Kirchhoffs laws of spectra Gustav Kirchhoff and Robert

Kirchhoff’s laws of spectra Gustav Kirchhoff and Robert Bunsen in the 1860 s

Chemical fingerprinting: Different hot gases give off different colors (or wavelengths). In other words: different elements have different lines in their spectra.

In 1913, Niels Bohr devised the modern theory of the atom. It explains spectral lines. Electron orbits have specific energies. Electrons jump between orbits when atoms gain or lose energy. Atoms therefore emit or absorb only specific amounts of energy, or colors (or wavelengths) of light.
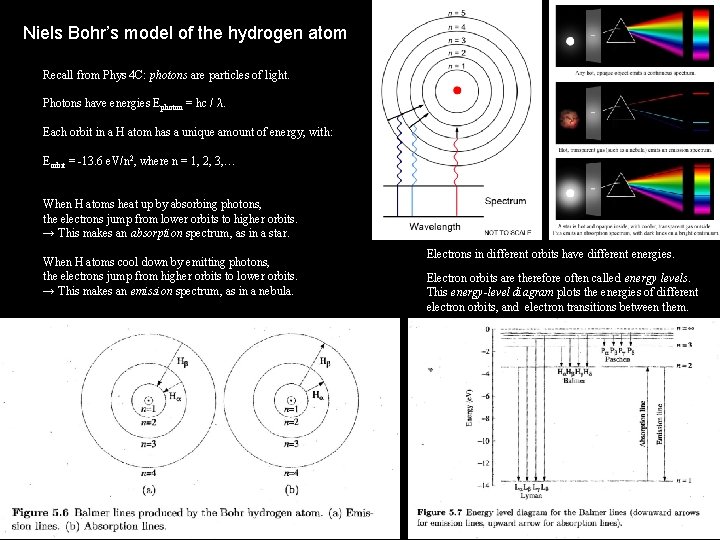
Niels Bohr’s model of the hydrogen atom Recall from Phys 4 C: photons are particles of light. Photons have energies Ephoton = hc / λ. Each orbit in a H atom has a unique amount of energy, with: Eorbit = -13. 6 e. V/n 2, where n = 1, 2, 3, … When H atoms heat up by absorbing photons, the electrons jump from lower orbits to higher orbits. → This makes an absorption spectrum, as in a star. When H atoms cool down by emitting photons, the electrons jump from higher orbits to lower orbits. → This makes an emission spectrum, as in a nebula. Electrons in different orbits have different energies. Electron orbits are therefore often called energy levels. This energy-level diagram plots the energies of different electron orbits, and electron transitions between them.



Extreme ultraviolet (EUV) photons have λ < 91. 2 nm and so always ionize hydrogen. Hot stars in both star death (left) and star birth (right) can light up the H gas surrounding them, similarly to how fluorescent lights work (although fluorescent lights use electrons to ionize the gas in them). Star death: a planetary nebula Star birth: a star-forming region


The Balmer lines (Hα, Hβ, Hγ, etc. ) are from electrons jumping into (for emission) and out of (for absorption) the n = 2 orbit. Hα, Hβ, and Hγ are visible light. The Lyman lines (Ly α, Ly β, Ly γ, etc. ) are from electrons jumping into/out of the n = 1 orbit. The Lyman series are all ultraviolet light (121. 6 nm to 91. 2 nm. ) NASA had a dedicated spacecraft, FUSE (the Far-Ultraviolet Spectroscopic Explorer) to observe the Lyman lines (in 1999 -2007). NASA’s Extreme Ultraviolet Explorer (EUVE) spacecraft observed at wavelengths with λ < 91. 2 nm. This corresponds to photon energies of >13. 6 e. V: energetic enough always to ionize hydrogen (in 1992 -2000).
- Slides: 9