysed processes. The overall effect is to reduce carbon dioxide to carbohydrates and oxidise water to oxygen:
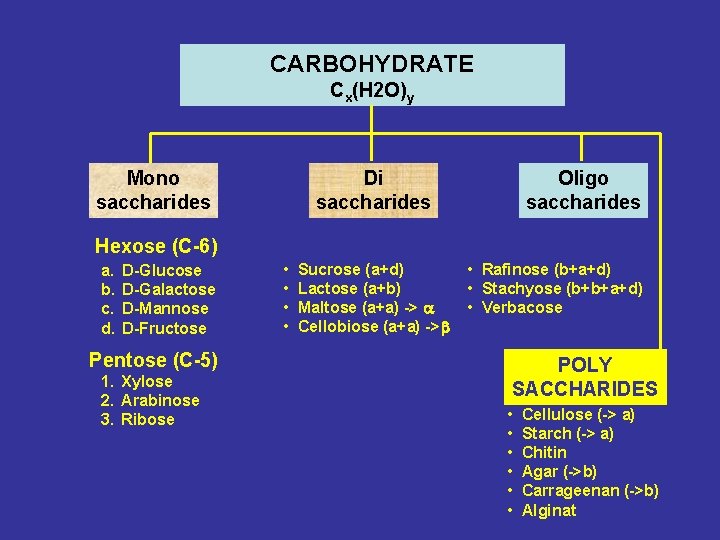
CARBOHYDRATE Cx(H 2 O)y Mono saccharides Di saccharides Oligo saccharides Hexose (C-6) a. b. c. d. D-Glucose D-Galactose D-Mannose D-Fructose Pentose (C-5) 1. Xylose 2. Arabinose 3. Ribose • • Sucrose (a+d) Lactose (a+b) Maltose (a+a) -> a Cellobiose (a+a) ->b • Rafinose (b+a+d) • Stachyose (b+b+a+d) • Verbacose POLY SACCHARIDES • • • Cellulose (-> a) Starch (-> a) Chitin Agar (->b) Carrageenan (->b) Alginat
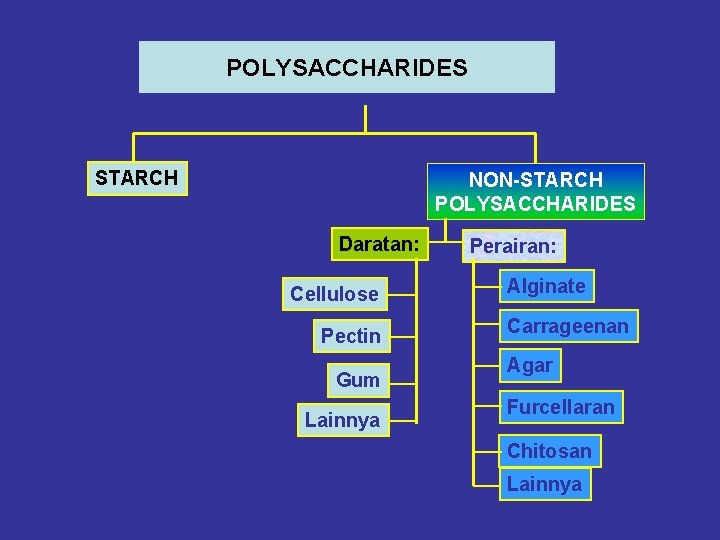
POLYSACCHARIDES STARCH NON-STARCH POLYSACCHARIDES Daratan: Cellulose Pectin Gum Lainnya Perairan: Alginate Carrageenan Agar Furcellaran Chitosan Lainnya
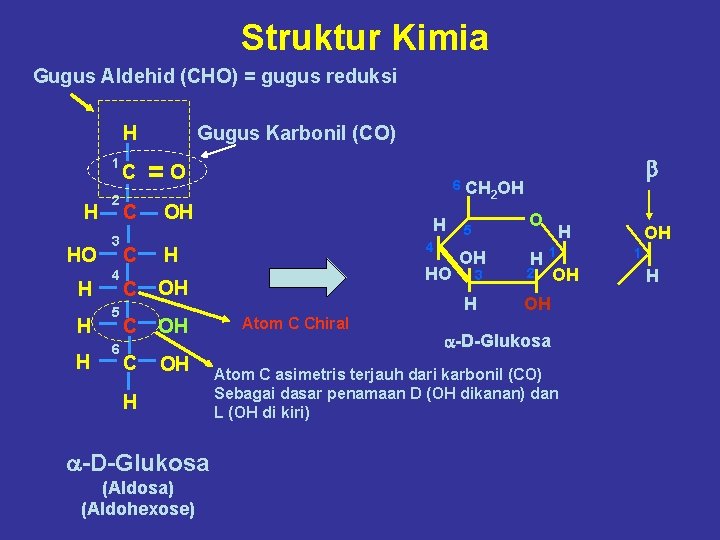
Struktur Kimia Gugus Aldehid (CHO) = gugus reduksi H 1 H HO H H H 2 3 4 5 6 C Gugus Karbonil (CO) =O C OH C OH H a-D-Glukosa (Aldosa) (Aldohexose) 6 CH H 4 HO 2 OH O 5 OH 3 H Atom C Chiral b H H 1 2 OH OH a-D-Glukosa Atom C asimetris terjauh dari karbonil (CO) Sebagai dasar penamaan D (OH dikanan) dan L (OH di kiri) OH 1 H
Struktur Kimia
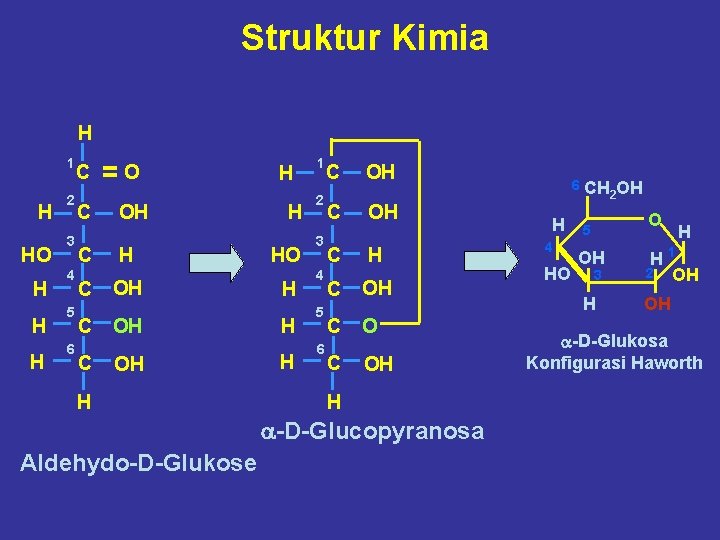
Struktur Kimia H 1 H HO H H H 2 3 4 5 6 C C C =O OH H OH OH OH H HO H H H 1 2 3 4 5 6 C OH C H C O C OH H a-D-Glucopyranosa Aldehydo-D-Glukose 6 CH H 4 HO 2 OH O 5 OH 3 H H H 1 2 OH OH a-D-Glukosa Konfigurasi Haworth

Struktur Kimia Gugus Aldehid (CHO) = gugus reduksi H 1 H HO H H H 2 3 4 5 6 C C C H Gugus Karbonil (CO) =O OH H OH OH OH H a-D-Glukosa (Aldosa) H H Reduksi • Na-Amalgam • Li-Al-hydride • hydrogenation HO H H H 1 2 3 4 5 6 C OH C H C OH H Sorbitol
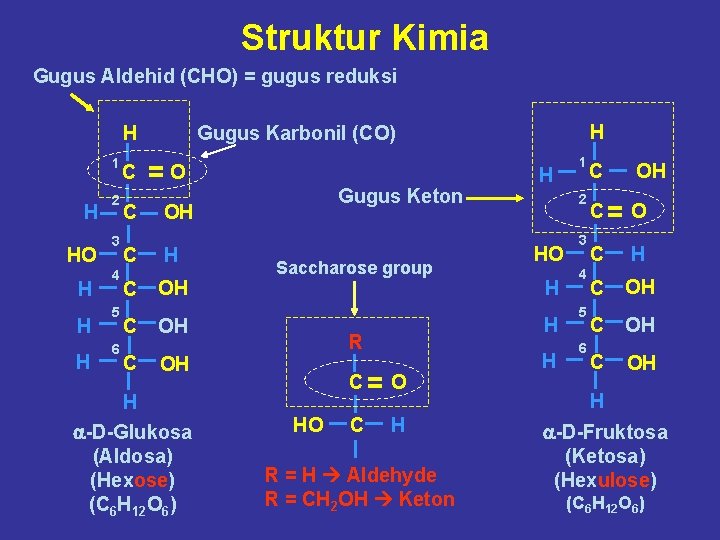
Struktur Kimia Gugus Aldehid (CHO) = gugus reduksi H 1 H HO H H H 2 3 4 5 6 C C C H Gugus Karbonil (CO) =O Gugus Keton OH H OH Saccharose group OH H a-D-Glukosa (Aldosa) (Hexose) (C 6 H 12 O 6) C= O HO C HO H H R OH H H R = H Aldehyde R = CH 2 OH Keton H 1 C 2 C= O 3 4 5 6 OH C OH H a-D-Fruktosa (Ketosa) (Hexulose) (C 6 H 12 O 6)
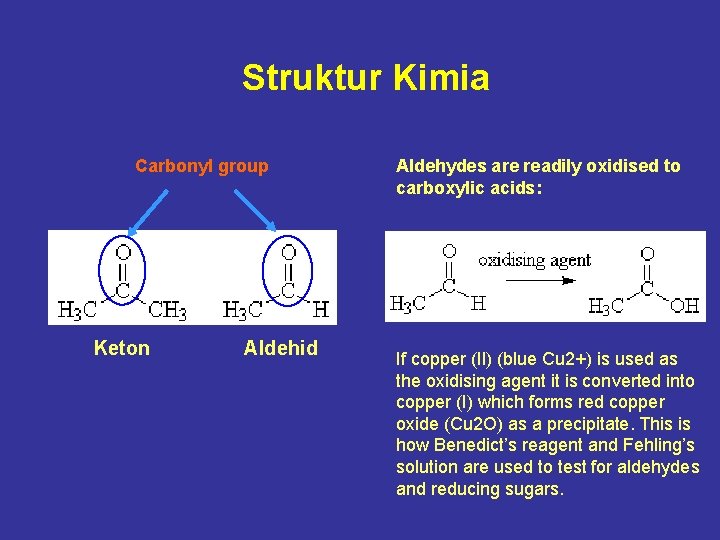
Struktur Kimia Carbonyl group Keton Aldehid Aldehydes are readily oxidised to carboxylic acids: If copper (II) (blue Cu 2+) is used as the oxidising agent it is converted into copper (I) which forms red copper oxide (Cu 2 O) as a precipitate. This is how Benedict’s reagent and Fehling’s solution are used to test for aldehydes and reducing sugars.
Struktur Kimia
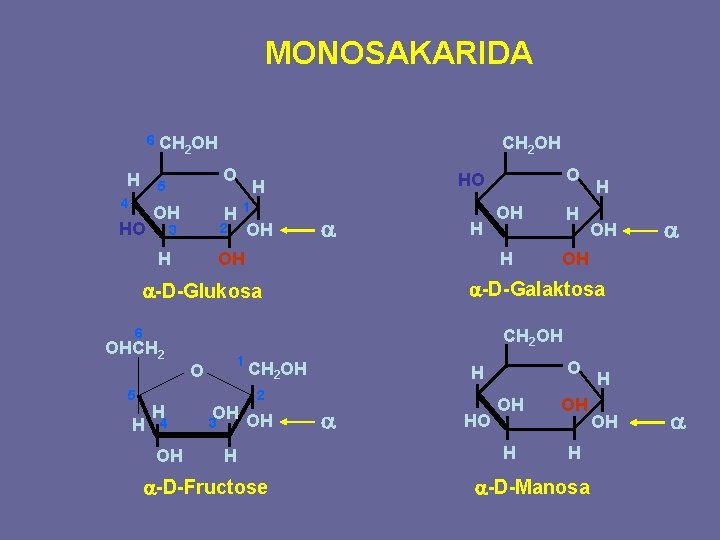
MONOSAKARIDA 6 CH H 2 OH O 5 4 HO CH 2 OH OH H 1 2 3 OH a H OH H 6 5 H H 4 OH OH a a-D-Galaktosa a-D-Glukosa OHCH 2 O HO H CH 2 OH O 1 CH 2 OH 2 OH OH 3 H a-D-Fructose O H a HO OH OH H H a-D-Manosa H OH a
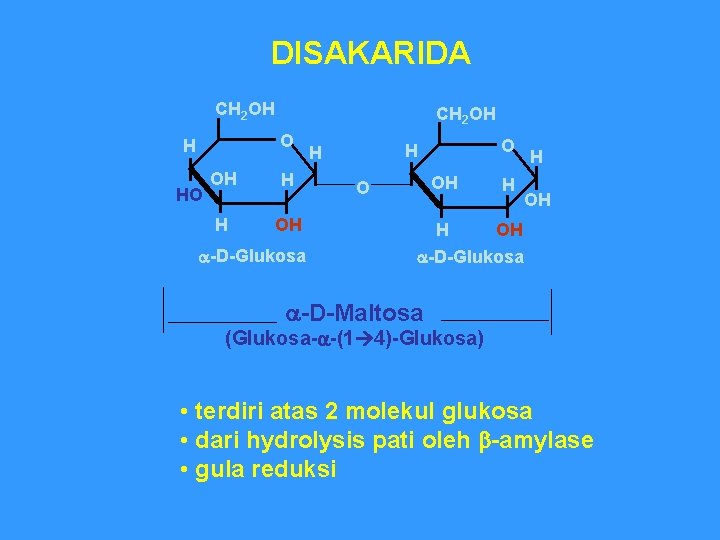
DISAKARIDA CH 2 OH O H HO OH H H OH a-D-Glukosa O H H OH OH H a-D-Glukosa a-D-Maltosa (Glukosa-a-(1 4)-Glukosa) • terdiri atas 2 molekul glukosa • dari hydrolysis pati oleh b-amylase • gula reduksi
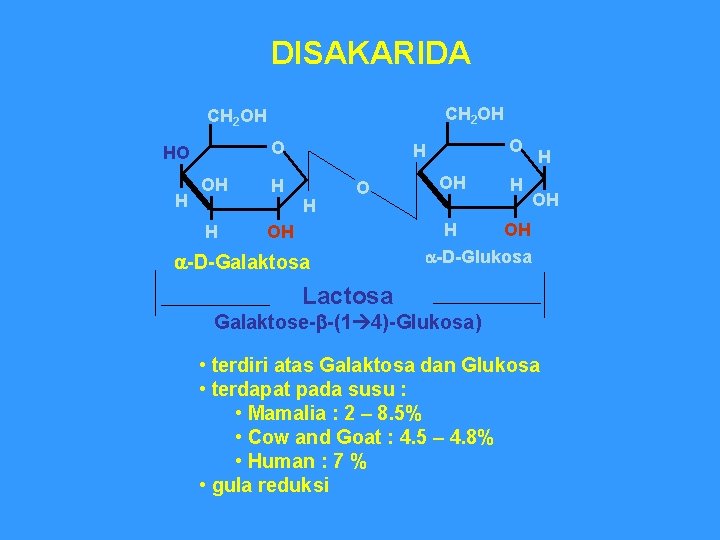
DISAKARIDA CH 2 OH O HO H OH H H O OH a-D-Galaktosa OH H H OH OH H a-D-Glukosa Lactosa Galaktose-b-(1 4)-Glukosa) • terdiri atas Galaktosa dan Glukosa • terdapat pada susu : • Mamalia : 2 – 8. 5% • Cow and Goat : 4. 5 – 4. 8% • Human : 7 % • gula reduksi
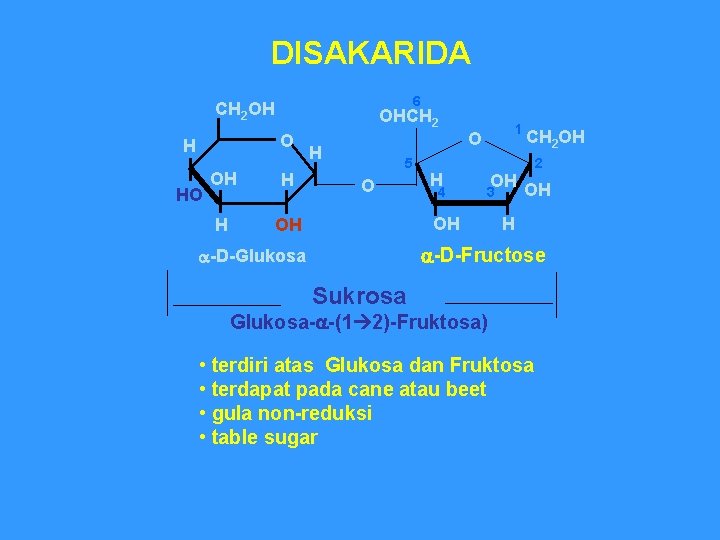
DISAKARIDA 6 CH 2 OH OHCH 2 O H HO OH H H OH H 5 O 1 O H 4 CH 2 OH 2 OH OH 3 OH H a-D-Fructose a-D-Glukosa Sukrosa Glukosa-a-(1 2)-Fruktosa) • terdiri atas Glukosa dan Fruktosa • terdapat pada cane atau beet • gula non-reduksi • table sugar
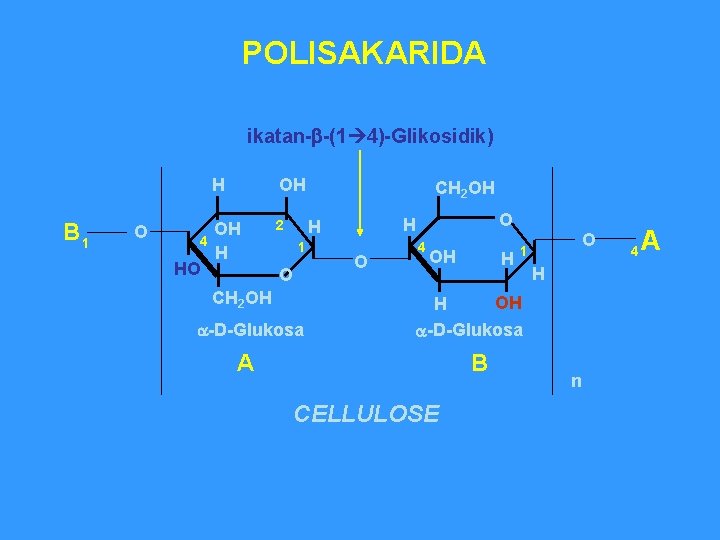
POLISAKARIDA ikatan-b-(1 4)-Glikosidik) B 1 O 4 HO H OH OH H 2 CH 2 OH 1 O CH 2 OH a-D-Glukosa O H H O 4 OH H O 1 H OH H a-D-Glukosa A B CELLULOSE n 4 A

POLISAKARIDA
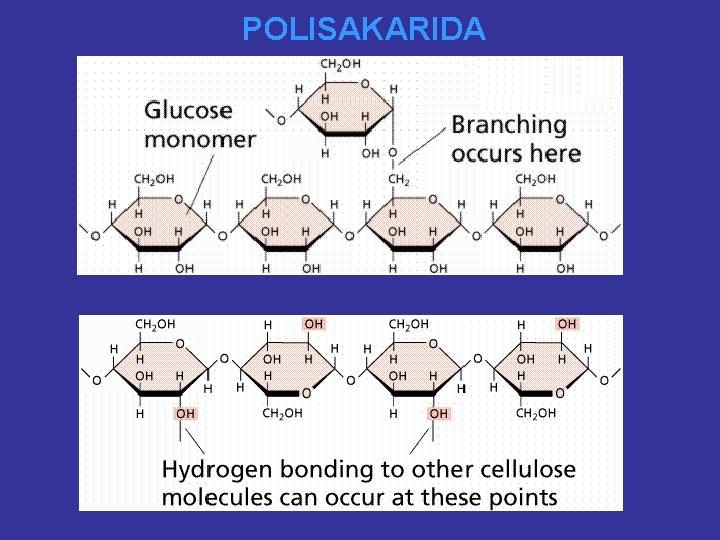
POLISAKARIDA

REAKSI-REAKSI KARBOHIDRAT 1. 2. HYDROLYSIS NON-ENZYMATIC BROWNING As shown in Table 4. 29, the C 1 and Cx factors, which were found to be endoand exo-1, 4 -β-glucanases respectively, hydrolyze cellulose to cellobiose. Since the C 1 factor is increasingly inhibited by its product, a cellobiase is needed so that cellulose breakdown is not rapidly brought to a standstill. However, cellobiase is also subject to product inhibition. Therefore, complete cellulose degradation is possible only if cellobiase is present in large excess or the glucose formed is quickly eliminated.
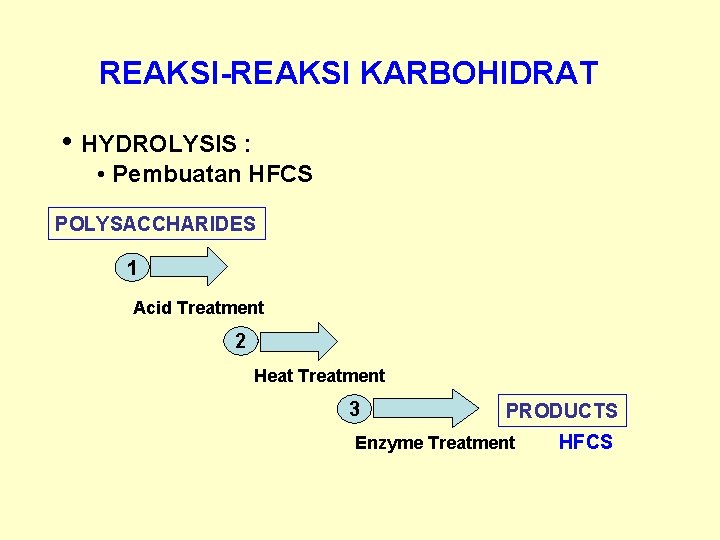
REAKSI-REAKSI KARBOHIDRAT • HYDROLYSIS : • Pembuatan HFCS POLYSACCHARIDES 1 Acid Treatment 2 Heat Treatment 3 PRODUCTS Enzyme Treatment HFCS
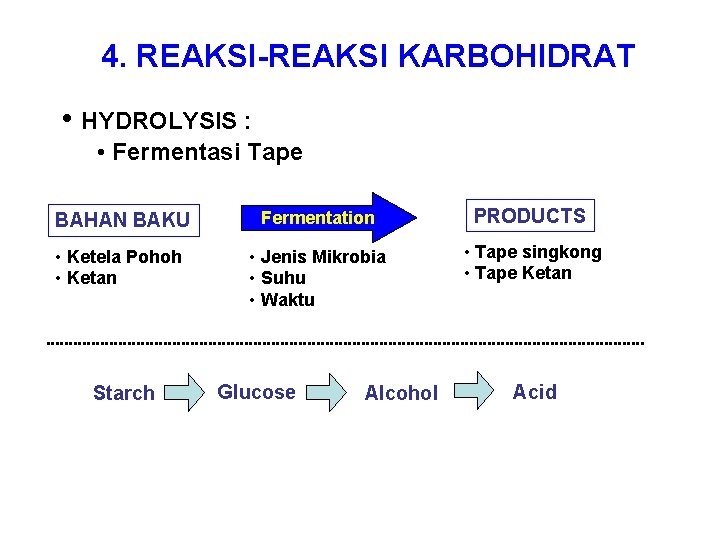
4. REAKSI-REAKSI KARBOHIDRAT • HYDROLYSIS : • Fermentasi Tape BAHAN BAKU Fermentation PRODUCTS • Ketela Pohoh • Ketan • Jenis Mikrobia • Suhu • Waktu • Tape singkong • Tape Ketan Starch Glucose Alcohol Acid
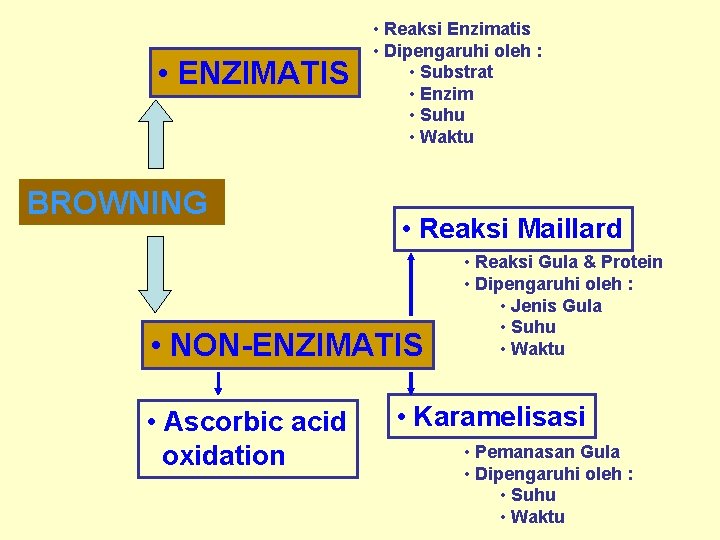
• ENZIMATIS BROWNING • Reaksi Enzimatis • Dipengaruhi oleh : • Substrat • Enzim • Suhu • Waktu • Reaksi Maillard • NON-ENZIMATIS • Ascorbic acid oxidation • Reaksi Gula & Protein • Dipengaruhi oleh : • Jenis Gula • Suhu • Waktu • Karamelisasi • Pemanasan Gula • Dipengaruhi oleh : • Suhu • Waktu

BROWNING Reaksi Maillard • Ilmuwan Perancis : Louis Maillard (1912) glucose + glycine • Carbonyl – Amine Reaction of amino acids/protein on reducing sugars • Related to aroma, taste and color • Roasting of coffee and cacao beans, baking of bread and cakes, toasting of cereals, cooking of meats •

BROWNING REDUCING SUGAR AMINO ACID CARBONYL AMINE • Dipengaruhi oleh : • Jenis Gula • Asam Amino • p. H • Suhu • Katalis • Kadar Air Melanoidins (Brown Pigments) 5 -Hydroxymethyl 2 -Furfuraldehyde (HMF)

REAKSI MAILLARD • • • Reaksi antara gula pereduksi dan Protein (asam amino) Dipengaruhi oleh suhu, waktu dan jenis gula Menghasilkan warna coklat Prosesnya berlangsung pada suasana basa Proses yang terjadi pada reaksi maillard: 1. Gugus karbonil pada gula menghasilkan Nglukosamin dan air 2. Gugus glukosamin yang tidak stabil mengalami pengaturan kembali membentuk ketosamin 3. Ketosamin mengalami proses lanjut: • Memproduksi air dan redukton • Menghasilkan diasetil, aspirin, pyruvaldehid, dan ikatan hidrolitik lain • Membentuk melanoidin.

MAILLARD REACTION (i) Initial stage (colourless) a. sugar-amine condensation b. Amadori rearrangement (ii) Intermediate stage (colourless to yellow) c. sugar dehydration d. sugar fragmentation e. amino acid degradation (iii)Final stage (highly coloured) f. aldol condensation g. aldehyde-amine polymerisation, formation of heterocyclic nitrogen compounds.
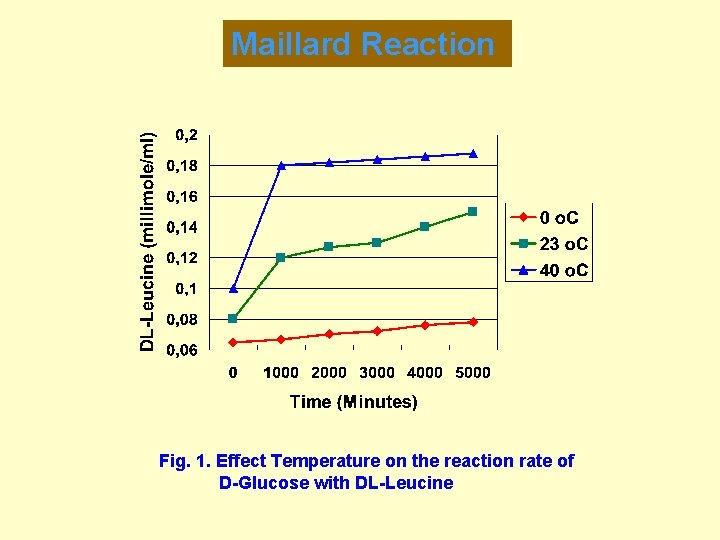
Maillard Reaction Fig. 1. Effect Temperature on the reaction rate of D-Glucose with DL-Leucine
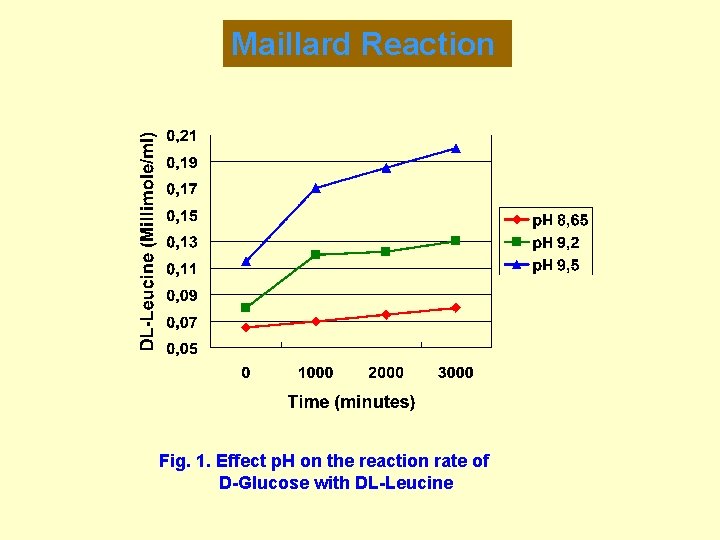
Maillard Reaction Fig. 1. Effect p. H on the reaction rate of D-Glucose with DL-Leucine

BROWNING REACTIONS Maillard Reaction : (Lysine + Sugars) SUCROSE + FRUCTOSE SUCROSE + LACTOSE

BROWNING NON ENZIMATIS : MAILLARD Produk : Bakpia

CARAMELIZATION • Caramelization is defined as thermal degradation of sugars leading to the formation of volatiles (caramel aroma) and brown-colored products (caramel colors). • The process is acid or base catalyzed and generally requires temperatures > 120 o. C at 9<p. H<3 (p. H < 3 or p. H > 9) • Caramelization occurs in food, when food surfaces are heated strongly, e. g. the baking and roasting processes, the processing of foods with high sugar content such as jams and certain fruit juices, or in wine production.

TEKNOLOGI KARBOHIDRAT SUMBER KARBOHIDRAT 1. Ketela Pohon 2. Beras Ketan 3. Jagung 4. Gandum TEKNOLOGI PROSES 1. Asam 2. Basa 3. Panas 4. Enzimatis 5. Mikrobiologis CARBOHYDRATE-BASED PRODUCT 1. Tape Ketela Pohon 2. Tape Ketan 3. HFCS 4. Karamel 5. Candy 6. Modified (CMC)

SIFAT FUNGSIONAL 1. SWEETNESS & SWEETENERS 2. HYGROSCOPICITY 1. Reduced Aw Preservative 2. Adsorbent Baby care products 3. Moisture Beauty care products 3. TEKSTURAL CONTRIBUTION 1. Rigidity : Roti 2. Viscosity : Saus
- Slides: 41