Knowledge Organiser CHEMISTRY Unit 4 Chemical Changes Keywords
- Slides: 2

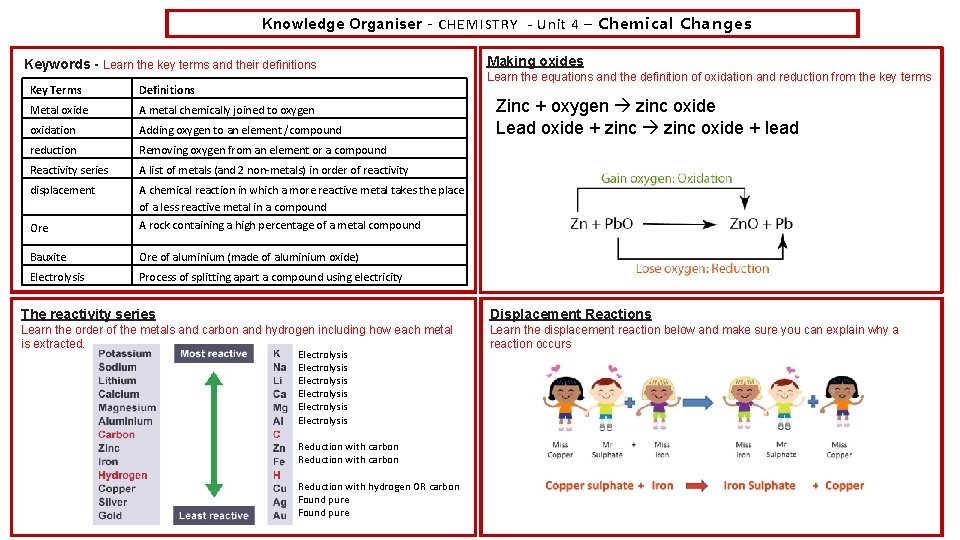
Knowledge Organiser – CHEMISTRY - Unit 4 – Chemical Changes Keywords - Learn the key terms and their definitions Key Terms Definitions Metal oxide A metal chemically joined to oxygen oxidation Adding oxygen to an element /compound reduction Removing oxygen from an element or a compound Reactivity series A list of metals (and 2 non-metals) in order of reactivity displacement Ore A chemical reaction in which a more reactive metal takes the place of a less reactive metal in a compound A rock containing a high percentage of a metal compound Bauxite Ore of aluminium (made of aluminium oxide) Electrolysis Process of splitting apart a compound using electricity Making oxides Learn the equations and the definition of oxidation and reduction from the key terms Zinc + oxygen zinc oxide Lead oxide + zinc oxide + lead The reactivity series Displacement Reactions Learn the order of the metals and carbon and hydrogen including how each metal is extracted. Learn the displacement reaction below and make sure you can explain why a reaction occurs Electrolysis Electrolysis Reduction with carbon Reduction with hydrogen OR carbon Found pure

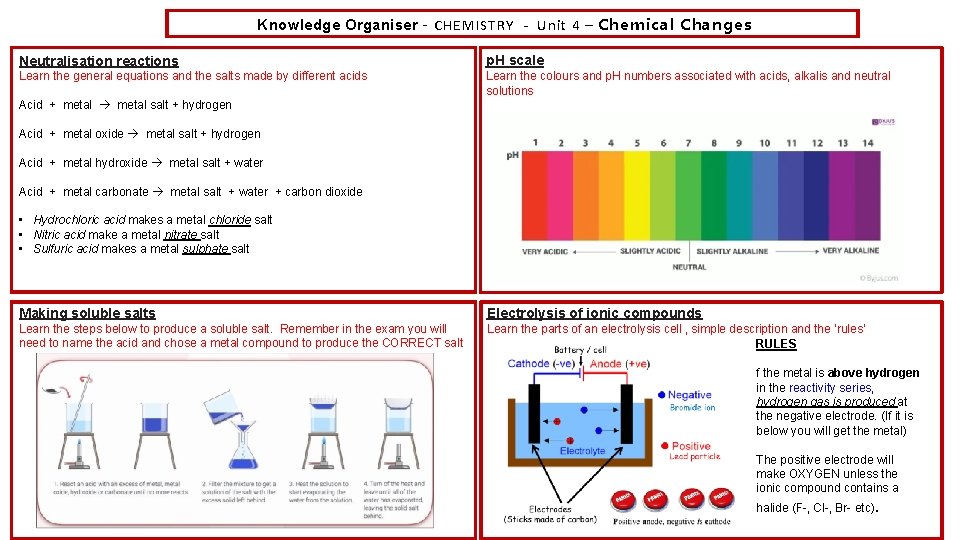
Knowledge Organiser – CHEMISTRY - Unit 4 – Chemical Changes Neutralisation reactions p. H scale Learn the general equations and the salts made by different acids Learn the colours and p. H numbers associated with acids, alkalis and neutral solutions Acid + metal salt + hydrogen Acid + metal oxide metal salt + hydrogen Acid + metal hydroxide metal salt + water Acid + metal carbonate metal salt + water + carbon dioxide • Hydrochloric acid makes a metal chloride salt • Nitric acid make a metal nitrate salt • Sulfuric acid makes a metal sulphate salt Making soluble salts Electrolysis of ionic compounds Learn the steps below to produce a soluble salt. Remember in the exam you will need to name the acid and chose a metal compound to produce the CORRECT salt Learn the parts of an electrolysis cell , simple description and the ‘rules’ RULES f the metal is above hydrogen in the reactivity series, hydrogen gas is produced at the negative electrode. (If it is below you will get the metal) The positive electrode will make OXYGEN unless the ionic compound contains a halide (F-, Cl-, Br- etc).