KLINICK SKSENOSTI LIEBY IMUNODEFICIENCI POMOCOU IVIG MUDR RAFF

KLINICKÉ SKÚSENOSTI LIEČBY IMUNODEFICIENCIÍ POMOCOU IVIG MUDR. Š. RAFFÁČ, MPH. , MUDR. J. GABZDILOVÁ, PHD. KIA UNLP, KHAOH LF UPJŠ A UNLP 5. FÓRUM IMUNODEFICIENCÍ, KOŠICE 28. 9. -29. 9. 2017

INTRAVENÓZNY IMUNOGLOBULÍN +L-PROLÍN § Polyvalenetný ľudský 10% Ig. G určený na iv. aplikáciu § L-prolín ako pomocná látka a p. H 4. 8 zabezpečujú dlhodobú stabilitu prípravku § Umožňuje jeho uskladnenie pri izbovej teplote (25ºC) § Môže byť podaný bezprostredne § L-proline vs glycine - prevencia agrgeácie a fragmentácie Ig. G § Mierne kyslé p. H roztoku bráni agregácii a tvorbe dimérov a predčasnej fragmentácii a strate špecifickej aktivity Ig. *In climate zones 3 and 4 where 30⁰C (86⁰F) stability data are required for room-temperature storage, the product will need to be stored at 2⁰C - 8⁰C (36⁰F-46⁰F). Please refer to the individual country label.

L-PROLÍN § L-proline je prirodzená aminokyselina a súčasť ľudského tela § Infúzia L –prolína nespôsobovala žiadne závažné neurologické vedľajšie účinky - Model na potkanoch (Irwin test) ani pri 2, 5 -10 - krát vyššej dávke ako je najvyššia dávka používaná v klinickej praxi 1. Hagan JB, Wasserman RL, Baggish JS, Spycher MO, Berger M, Shashi V, Lohrmann E, Sullivan KE. Safety of L-proline as a stabilizer for immunoglobulin products. Expert Rev Clin Immunol. 2012; 8(2): 169– 178. 2. Bolli R, et al. L-Proline reduces Ig. G dimer content and enhances the stability of intravenous immunoglobulin (IVIG) solutions. Biologicals 2010; 38(1): 150– 157.

NÍZKY OBSAH DIMÉROV IG G § V produktoch ivig v dôsledku interakcie protilátok od rôznych darcov dochádza prostredníctvom ich Fab oblasti k tvorbe dimérov 1, 2 § Vysoký obsah dimérov môže ovplyvniť klinickú toleranciu Ivig 3 § L-proline je amphifilná molekula , ktorá inhibuje tvorbu dimérov. Ig. G naviazaním sa miesta Ig G molekúl a tak ich satbilizuje v monomérnej forme. § Väzba je reverzibilná a neovplyvňuje vlastnosti lieku 4 1. Tankersley DL, et al. Immunoglobulin G dimer: an idiotype–anti-idiotype complex. Mol Immunol 1988; 25(1): 41– 48. 2. Roux KH, Tankersley DL. A view of the human idiotypic repertoire. Electron microscopic and immunologic analyses of spontaneous idiotype-anti-idiotype dimers in pooled human Ig. G. J Immunol 1990; 44(4): 1387– 1395. 3. Spycher MO, et al. Well-tolerated liquid intravenous immunoglobulin G preparations (IVIGs) have a low immunoglobulin G dimer (Ig. G-dimer) content. J Autoimmun 1999; 96(Suppl. 1): 96. 4. Bolli R, et al. L-Proline reduces Ig. G dimer content and enhances the stability of intravenous immunoglobulin (IVIG) solutions. Biologicals 2010; 38(1): 150– 157; figure reprinted with permission. †© 2009 The International Association for Biologicals. Published by Elsevier Ltd. All rights reserved.

ĎAŠIE VÝHODY - NÍZKY OBSAH SODÍKA A IG A § Nízky obsah Na umožňuje podávanie lieku aj u pacientov, kde je potrebná reštrikcia prijímu Na § Obsah Ig A je nízky ≤ 25 mg/L 1 1. Privigen SPC available at: http: //www. ema. europa. eu/docs/en_GB/document_library/EPAR_-_Product_Information/human/000831/WC 500043077. pdf. Accessed December 2014.

REGISTROVANÉ INDIKÁCIE IVIG Ø Primárne imunodeficiencie (PID) Ø Skundárne imunodeficiencie (SID) Ø Substitučná terapia u dospelých(≥ 18 r. ) pacientov s PID Ø Substitučná terapia pacientov s mnohopočetným myelómom a CLL spojených s ťažkou sekundárnou hypogamaglobulinémiou a opakovaným infekciami Ø Hypogamaglobulinémia u pacientov pred a po alogénnej transplantácii hematopoetických kmeňových buniek Ø Primárna imúnna trombocytopénia (ITP) Ø Chronická inflamatórna demyelinizačná inflamatórna polyneuropatia (CIDP) Ø Guillain-Barré syndróme (GBS) Ø Kawasaki choroba

PRIMÁRNE IMUNODEFICIENCIE (PID) v Poskytuje náhradu širokého spektra Ig G proti vírusom , baktériam a ich toxínom v Ochrana pred infekciou u pacientov s PID pri rough levels Ig. G≥ 4 g/L resp. Ig. G ≥ 6 g/L 1, 2 v Prevalenica PID : 3. 9– 5. 1: 10, 0003 Prevalence of common types of PID 4 -9 X-linked agammaglobulinaemia 1: 250, 000 Ig. G subclass deficiency 1: 10, 000 Selective Ig. A deficiency 1: 142– 18, 500 Common variable immunodeficiency 1: 50, 000 Paediatric severe combined immunodeficiency 1: 50, 000– 100, 000 Wiskott Aldrich syndrome 1: 250, 000 Úvodná liečba 400– 800 mg/kg nasledovanámini. 200 mg/kg bw každé 3– 4 týždne Udržiavacia liečba: 200– 800 mg/kg bw /mesiac 1. Buckley RH. Primary immunodeficiency diseases due to defects in lymphocytes. NEJM 2000; 343(18): 1313 -1324. 2. Mc. Cluskey DR, Boyd NA. Prevalence of primary hypogammaglobulinaemia in Northern Ireland. Proc R Coll Physicians Edinb 1989; 19: 191– 194. 3. Kobrynski L, Waltenburg Powell. R. Prevalence and morbidity of primary immunodeficiency diseases, United States 2001– 2007. J Clin Immunol 2014; 34: 954– 961. 4. Al-Herz W, Bousfiha A, Casanova J-L et al. Primary immunodeficiency diseases: an update on the classification from the International Union of Immunological Societies Expert Committee for Primary Immunodeficiency. Frontiers Immunol 2011; 2: 54. 5. Schwartz RA, Lin RY, Vafaie J. Immunoglobulin G deficiency, 2014. Available at http: //emedicine. medscape. com/article/136897 -overview. Accessed October 2014. 6. Dolina MY, Bascom R. Immunoglobulin A deficiency, 2014. Available at http: //emedicine. medscape. com/article/136580 -overview. Accessed October 2014. 7. Schwartz RA, Modak RM. Common variable immunodeficiency, 2014. Available at http: //emedicine. medscape. com/article/1051103 -overview. Accessed October 2014. 8. Schwartz RA, Sinha S. Pediatric severe combined immunodeficiency, 2014. Available at: http: //emedicine. medscape. com/article/888072 -overview. Accessed October 2014. 9. Dibbern DA, Routes JM. Wiskott-Aldrich syndrome, 2014. Available at: http: //emedicine. medscape. com/article/137015 -overview. Accessed October 2014.

SEKUNÁRNE IMUNODEFICIENCIE (SID) Ø B-cell lymphoproliferatívne ochorenia 1 o IVIG dávka: 200– 400 mg/kg bw / 3– 4 t. o Substitučná terapia pacientov s mnohopočetným myelómom a CLL spojených s ťažkou sekundárnou hypogamaglobulinémiou a opakovaným infekciami o Hypogamaglobulinémia u pacientov pred a po alogénnej transplantácii hematopoetických kmeňových buniek Ø CONGENITAL AIDS WITH RECURRENT INFECTIONS IN CHILDREN 1 – IVIG dávka: 200– 400 mg/kg bw / 3– 4 t. 1. Privigen Summary of Product Characteristics (EMA). April 2011. Available at: http: //www. ema. europa. eu/docs/en_GB/document_library/EPAR__Product_Information/human/000831/WC 500043077. pdf. Accessed December 2014.
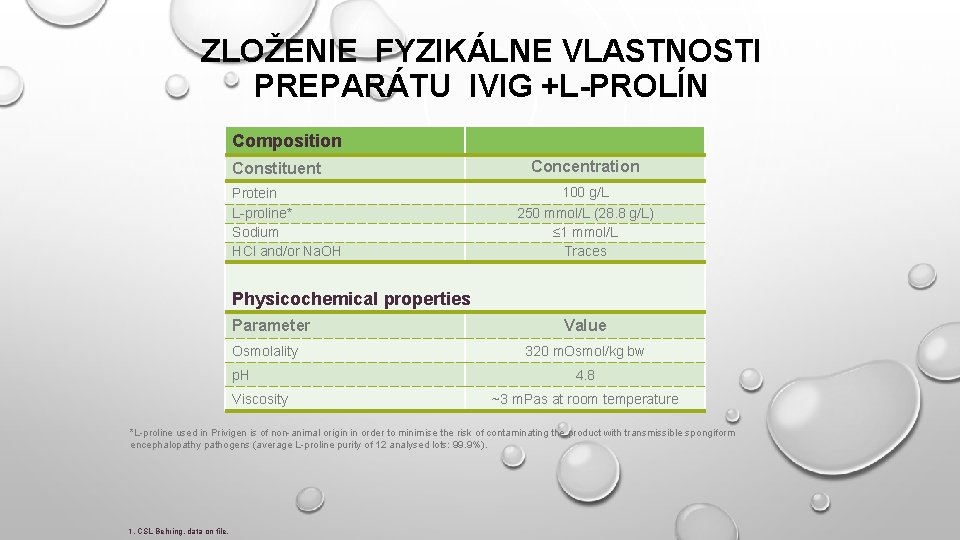
ZLOŽENIE FYZIKÁLNE VLASTNOSTI PREPARÁTU IVIG +L-PROLÍN Composition Constituent Protein L-proline* Sodium HCl and/or Na. OH Concentration 100 g/L 250 mmol/L (28. 8 g/L) ≤ 1 mmol/L Traces Physicochemical properties Parameter Osmolality p. H Viscosity Value 320 m. Osmol/kg bw 4. 8 ~3 m. Pas at room temperature *L-proline used in Privigen is of non-animal origin in order to minimise the risk of contaminating the product with transmissible spongiform encephalopathy pathogens (average L-proline purity of 12 analysed lots: 99. 9%). 1. CSL Behring, data on file.

MECHANIZMUS ÚČINKU Substitúcia Ig. G u PID a SID Blokáda a imunomodulácia Fc receptorov Vychytávanie komplementu o ovplyvnenie je aktivácie a vychytávania Anti-idiotypická neutralizícia patologických auto alebo allo- protilátok. Down-reguláciaauto-Ab tvorby B–ly Akcelerovaný katabolizmus patologických auto-Ab Regulácia apoptózy

KAZUISTIKA: Muž, 1958 RA: negat OA: od 18 r. sledovaný dermatológom a imunológom-alopécia, zistený deficit imunity, ekzemy, rosacea faciei, angiolog. - Raynaudov sy, reumatolog. - artralgie, od 6/2012 chron. gastritída APCA pozit. , susp. perniciózna anémia, deficit vitamínu B 12, trombocytopénia a leukopénia v. s. na autoimunitnom podklade. Demyelinizačné lézie CNS vs. na podklade vaskulopatie. Hepatosplenomegália s presiaknutím steny terminálneho ilea - USG verifikovaná. Asthma bronchiale, veruccae vulg. , laktátová intoleranica, art. hypertenzia 2. stp. ESH/ESC. 7/2012 extirpácia pleomorfného adenómu gl. parotis. → Primárny imunodeficit humor. imunity CVID s poklesom hladín Ig. G, a Ig. M na substitučnej liečbe s orgánovými známkami autoimunitného postihnutia AA: Saliciláty LA: B-blokátor, statín, ivig 10% + prolín→ sc. Ig +prolín, transfer faktor B lymfocytov, fexofenadín, pentoxyfiln, salbutamol inh. , salmeterol+flutikazón inh. , azelastín gutt oc. , vit B 12 + B 1.
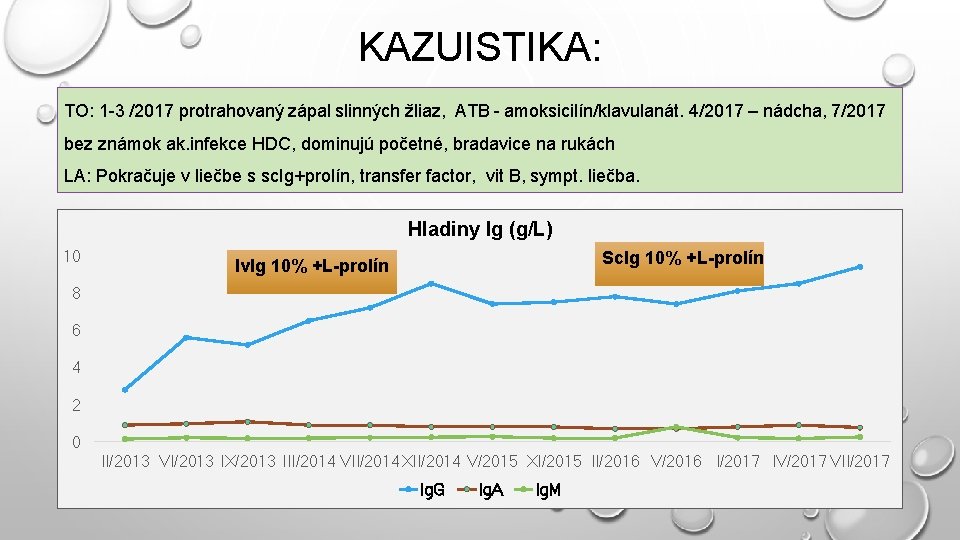
KAZUISTIKA: TO: 1 -3 /2017 protrahovaný zápal slinných žliaz, ATB - amoksicilín/klavulanát. 4/2017 – nádcha, 7/2017 bez známok ak. infekce HDC, dominujú početné, bradavice na rukách LA: Pokračuje v liečbe s sc. Ig+prolín, transfer factor, vit B, sympt. liečba. Hladiny Ig (g/L) 10 Sc. Ig 10% +L-prolín Iv. Ig 10% +L-prolín 8 6 4 2 0 II/2013 VI/2013 IX/2013 III/2014 VII/2014 XII/2014 V/2015 XI/2015 II/2016 V/2016 I/2017 IV/2017 VII/2017 Ig. G Ig. A Ig. M

ZÁVER • SUBSTITUČNÁ LIEČBA PID IVIG 10% +L- PROLÍNOM POSKYTUJE PRE PACIENTOV EFEKTÍVNU LIEČBU • UMOŽŇUJE BEZPROSTREDNÉ PODANIE LIEKU • JE SPOJENÁ S MINIMALIZÁCIOU REAKCIÍ PRI APLIKÁCII LIEKU A DOBRÝM KLINICKÝM EFEKTOM

CONCLUSIONS IVIG +L-PROLÍN PIVOTAL PID STUDY 1 OBJECTIVE: TO ASSESS THE SAFETY AND EFFICACY OF PRIVIGEN IN PATIENTS WITH PRIMARY IMMUNODEFICIENCY (PID) PATIENTS – 80 PATIENTS WITH COMMON VARIABLE IMMUNODEFICIENCY (CVID) OR X-LINKED AGAMMAGLOBULINAEMIA (XLA) ON STABLE IVIG THERAPY IN 19 STUDY SITES IN THE USA AND EUROPE INCLUSION CRITERIA – MALES AND FEMALES BETWEEN 3 AND 69 YEARS OF AGE WHO HAD EITHER CVID OR XLA – HAVE RECEIVED REGULAR IVIG THERAPY EVERY 3 TO 4 WEEKS FOR AT LEAST 6 MONTHS PRIOR TO SCREENING AND TO HAVE HAD AT LEAST ONE DOCUMENTED SERUM IGG TROUGH LEVEL OF ≥ 4 G/L DURING THIS TIME EXCLUSION CRITERIA – PREVIOUS SERIOUS ADVERSE REACTION TO IMMUNOGLOBULINS OR OTHER BLOOD PRODUCTS – SELECTIVE IGA DEFICIENCY OR KNOWN ANTIBODIES TO IGA – HYPERPROLINAEMIA – PREGNANCY, PLANNING A PREGNANCY OR BREASTFEEDING 1. Stein MR, et al. Ig. Pro 10 in PID Study Group. Safety and efficacy of Privigen, a novel 10% liquid immunoglobulin preparation for intravenous use, in patients with primary immunodeficiencies. J Clin Immunol 2009; 29: 137 -144.
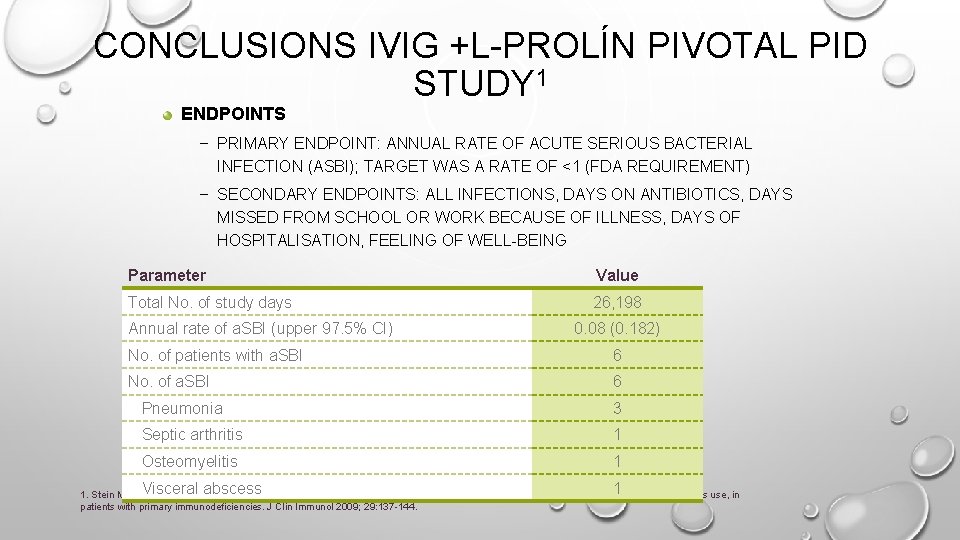
CONCLUSIONS IVIG +L-PROLÍN PIVOTAL PID STUDY 1 ENDPOINTS – PRIMARY ENDPOINT: ANNUAL RATE OF ACUTE SERIOUS BACTERIAL INFECTION (ASBI); TARGET WAS A RATE OF <1 (FDA REQUIREMENT) – SECONDARY ENDPOINTS: ALL INFECTIONS, DAYS ON ANTIBIOTICS, DAYS MISSED FROM SCHOOL OR WORK BECAUSE OF ILLNESS, DAYS OF HOSPITALISATION, FEELING OF WELL-BEING Parameter Value Total No. of study days 26, 198 Annual rate of a. SBI (upper 97. 5% CI) 0. 08 (0. 182) No. of patients with a. SBI 6 No. of a. SBI 6 Pneumonia 3 Septic arthritis 1 Osteomyelitis 1 Visceral abscess 1 1. Stein MR, et al. Ig. Pro 10 in PID Study Group. Safety and efficacy of Privigen, a novel 10% liquid immunoglobulin preparation for intravenous use, in patients with primary immunodeficiencies. J Clin Immunol 2009; 29: 137 -144.

CONCLUSIONS IVIG +L-PROLÍN PIVOTAL PID STUDY 1 • • • The first 10% liquid IVIG preparation stabilised with the natural amphiphilic amino acid l-proline The study Stein et al. , (2009) demonstrates the safety and efficacy of privigen in PID patients The study met the efficacy and safety endpoints • ASBI (ac. Seriouse bacterial infections) rate was 0. 08, upper 97% CI: 0. 182, below the target set by FDA (annual rate of ASBI <1) • The proportion of privigen infusions with temporally associated AEs was 0. 21, below the limit of 0. 4 set by the FDA • • 86% of the infusions were administered at 8 mg/kg bw/min Premedication to alleviate potentially infusion-related AEs was infrequently used (8 of 80 patients; 10%) The observed types, frequencies and severities of all aes potentially related to privigen were as expected and indicated good tolerability Privigen treatment resulted in serum igg trough concentrations that remained stable throughout the entire treatment period 1. Stein MR, et al. Ig. Pro 10 in PID Study Group. Safety and efficacy of Privigen, a novel 10% liquid immunoglobulin preparation for intravenous use, in patients with primary immunodeficiencies. J Clin Immunol 2009; 29: 137 -144.

ĎAKUJEM ZA POZORNOSŤ
- Slides: 17