KINGDOM OF SAUDI ARABIA Ministry of Higher Education

KINGDOM OF SAUDI ARABIA Ministry of Higher Education KING ABDULAZIZ UNIVERSITY ﻛﻠﻴﺔ ﺍﻟﻬﻨﺪﺳﺔ ﺑﺮﺍﺑﻎ Faculty of Engineering Rabigh Branch Introduction to Chemical Engineering “CHEN 201” Lecture 1 “Introduction” Mohamed Ismail Bassyouni, Ph. D. “Head of the Chemical Engineering Department” 1 Sep. 2015

Introduction to Materials Science & Engineering Course Objective. . . Introduce fundamental concepts in Chemical engineering calculations You will learn about: • Chemical Processes • Mass Balance • Energy Balance This course will help you to: • Carry out chemical engineering calculations • realize Chemical engineering processes 2

LECTURES Instructor: Dr. Mohamed Ismail Bassyouni Tutor: Eng. Ahmed Almalki E-mail : migb 2000@hotmail. com Emal: ahmed. lk 177@gmail. com Time: Monday & Wednesday: 14 -1650 am Location: R 3 Activities: • Present new material • Announce reading and homework • Take quizzes and midterms* 3

TEACHING Name Office Tel. Dr Mohamed Bassyouni 306 X-XXXX E-mail migb 2000@Hotmail. com Teaching Assistant will -Participate in recitation sessions -Chemical Engineering (Calculations) , 4

COURSE MATERIALS (with text) Textbook: Hammelblau D. M. and Riggs J. B. , Basic Principles and Calculations in Chemical Engineering, 7 th Edition, John Wiley, 2004 Reference: Felder M. , Rousaou W, Elementary Principles of Chemical Processes 5 th Edition, John Wiley, 1986 5

GRADING 1 - Weekly in-lecture quizzes 20% Based on core homework problems Your lowest quiz grade will be dropped 2 - Midterm 20% 3 - Assignment and homework 20% 5 - Final 40% 6
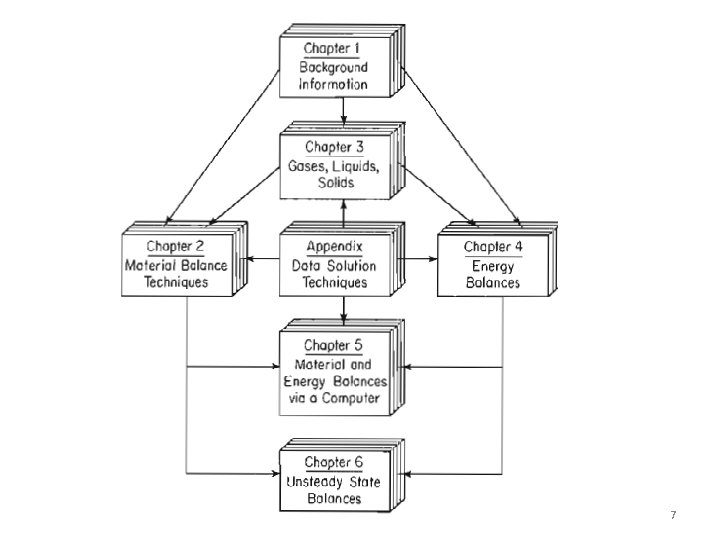
7

A chemist in your company's research and development division has discovered that if he mixes two reactants in a certain proportion at an elevated temperature. He obtains a product significantly more valuable than both reactants. The company contemplates manufacturing the product using a process based on this reaction. At this point the matter becomes an engineering problem or. more precisely. hundreds of engineering problems. A+B C 1. What type of reactor should be used? A long pipe? A large tank? Several smaller tanks? An extremely large test tube? How large? Made of what? Does it have to be heated? If so. how much and how? With an electrical heater inside or outside the reactor? By passing a hot fluid through a heating coil in the reactor'? By heating the reactants before they get into the reactor? Does the reaction supply its own heat, so that heating is needed only for startup? If so, can the reactor "run away" and possibly explode? Should control measures be introduced to prevent this? What kind? 2. Where should the reactants be obtained? Buy them, or make them? In what proportions should they be fed to the reactor? 8

3. Should the reactor effluent, which contains the product and unconsumed reactants. be sold as is, or should the product be separated from the reactants and the latter be sent back to the reactor? If separation is desirable. how can it be accomplished? Heat the mixture and draw off and condense the vapor, which will be richer in the more volatile substances than the original mixture? Add another substance that extracts the product and is immiscible with the reactants, and then separate the two phases mechanically? If all of the process materials are gases at the reaction temperature, can the mixture be cooled to a temperature at which the product condenses but the reactants do not, or vice versa, or if they are liquids can the mixture be cooled to a temperature at which the product crystallizes? If one of these alternatives is chosen, what kind of equipment is needed'? What size? What materials? What are the heating or cooling requirements? Are controls needed to keep the operation of the process within rigid limits? What kind of controls? Should they be manual or automatic? 4. How should the reactant and product streams be moved to and from the reactor and any heating, cooling, and separation equipment involved in the process? By gravity from a raised feed tank? With pumps, or blowers, or compressors. or conveyor belts? What kinds'? How big? In pipes made of what? 9

5. Is enough known about the reaction system to be able to answer all of these questions. Or should additional laboratory studies be carried out'? What studies? Can the laboratory data be used directly to design the industrial plant, or should a smaller pilot plant be constructed first to test the design? How much smaller? 6. What can possibly go wrong with the process, and what can be done if and when it does? 7. Are waste products produced by the process? In what quantities? Are they potentially harmful if released untreated into the environment? If so, in what way? What should be done to reduce pollution hazards? Chemically treat the wastes? Dump liquid and solid wastes into containers, seal and cart them out to sea? Disperse gases in the tmosphere with a high stack? Precipitate solids electrostatically from gas exhausts? 8. How much of the process should be automated. and how should the automation be done'? 9. How much will all of this cost? For how much can the product be sold, and to whom'? How much money will the process net each year? Is it enough to make it worthwhile? If so, where should the plant be built? 10

10. Once the plant has been built, what procedure should be followed for startup? 11. Six months later when startup has been achieved, why is the product not coming out the way it did in the laboratory? Is it an equipment malfunction or a change in conditions somewhere between the laboratory and industrial process? How can we find out? What can be done to correct the problem? Is it necessary to shut down the operation for modifications? 12. Is it significant or just a coincidental series of bad breaks that there have been three explosions and four fires within six months in the reactor unit? In either case, how do we stop them from recurring? 13. All sorts of other things are going wrong with the process operation. Why weren't they on the list of things that could possibly go wrong? What can be done about them? 14. When the process finally starts working perfectly and the next day an order comes down to change the product specifications, how can it be done without redesigning the entire process? Why didn't they think of this before they built the plant? 11
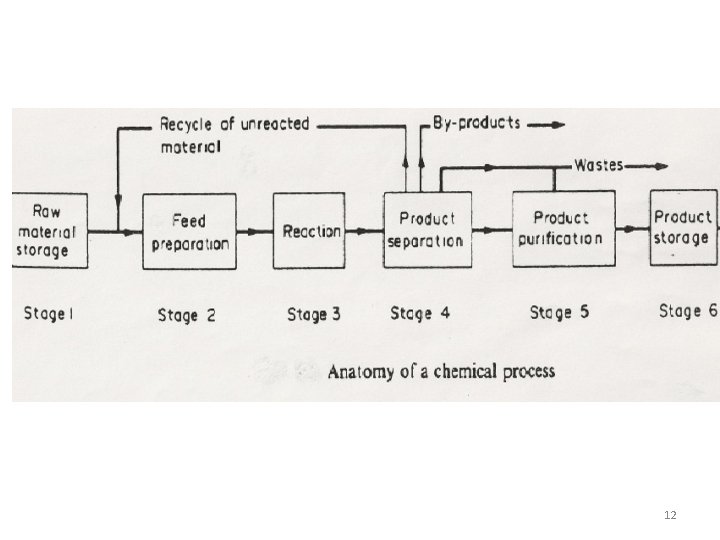
12

Questions are Welcome 13

KINGDOM OF SAUDI ARABIA Ministry of Higher Education KING ABDULAZIZ UNIVERSITY ﻛﻠﻴﺔ ﺍﻟﻬﻨﺪﺳﺔ ﺑﺮﺍﺑﻎ Faculty of Engineering Rabigh Branch Introduction to Chemical Engineering “Ch. E 201” Lecture 2 “Unites and Dimensions” Assis. Prof. Mohamed Ismail Bassyouni “Head of the Chemical Engineering Department” 14 Sep 2011

Dimensions & Units Dimensions: are our basic concepts of measurement such as -Length -Time -Mass -Temp…. etc. Units: are the means of expressing the diemntions such as: -Feet -Centimeters -hours Seconds, …etc. 15

Dimensions & Units • All measured property has a value and a unit • A dimension is a property that can be measured, or calculated by multiplying or dividing other dimensions. • Measurable units are specific values of dimensions that have been defined by convention, custom, or law. 16

• In engineering one will come across many dimensionless groups. • It is important to note that the numerical value for a dimensionless group is independent of the units chosen for the primary quantities, provided the units are consistent. • Dimensional Homogeneity ---- every valid equation must be "dimensionally homogeneous" --- all additive terms must have the same dimension. 17
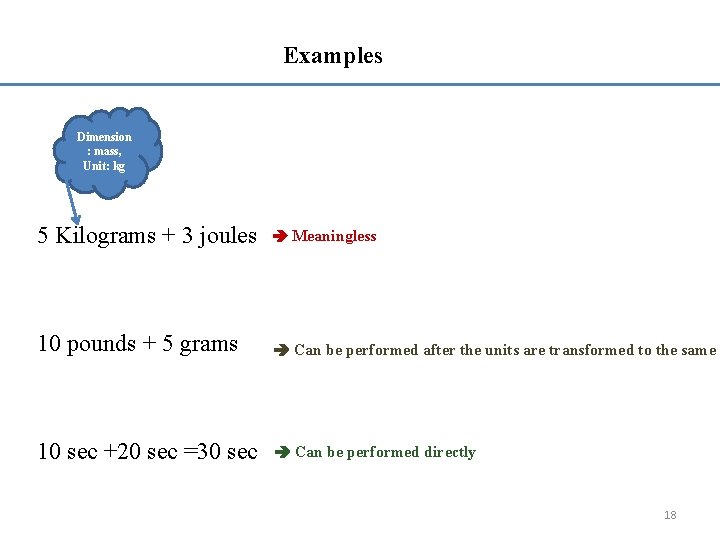
Examples Dimension : mass, Unit: kg 5 Kilograms + 3 joules Meaningless 10 pounds + 5 grams Can be performed after the units are transformed to the same 10 sec +20 sec =30 sec Can be performed directly 18

Systems of units • Base Units • Multiple Units • Derived Units • International System of Units: (SI) system • Centimetre-gram-second system: CGS System • American Engineering System 19

Base units for mass, length, time, temperature, electrical current, and light intensity. 20
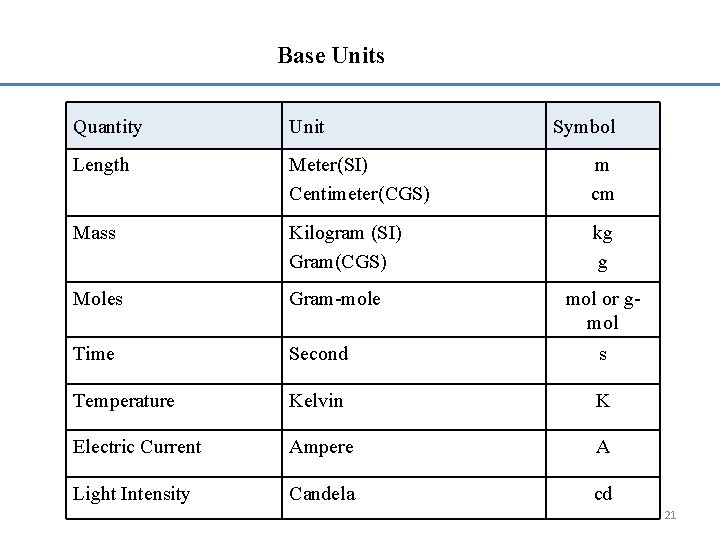
Base Units Quantity Unit Symbol Length Meter(SI) Centimeter(CGS) m cm Mass Kilogram (SI) Gram(CGS) kg g Moles Gram-mole Time Second s Temperature Kelvin K Electric Current Ampere A Light Intensity Candela cd mol or gmol 21

Multiple Unites Which are defined as multiples or fractions of base units such as minutes, hours, and milliseconds, all of which are defined in terms of the base unit of a second. Multiple units are defined for convenience rather than necessity: it is simply more convenient to refer to 3 yr than to 94, 608, 000 s. 22

Multiple Units • • Tera (T) = 1012 Giga (G) = 109 Mega (M) = 10 6 Kilo (k) = 103 Centi (c) = 10 -2 Milli (m) = 10 -3 Micro (μ) = 10 -6 Nano (n) =10 -9 23

Derived units Otained in one of two ways: (a) By multiplying and dividing base or multiple units (cm 2, ftlmin, kg' m/s 2 , etc. ). Derived units of this type are referred to as compound units. (b) As defined equivalents of compound units (e. g. , 1 erg (lg'cm/s 1 lbf " 32. 174 Ibm ·ft/s 2). 24
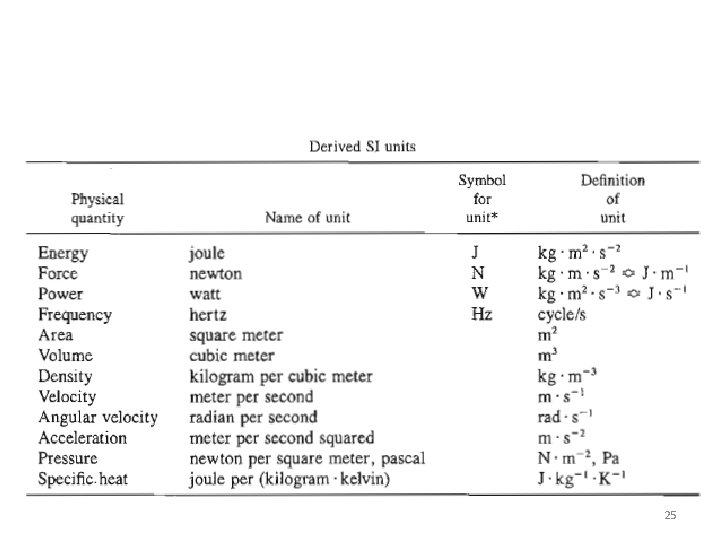
25
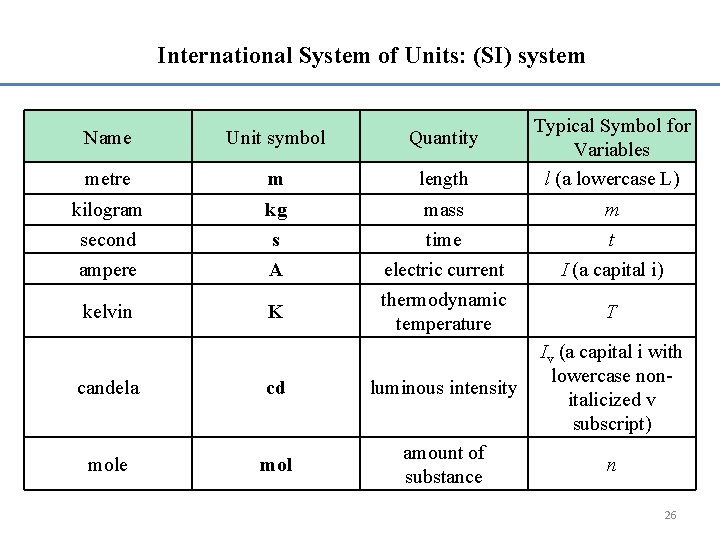
International System of Units: (SI) system Name Unit symbol Quantity metre m length kilogram second ampere kg s A kelvin K mass time electric current thermodynamic temperature Typical Symbol for Variables l (a lowercase L) m t I (a capital i) T candela cd luminous intensity Iv (a capital i with lowercase nonitalicized v subscript) mole mol amount of substance n 26
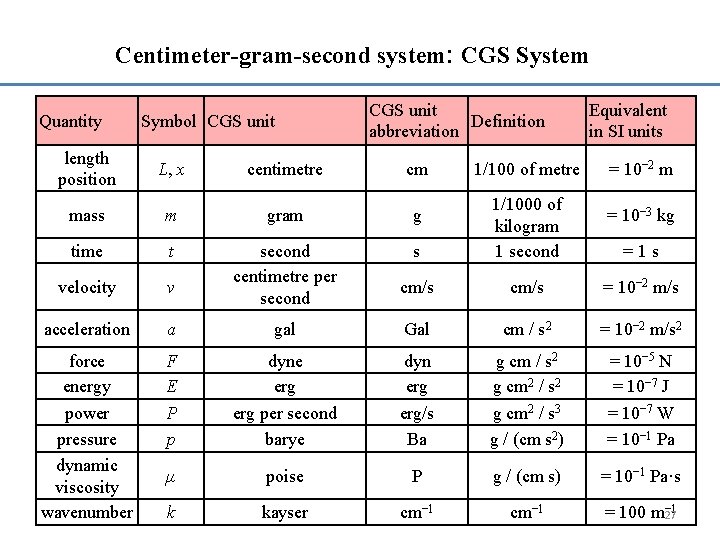
Centimeter-gram-second system: CGS System Quantity Symbol CGS unit Definition abbreviation Equivalent in SI units length position L, x centimetre cm mass m gram g time t s velocity v second centimetre per second 1/1000 of kilogram 1 second cm/s = 10− 2 m/s acceleration a gal Gal cm / s 2 = 10− 2 m/s 2 force energy power pressure dynamic viscosity wavenumber F E P p dyne erg per second barye dyn erg/s Ba g cm / s 2 g cm 2 / s 3 g / (cm s 2) = 10− 5 N = 10− 7 J = 10− 7 W = 10− 1 Pa μ poise P g / (cm s) = 10− 1 Pa·s k kayser cm− 1 − 1 = 100 m 27 1/100 of metre = 10− 2 m = 10− 3 kg = 1 s

28

Convert an acceleration of 1 cm/s 2 to its equivalent in km/yr 2. 29

Questions are Welcome 30

KINGDOM OF SAUDI ARABIA Ministry of Higher Education KING ABDULAZIZ UNIVERSITY ﻛﻠﻴﺔ ﺍﻟﻬﻨﺪﺳﺔ ﺑﺮﺍﺑﻎ Faculty of Engineering Rabigh Branch Introduction to Chemical Engineering “Ch. E 201” Lecture 3 “Important terms” Assis. Prof. Mohamed Ismail Bassyouni “Head of the Chemical Engineering Department” 31 Sep. 2011

Dimensional consistency A basic principle exists that equations must be dimensionally consistent Example : Van der waals equation a will have the unit: atm (cm)6 b will have the unit: cm 3 32

Non dimensional groups (cm) (s) (g) (cm) (s) (cm) 3 (g) All units cancel out 33

Important terms a) Precision: refers to the degree of dispersion (or deviations) of measurements from their true values b)Accuracy: refers to how close measures value is to its true value c) Mole and mole fraction d) Density and specific gravity e)Flow rate f) Temperature g) Pressure 34
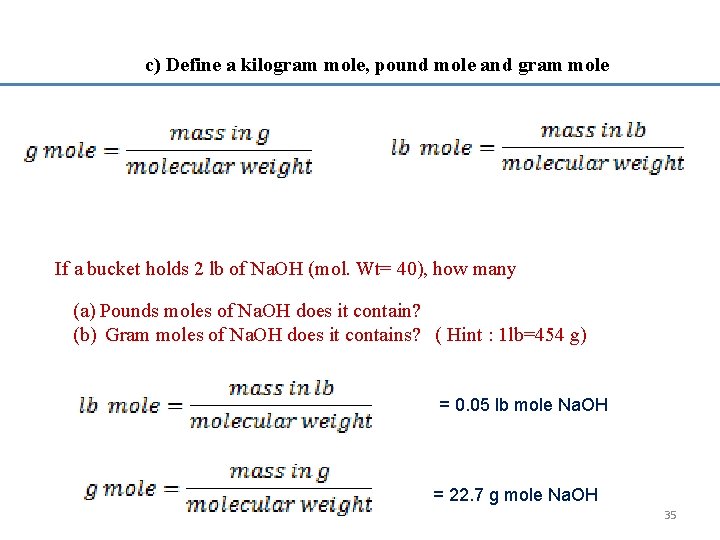
c) Define a kilogram mole, pound mole and gram mole If a bucket holds 2 lb of Na. OH (mol. Wt= 40), how many (a) Pounds moles of Na. OH does it contain? (b) Gram moles of Na. OH does it contains? ( Hint : 1 lb=454 g) = 0. 05 lb mole Na. OH = 22. 7 g mole Na. OH 35
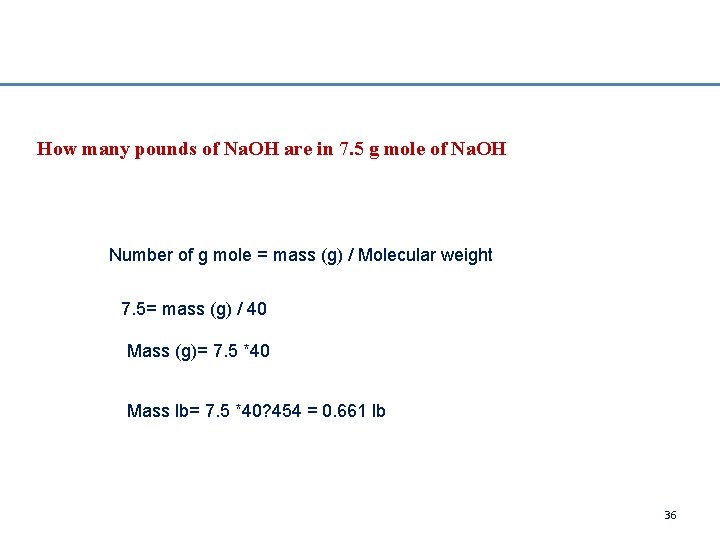
How many pounds of Na. OH are in 7. 5 g mole of Na. OH Number of g mole = mass (g) / Molecular weight 7. 5= mass (g) / 40 Mass (g)= 7. 5 *40 Mass lb= 7. 5 *40? 454 = 0. 661 lb 36

Mass and Mole Fraction 37

d) Density -The density of a substance is the mass per unit volume of the substance (kg/m 3 , g/cm 3, Ibm/ft 3, etc. ). -Densities of pure solids and liquids are essentially independent of pressure and vary relatively slightly with temperature specific gravity The specific gravity of a substance is the ratio of the density p of the substance to the density Pref of a reference substance at a specific condition: SG = ρ/ ρref Specific volume The specific volume of a substance is the volume occupied by a unit mass of the substance; it is the inverse of density 38
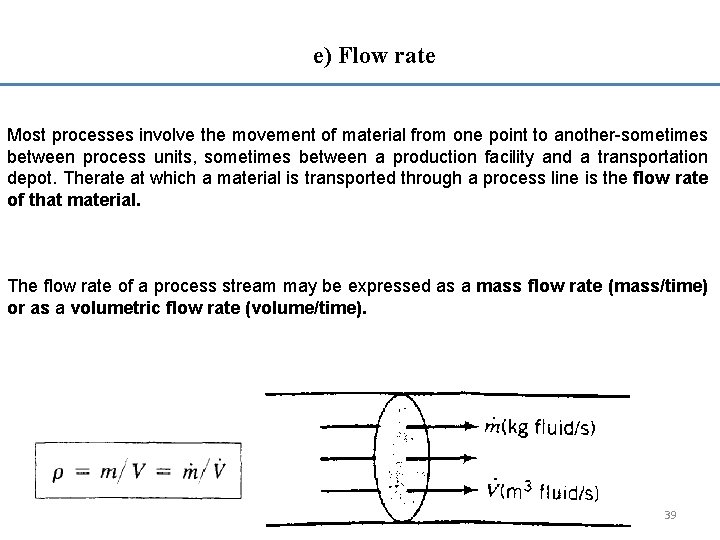
e) Flow rate Most processes involve the movement of material from one point to another-sometimes between process units, sometimes between a production facility and a transportation depot. Therate at which a material is transported through a process line is the flow rate of that material. The flow rate of a process stream may be expressed as a mass flow rate (mass/time) or as a volumetric flow rate (volume/time). 39

f) Temperature and pressure do not have large influences on the densities of solids and liquids. Nevertheless, the fact that mercury in a thermometer rises or falls with changing temperature shows that the effect of temperature on liquid density is measurable. the dependence of the volume of mercury on temperature as 40

The process variables The quantities used to describe a process are called process variables. To design or analyze a process, we need to know the amounts, compositions, and condition of materials entering, leaving, and within the process The process variables of interest to chemical engineers are: -Mass and Volume -Composition -Pressure -Temperature 41

Questions are Welcome 42

1 - Chemical composition • Most materials are mixtures of various species. • The physical properties of a mixture depend strongly on the mixture composition. 1. 1 Moles and Molecular Weight The atomic weight : of an element is the mass of an atom on a scale that assigns 12 C (the isotope of carbon whose nucleus contains six protons and six neutrons) a mass of exactly 12. The molecular weight of a compound is the sum of the atomic weights of the atoms that constitute a molecule of the compound: 43

Atomic weight of O = 16 Molecular weight of O 2 = 32 Example: Number of moles of 34 kg of ammonia (NH 3 : M = 17) 44

How many of each of the following are contained in 100. 0 g of CO 2 (M = 44. 01, atomic weight of Carbon = 12, Oxygen = 16)? (1) mol CO 2 ; (2) lb-moles CO 2 ; ( Hint : 1 lb=454 g) (3) mol C; (4) mol O; (5) mol O 2; (6) g O; (7) g O 2; (8) molecules of CO 2. 45

46

1. 2 Mass and Mole Fractions and Average Molecular Weight • Process streams occasionally contain one substance, but more often they consist of mixtures of liquids or gases, or solutions of one or more solutes in a liquid solvent. • Composition of a mixture of substances, including a species A. • The percent by mass of A is 100 x. A, • and the mole percent of A is 100 y. A 47

A solution contains 15% A by mass (XA = 0. 15) and 20 mole% B (YB = 0. 20). 1. Calculate the mass of A in 175 kg of the solution. 2. Calculate the mass flow rate of A in a stream of solution flowing at a rate of 53 Ibm/h. 3. Calculate the molar flow rate of B in a stream flowing at a rate of 1000 mol/min. 4. Calculate the total solution flow rate that corresponds to a molar flow rate of 28 kmol B/s. 5. Calculate the mass of the solution that contains 300 Ibm of A. 48

49
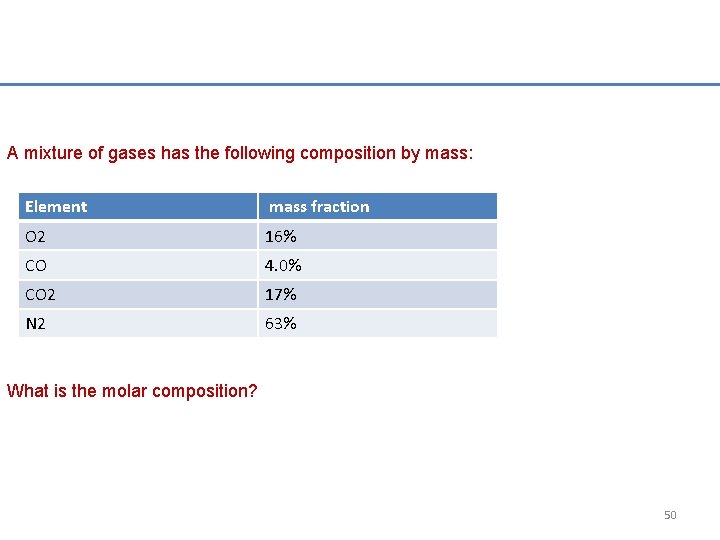
A mixture of gases has the following composition by mass: Element mass fraction O 2 16% CO 4. 0% CO 2 17% N 2 63% What is the molar composition? 50

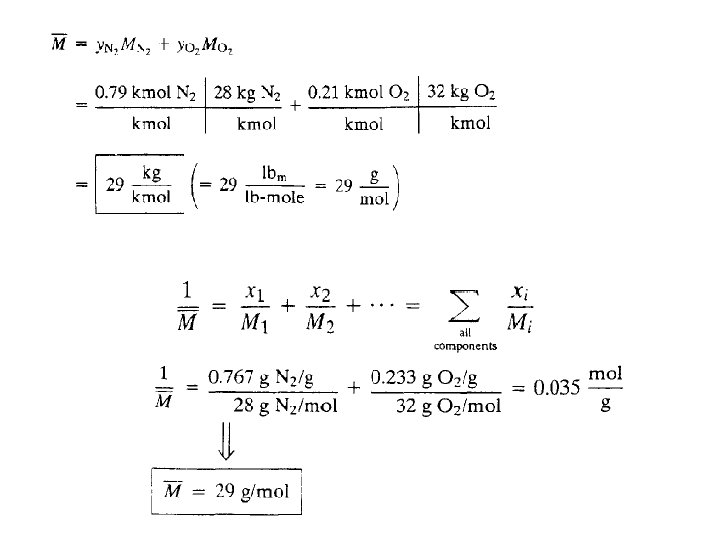
The average molecular weight (or mean molecular weight) of a mixture : is the ratio of the mass of a sample of the mixture (mt) to the number of moles of all species (nt) in the sample. 51

Calculate the average molecular weight of air: (1) from its approximate molar composition of 79% N 2, 21% O 2 (2) from its approximate composition by mass of 76. 7% N 2, 23. 3% O 2· 52
53

1. 3 Concentration 1 -The Molarity of a solution is the value of the molar concentration of the solute expressed in gram-moles solute/liter solution (e. g. , a 2 -molar solution of A contains 2 mol A/liter solution). 2 -Parts per Million and Parts per Billion 54
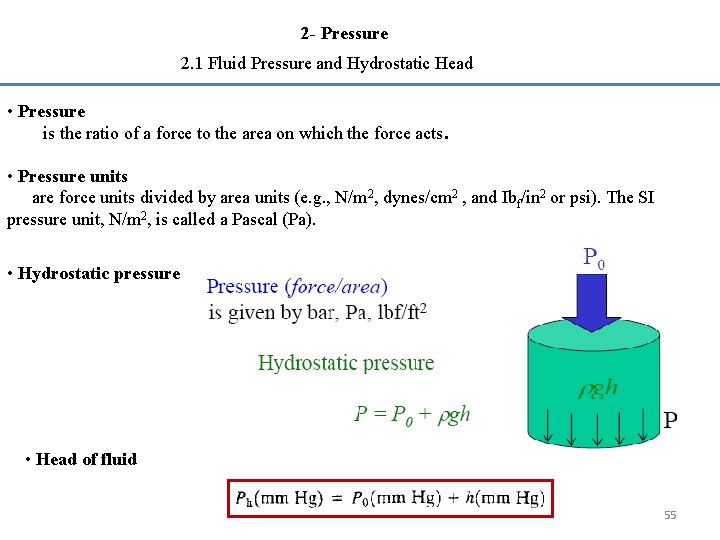
2 - Pressure 2. 1 Fluid Pressure and Hydrostatic Head • Pressure is the ratio of a force to the area on which the force acts. • Pressure units are force units divided by area units (e. g. , N/m 2, dynes/cm 2 , and Ibf/in 2 or psi). The SI pressure unit, N/m 2, is called a Pascal (Pa). • Hydrostatic pressure • Head of fluid 55
The fluid pressures referred to so far are all absolute pressures, Many pressure-measuring devices give the gauge pressure Psia : Pound /square inch absolute Psig: Pound /square inch gauge 56
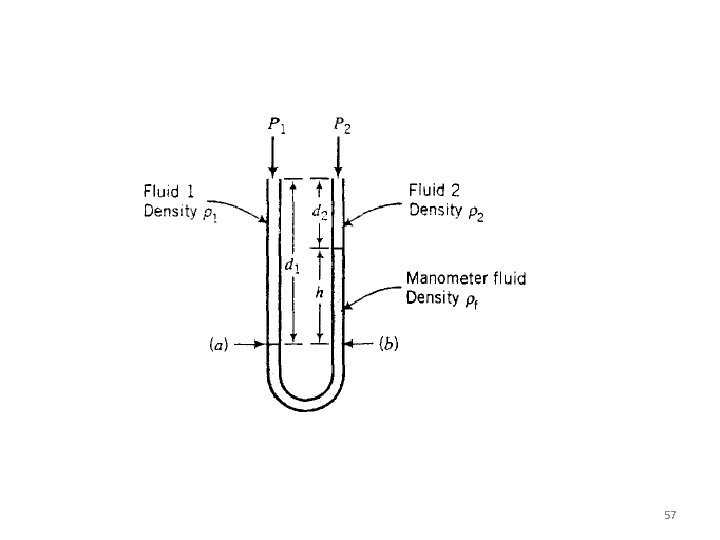
57
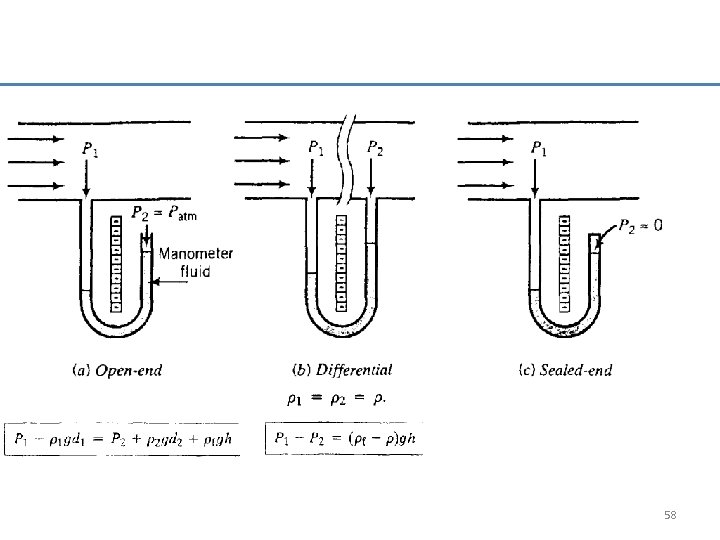
58
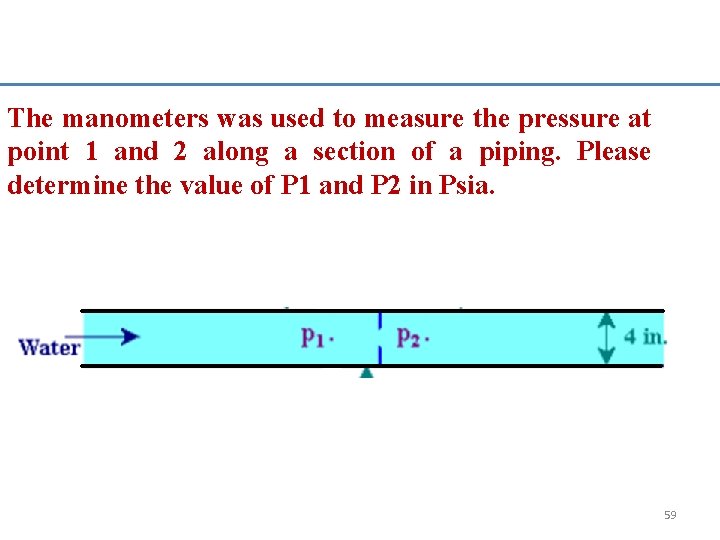
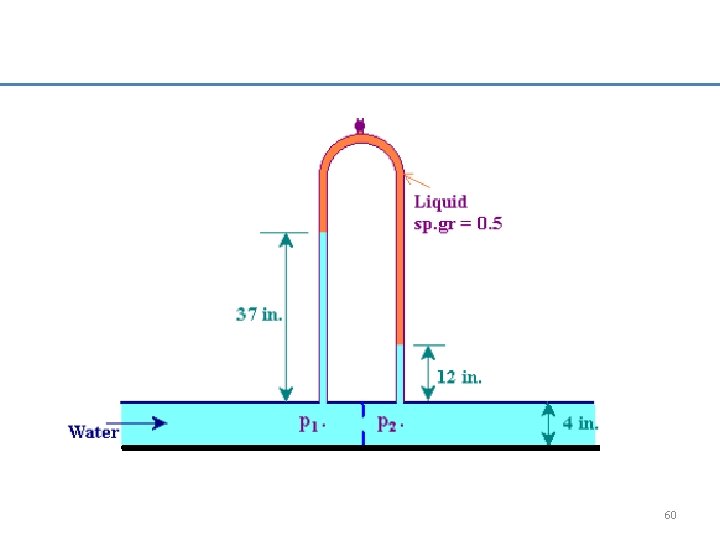
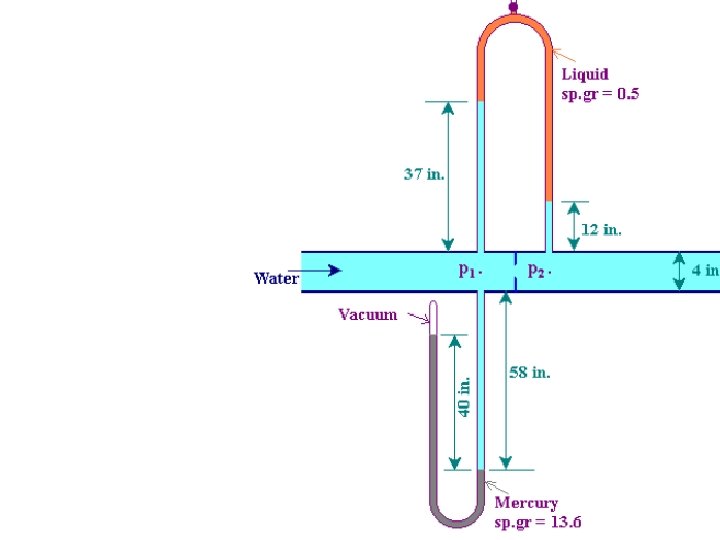
The manometers was used to measure the pressure at point 1 and 2 along a section of a piping. Please determine the value of P 1 and P 2 in Psia. 59
60
61

3 - Temperature 62

Questions are Welcome 63

KINGDOM OF SAUDI ARABIA Ministry of Higher Education KING ABDULAZIZ UNIVERSITY Faculty of Engineering Rabigh Branch ﻛﻠﻴﺔ ﺍﻟﻬﻨﺪﺳﺔ ﺑﺮﺍﺑﻎ Introduction to Chemical Engineering “Ch. E 201” Lecture 5 “Material Balance” Assis. Prof. Mohamed Ismail Bassyouni “Head of the Chemical Engineering Department” 64 Oct. 2011

Law of conservation The mass can neither be created nor destroyed 65

Process Classification 1. Batch Process The feed is charged (fed) into a vessel at the beginning of the process and the vessel contents are removed sometime later. No mass crosses the system boundaries between the time the feed is charged and the time the product is removed. 2. Continuous Process The inputs and outputs flow continuously throughout the duration of the process. 3. Semibatch process Any process that is neither batch nor continuous 66

Process Classification Batch Process Continuous Process Semi batch Process Example Rapidly add reactants to a tank and remove the products and unconsumed reactants sometime later when the system has come to equilibrium. Pump a mixture of liquids into a distillation column at a constant rate and steadily withdraw product streams from the top and bottom of the column. Allow the contents of a pressurized gas container to escape to the atmosphere; slowly blend several liquids in a tank from which nothing is being withdrawn. 67

Steady State and Unsteady State (Transient Process) i. Steady State Processes • If the values of all the variables in a process (i. e. , all temperatures, pressures, volumes, flow rates) do not change with time, except possibly for minor fluctuations about constant mean values, the process is said to be operating at steady state. ii. Unsteady State (Transient Process) • If any of the process variables change with time, transient or unsteady-state operation is said to exist. Continuous processes are usually run as close to steady state as possible. • Unsteady-state (transient) conditions exist during the start-up of a process and following changes-intentional or otherwise-in process operation conditions 68
Steady State and Unsteady State (Transient Process) • Batch Process • Semibatch process • Continuous Process • Unsteady state or Steady state 69

Steady State and Transient Process Example 1. A balloon filled with air at a steady rate of 2 g/min 2. A bottle of milk is taken from the refrigerator and left on the kitchen table. 3. Water is boiled in an open flask. 4. Carbon monoxide and steam are fed into a tubular reactor at a steady rate and react to form carbon dioxide and hydrogen. Products and unused reactants are withdrawn at the other end. The reactor contains air when the process is started up. The temperature of the reactor is constant, and the composition and flow rate of the entering reactant stream are also independent of time. Classify the process (a) initially and (b) after a long period of time has elapsed. Results 1. 2. 3. 4. Semibatch, Unsteady state Batch, Unsteady state Semibatch, Unsteady state (a) Continuous, non-steady state (b) Continuous , Steady State ? 70

Steady State and Transient Process Example 1. A balloon filled with air at a steady rate of 2 g/min 2. A bottle of milk is taken from the refrigerator and left on the kitchen table. 3. Water is boiled in an open flask. 4. Carbon monoxide and steam are fed into a tubular reactor at a steady rate and react to form carbon dioxide and hydrogen. Products and unused reactants are withdrawn at the other end. The reactor contains air when the process is started up. The temperature of the reactor is constant, and the composition and flow rate of the entering reactant stream are also independent of time. Classify the process (a) initially and (b) after a long period of time has elapsed. Results 1. 2. 3. 4. Semibatch Batch Semibatch (a) Continuous, non-steady state (b) Continuous , Steady State 71

Open and Closed Systems For Systems: We must define the boundary of the system Systems are of two types : closed and open Closed system : material is not crossing the boundary Open system: material is crossing the boundary 72
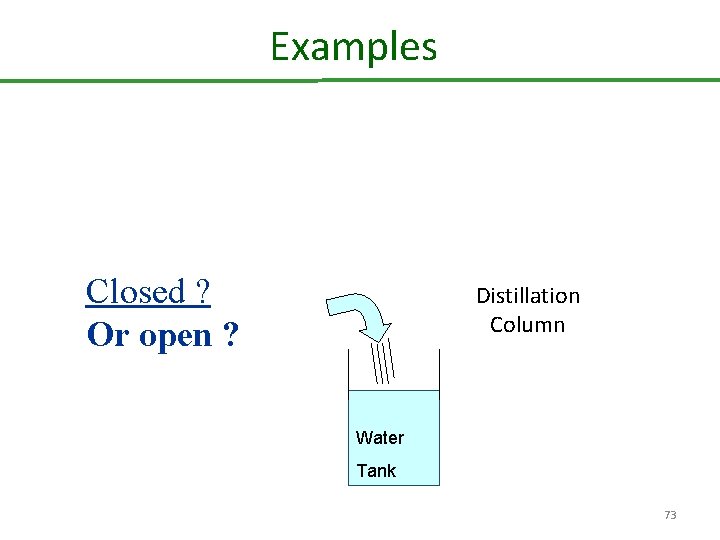
Examples Closed ? Or open ? Distillation Column Water Tank 73

The General Balance Equation moin (CH 4 Kg/hr) Process Unit moout (CH 4 Kg/hr) Suppose methane is a component of both the input and output streams of a continuous process unit, and that in an effort to determine whether the unit is performing as designed, the mass flow rates of methane in both streams are measured and found to be different There are several possible explanations for the observed difference between the measured flow rates: 1. Methane is being consumed as a reactant or generated as a product within the unit. 2. Methane is accumulating in the unit-possibly adsorbing on the walls. 3. Methane is leaking from the unit. 4. The measurements are wrong. 74

Balance on Conserved Quantities Differential balances, or balances that indicate what is happening in a system at an instant in time. Each term of the balance equation is a rate (rate of input, rate of generation, etc. ) and has units of the balanced quantity unit divided by a time unit (people/yr, g SO 2/s, barrels/day). This is the type of balance usually applied to a continuous process. Integral balances, or balances that describe what happens between two instants of time. Each term of the equation is an amount of the balanced quantity and has the corresponding unit (people, g SO 2, barrels). This type of balance is usually applied to a batch process, with the two instants of time being the moment after the input takes place and the moment before the product is withdrawn. 75

Each year 50, 000 people move into a city, 75, 000 people move out, 22, 000 are born, and 19, 000 die. Write a balance on the population of the city Let P denote people: input + generation - output - consumption = accumulation Each year the city's population decreases by 22, 000 people. 76

Material Balance Rules The following rules may be used to simplify the material balance equation: 1. If the balanced quantity is total mass, set generation = 0 and consumption = 0. Except in nuclear reactions, mass can neither be created nor destroyed. 1. If the balanced substance is a nonreactive species (neither a reactant nor a product), set generation = 0 and consumption = 0. 1. If a system is at steady state, set accumulation = 0, regardless of what is being balanced. By definition, in a steady-state system nothing can change with time, including the amount of the balanced quantity. 77

Balances on Continuous (Steady-State), and Batch (Integral balance) Processes For continuous processes at steady-state, the accumulation term in the general balance equation equals zero, and the equation simplifies to Input + Generation = Output + Consumption For integral balance on batch process Accumulation = final output – initial input = generation – consumption Equating these two expression for the accumulation field Input + Generation = Output + Consumption 78

Questions are Welcome 79

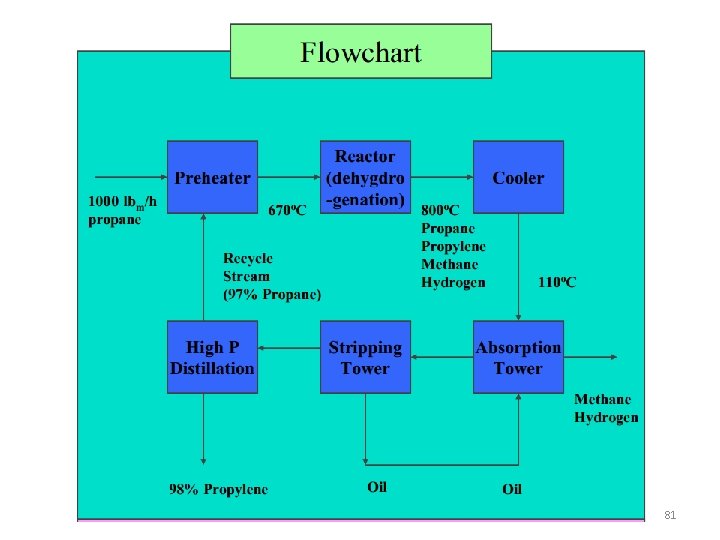
Developing Flowchart to Solve the Problem The catalytic dehydrogenation of propane is carried out in a continuous packed bed reactor. One thousand kilograms per hour of pure propane is preheated to a temperature of 670 o. C before it passes into the reactor. The reactor effluent gas, which includes propane, propylene, methane, and hydrogen, is cooled from 800°C to 110 o. C and fed to an absorption tower, where the propane and propylene are dissolved in oil. The oil then goes to a stripping tower in which it is heated, releasing the dissolved gases; these gases are recompressed and sent to a distillation column in which the propane and propylene are separated. The propane stream is recycled back to join the feed to the reactor preheater. The product stream from the distillation column contains 98% propylene, and the recycle stream is 97% propane. The stripped oil is recycled to the absorption tower. 80
81
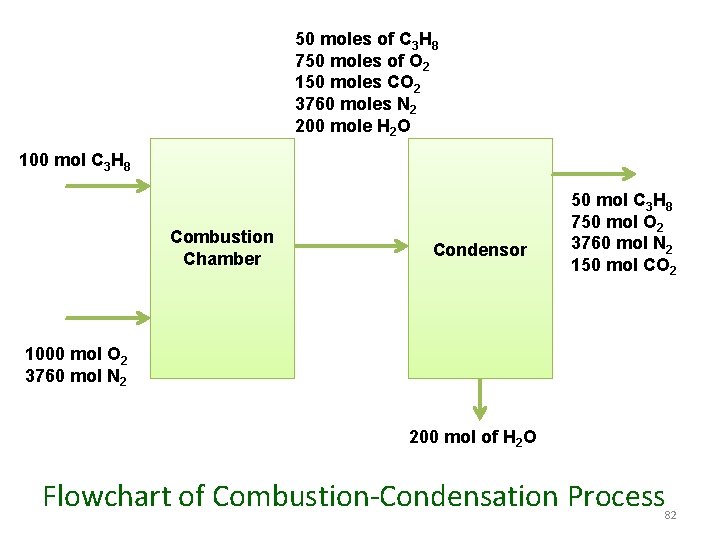
50 moles of C 3 H 8 750 moles of O 2 150 moles CO 2 3760 moles N 2 200 mole H 2 O 100 mol C 3 H 8 Combustion Chamber Condensor 50 mol C 3 H 8 750 mol O 2 3760 mol N 2 150 mol CO 2 1000 mol O 2 3760 mol N 2 200 mol of H 2 O Flowchart of Combustion-Condensation Process 82

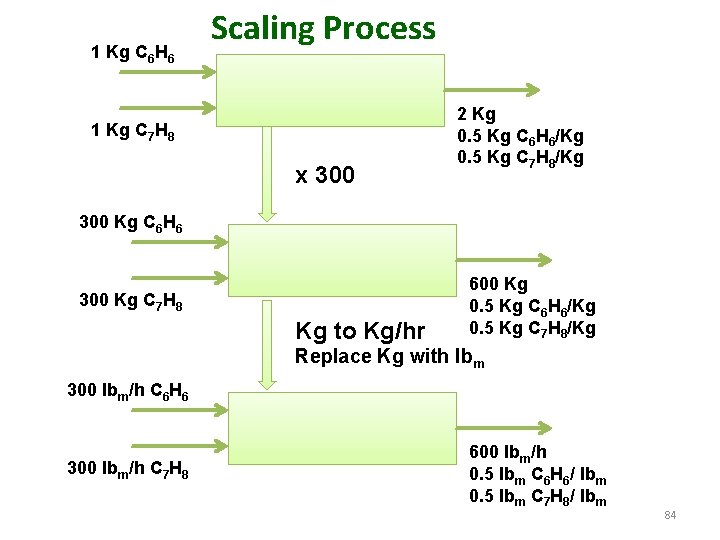
Flowchart scaling and basis of calculation The procedure of changing the values of all stream amounts or flow rates by a proportional amount while leaving the stream compositions unchanged is referred to as scaling the flowchartscaling up if the final stream quantities are larger than the original quantities, scaling down if they are smaller. Example: Suppose a kilogram of benzene is mixed with a kilogram of toluene. The output from this simple process is obviously 2 kg of a mixture that is 50% benzene by mass. 1 Kg C 6 H 6 1 Kg C 7 H 8 2 Kg 0. 5 Kg C 6 H 6/Kg 0. 5 Kg C 7 H 8/Kg 83
1 Kg C 6 H 6 Scaling Process 1 Kg C 7 H 8 x 300 2 Kg 0. 5 Kg C 6 H 6/Kg 0. 5 Kg C 7 H 8/Kg 300 Kg C 6 H 6 300 Kg C 7 H 8 Kg to Kg/hr 600 Kg 0. 5 Kg C 6 H 6/Kg 0. 5 Kg C 7 H 8/Kg Replace Kg with lbm 300 lbm/h C 6 H 6 300 lbm/h C 7 H 8 600 lbm/h 0. 5 lbm C 6 H 6/ lbm 0. 5 lbm C 7 H 8/ lbm 84

Basis of Calculation • A basis of calculation is an amount (mass or moles) or flow rate (mass or molar) of one stream or stream component in a process. The first step in balancing a process is to choose a basis of calculation; all unknown variables are then determined to be consistent with this basis. • If a stream amount or flow rate is given in a problem statement, it is usually most convenient to use this quantity as a basis of calculation. • If no stream amounts or flow rates are known, assume one, preferably that of a stream with a known composition. If mass fractions are known, choose a total mass or mass flow rate of that stream (e. g. 100 kg or 100 kg/h) as a basis; if mole fractions are known, choose a total number of moles or a molar flow rate. 85

Balancing a Process 1. The maximum number of independent equations that can be derived by writing balances on a nonreactive system equals the number of chemical species in the input and output streams. 2. Write balances first that involve the fewest unknown variables. 86

Degree-of-Freedom Analysis • Before you do any lengthy calculations, you can use a properly drawn and labeled flowchart to determine whether you have enough information to solve a given problem. The procedure for doing so is referred as Degree-of-Freedom Analysis • To perform a degree-of-freedom analysis, draw and completely label a flowchart, count the unknown variables on the chart, then count the independent equations relating them, andsubtract the second number from the first. The result is the number of degrees of freedom of the process. ndf = nunknowns – nindependent eqns • There are three possibilities: 87

Degree-of-Freedom Analysis 1. If ndf = 0, there are n independent equations in n unknowns and the problem can in principle be solved. 2. If ndf > 0, there are more unknowns than independent equations relating them, and at least ndf additional variable values must be specified before the remaining variable values can be determined. 3. If ndf < 0, there are more independent equations than unknowns. Either the flowchart is incompletely labeled or the problem is overspecified with redundant and possibly inconsistent relations. 88

Degree-of-Freedom Analysis Sources of equations relating unknown process stream variables include the following 1. 2. 3. 4. 5. 6. Material balances An Energy Balance Process Specifications Physical properties and laws Physical Constraints Stoichiometric relations 89

General Procedure for Single-Unit Process Material Balance Calculations 1. Choose as a basis of calculation an amount or flow rate of one of the process streams. 2. Draw flowchart and fill in all known variable values, including the basis of calculation. Then label unknown stream variables on the chart. 3. Express what the problem statement asks you to determine in terms of the labeled variables. 4. If you are given mixed mass and mole units for a stream (such as a total mass flow rate and component mole fractions or vice versa), convert all quantities to one basis or the other. 5. Do the degree-of-freedom analysis. 90

General Procedure for Single-Unit Process Material Balance Calculations 6. If the number of unknowns equals the number of equations relating them (i. e. , if the system has zero degrees of freedom), write the equations in an efficient order (minimizing simultaneous equations) and circle the variables for which you will solve 7. Solve the equations 8. Calculate the quantities requested in the problem statement if they have not already been calculated. 9. If a stream quantity or flow rate ng was given in the problem statement and another value nc was either chosen as a basis or calculated for this stream, scale the balanced process by the ratio ng / nc to obtain the final result. 91

Recycle and Bypass There are several reasons for using recycle in a chemical process 1. Recovery and reusing unconsumed reactants 2. Recovery of catalyst 3. Dilution of a process stream 4. Control of process variable 5. Circulation of a working fluid A procedure that has several features in common with recycle is bypass, in which a fraction of the feed to a process unit is diverted around the unit and combined with the output stream from the unit. By varying the fraction of the feed that is bypassed, we can vary the composition and properties of the product. 92
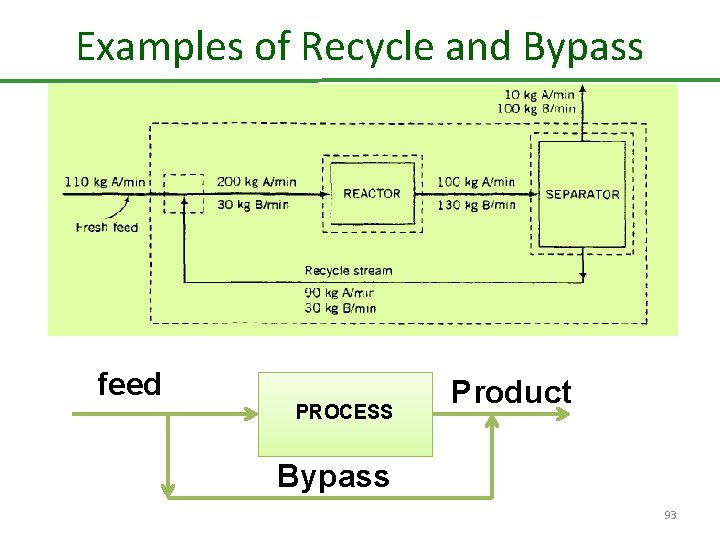
Examples of Recycle and Bypass feed PROCESS Product Bypass 93

Chemical Reaction Stoichiometry • Stoichiometry is theory of the proportions in which chemical species combine with one another. The stoichiometric equation of a chemical reaction is a statement of the relative number of molecules or moles of reactants and products that participate in the reaction. 2 SO 2 + O 2 2 SO 3 • For every two molecules (g-moles, Ib-moles) of S 02 that react, one molecule (g-mole, lb-mole) of O 2 reacts to produce two molecules (g-moles, lb-moles) of S 03. The numbers that precede the formulas for each species are the stoichiometric coefficients of the 94 reaction components.

Chemical Reaction Stoichiometry • A valid stoichiometric equation must be balanced; that is, the number of atoms of each atomic species must be the same on both sides of the equation, since atoms can neither be created nor destroyed in chemical reactions • The stoichiometric ratio of two molecular species participating in a reaction is the ratio of their stoichiometric coefficients in the balanced reaction equation. This ratio can be used as a conversion factor to calculate the amount of a particular reactant (or product) that was consumed (or produced), given a quantity of another reactant of product that participated in the reaction 95

Limiting and Excess Reactants • Two reactants, A and B, are said to be present in stoichiometric proportion if the ratio (moles A present)/(mole B present) equals the stoichiometric ratio obtained from the balanced reaction • The reactant that would run out if a reaction proceeded to completion is called the limiting reactant, and the other reactants are termed excess reactants. A reactant is limiting if it is present in less than its stoichiometric proportion relative to every other reactant. 96

Chemical Reaction The Limiting reactant In chemical reaction one of the reactant will consume first so it is called the limiting reagent Other reactants remain in excess The amount of product determined by the amount of limiting reagent

Chemical Reaction Determination of Limiting Reactant 1 - Calculate the number of moles of each reactant (if not given) 2 - Divide that moles to there factors in chemical reaction (ratio) 3 - Compare the ratios, the smallest is the limiting reagent

Chemical Reaction Determination of Limiting Reactant Example Determine the limiting reactant for the following reaction if 400 g Fe mixed with 300 g O 2. 400/56 = 7. 14 300/32 = 9. 375 Stoichiometric amount of oxygen = 7. 14*3/4= 5. 25 moles < 9. 375 So Fe is limiting reactant.

Chemical Reaction The Reaction Yield Theoretical Yield (T. Y. ): the amount of product produced when all limiting reagent reacted, or it is the yield calculated from equation Actual Yield (A. Y. ): the amount of product obtained actually Due to many factors affecting on reaction, A. Y. is always less than T. Y. so we can calculate the % yield

Chemical Reaction The Reaction Yield Example Calculate the % yield of urea, if 119 g of ammonia reacts with 80 g of CO 2 producing 100 g of urea according to the following equation % yield = actual/ theoritical = 100 g/109. 9 = 90. 99%

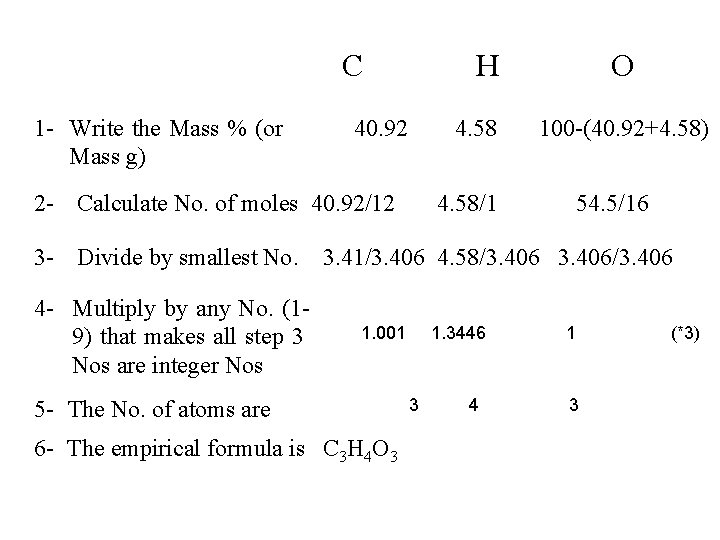
Combustion Analysis Determination of Empirical Formula ascorbic acid contains 40. 92% C and 4. 58% H on combustion (note that % O = 100 – 40. 92 – 4. 58 = 54. 5 %) The empirical formula can be determined as follows MASS MOLE RATIO
C H 1 - Write the Mass % (or 40. 92 4. 58 Mass g) O 100 -(40. 92+4. 58) 2 - Calculate No. of moles 40. 92/12 4. 58/1 54. 5/16 3 - Divide by smallest No. 3. 41/3. 406 4. 58/3. 406 3. 406/3. 406 4 - Multiply by any No. (19) that makes all step 3 Nos are integer Nos 1. 001 5 - The No. of atoms are 6 - The empirical formula is C 3 H 4 O 3 1. 3446 3 4 1 3 (*3)

Determine its empirical formula for 0. 15 mole H , 0. 05 mole C, and 0. 025 mole O 3 - Divide by smallest No. 0. 15/0. 025 0. 025/0. 025 4 - Multiply by any No. (19) that makes all step 3 Nos are integer Nos 6 5 - The No. of atoms are 6 - The empirical formula is C 2 H 6 O 2 1

Combustion Analysis Determination of Molecular formula Relation between molecular formula and empirical formula is

Combustion Analysis Determination of Molecular formula Example What is the molecular formula of ascorbic acid if its molecular weight is 176 g/mol

Chemical Reaction It is a process in which one or more substances converted to other substances This change involve a change in energy Only change are happened no matter or energy created or destroyed The physical conditions can be written in the chemical equation (s) for Solid (l) for Liquid (g) for Gas (aq) for Aqueous solution (ppt) for precipitation

Chemical Reaction The chemical equation must be balanced Balancing Chemical Equation e. g. balance the following equation

H. W. What is the coefficient of H 2 O when the equation is balanced: _ Al 4 C 3 + _ H 2 O _ Al(OH)3 + 3 CH 4 a. 13 b. 4 c. 6 D 12 H. W. What are the coefficients of Al 4 C 3 , H 2 O and Al(OH)3, respectively, when the equation is balanced: _ Al 4 C 3 + _ H 2 O _ Al(OH)3 + 3 CH 4 a. 4, 1, 5 B 1, 12, 4 a. 1, 24, 4 b. 4, 12, 1 109

Chemical Reaction Stoichiometry of Reaction The following equation can be read as follows 1 molecule of sugar + 6 molecules of O 2 gas gives 6 molecules of CO 2 and 6 molecules of water 1 mole of sugar + 6 moles of O 2 gas gives 6 moles of CO 2 and 6 moles of water 180 g of sugar + 192 g of O 2 gas gives 264 g of CO 2 and 108 g of water

Chemical Reaction Stoichiometry of Reaction Not as Follows 1 g of sugar + 6 g of O 2 gas gives 6 g of CO 2 and 6 g of water So this numbers means the number of moles or number of molecules not the number of grams, if you want the number of grams you must convert the number of moles to grams using the molar mass (Mw or Aw) and vice versa

Chemical Reaction The Amount of Reactants and Product The amount of reactants or products can be calculated from balanced chemical equation The excess of one of reactants can’t do anything for the amount of product produced Example How many grams of water produced when 7. 00 g of oxygen react with excess of hydrogen?

Chemical Reaction

Chemical Reaction Examples 1 -Calculate the number of moles of CO 2 resulted from the reaction of 4 moles of C 2 H 6 with excess oxygen according to the equation

Chemical Reaction Examples 2 -Calculate the mass of chlorine that reacts with 1. 220 g of hydrogen to form hydrogen chloride according the following equation:

Questions are Welcome 116

KINGDOM OF SAUDI ARABIA Ministry of Higher Education KING ABDULAZIZ UNIVERSITY Faculty of Engineering Rabigh Branch ﻛﻠﻴﺔ ﺍﻟﻬﻨﺪﺳﺔ ﺑﺮﺍﺑﻎ Introduction to Chemical Engineering “Ch. E 201” Lecture 5 Chemical Equilibria (Stoichiometry) Assis. Prof. Mohamed Ismail Bassyouni Syed Waheed ul Hasan 117 Dec. 2011

Chemical Equilibrium Given a set of reactive species and reaction conditions: (a) The field of chemical equilibrium thermodynamics concerns itself with what will be the final (equilibrium) composition of the reaction mixture (b) Chemical kinetics deals with how long will the system take to reach a specified state short of equilibrium? Irreversible Reaction: The reaction that proceeds only in a single direction (from reactants to products) and the concentration of the limiting reactant eventually approaches zero, is called an irreversible reaction. 118

Chemical Equilibrium Reversible Reaction: In a reversible reaction, reactants form products and products undergo the reverse reactions to reform the reactants. For example, consider the reaction in which ethylene is hydrolyzed to ethanol: C 2 H 4 + H 2 O C 2 H 5 OH 119

Calculation of an Equilibrium Composition If the water-gas shift reaction: CO(g)+H 2 O(g) CO 2(g)+H 2(g) proceeds to equilibrium at a temperature T(K), the mole fractions of the four reactive species satisfy the relation: where K(T) is the reaction equilibrium constant. At T = 1105 K, K = 1. 00. Suppose the feed to a reactor contains 1. 00 mol of CO, 2. 00 mol of H 2 O, and no CO 2 or H 2, and the reaction mixture comes to equilibrium at 1105 K. Calculate the equilibrium composition and the fractional conversion of the limiting reactant. 120

Solution The strategy is to express all mole fractions in terms of a single variable (ξe, the extent of reaction at equilibrium), substitute in the equilibrium relation, solve for ξe, and back-substitute to calculate the mole fractions and any other desired quantity. From the definition of extent of reaction, we can write Number of gram mole of CO present at equilibrium: n. CO = 1 mol – ξe Similarly n. H 2 O = 2 mol - ξe n. CO 2 = ξe n. H 2 = ξe 121

Solution ntotal = 3 mol From which the composition of the reaction mixture can be given by: y. CO = (1 mol – ξe) / 3 mol Similarly y. H 2 O = (2 mol - ξe ) / 3 mol y. CO 2 = (ξe ) / 3 mol y. H 2 = (ξe ) / 3 mol Substitution of these expressions into the equilibrium relation (with K = 1. 00) yields 122

Solution This equation may be rewritten as a standard quadratic equation ξe 2= 2 - 3ξe + ξe 2 2 - 3 ξe = 0 ξe = 2/3 ξe = 0. 667 mol. This quantity may in turn be substituted back into the expression for yi to yield y. CO = 0. 111 y. H 2 O = 0. 444 y. CO 2 = 0. 222 y. H 2 = 0. 222 123

Solution Finding the limiting reactant: Mole of CO provided = 1 Moles of CO/ stoichiometric coefficient = 1/1 Mole of H 2 O provided = 2 Moles of H 2 O/ stoichiometric coefficient = 2/1 = 2 Since the moles fed over stoichiometric coefficient of CO is smaller than water, its our limiting reactant. 124

Solution At equilibrium n. CO = 1 mol – ξe n. CO = 1 - 0. 667 mol = 0. 333 mol The fractional conversion of CO at equilibrium: fco = (1 -0. 333) mol CO reacted / (I mol CO fed) fco = 0. 667 125

Multiple Reactions Reactants can usually combine in more than one way, and the product once formed may react to yield something less desirable. This Truth results in (a) Economic loss (b) A greater quantity of raw materials must be fed to the reactor to obtain a specified product yield. 126
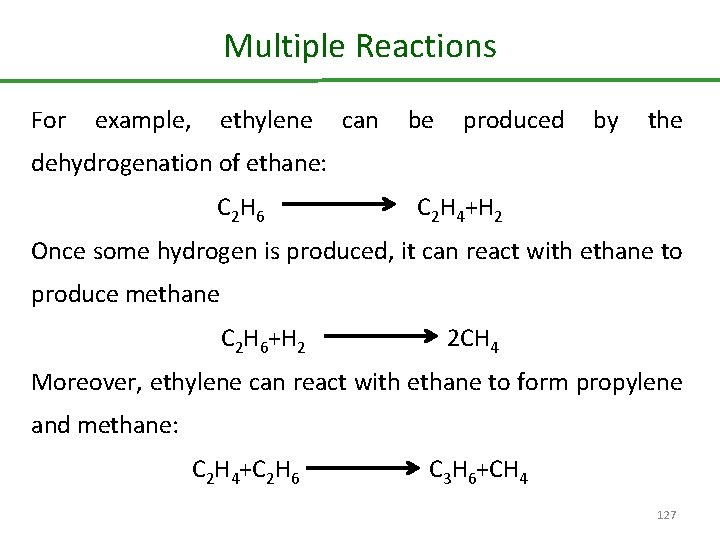
Multiple Reactions For example, ethylene can be produced by the dehydrogenation of ethane: C 2 H 6 C 2 H 4+H 2 Once some hydrogen is produced, it can react with ethane to produce methane C 2 H 6+H 2 2 CH 4 Moreover, ethylene can react with ethane to form propylene and methane: C 2 H 4+C 2 H 6 C 3 H 6+CH 4 127

Multiple Reactions Since the object of the process is to produce ethylene, only the first of these reactions may be regarded as desirable; the second one consumes the reactant without yielding the desired product and the third consumes both the reactant and the desired product. The engineer must consider not only how to maximize the production of the desired product (C 2 H 4), but also how to minimize the production of undesired by-products (CH 4, C 3 H 6) 128

Yield and Selectivity The terms yield and selectivity are used to describe the degree to which a desired reaction predominates over competing side reactions. 129

Extent of Reactions If a set of reactions takes place in a batch or continuous steady-state reactor and vij is the stoichiometric coefficient of substance i in reaction j (negative for reactants, positive for products), we may then write ni = nio + ∑vijξj For a single reaction it reduces to ni = nio + viξ 130

Extent of Reaction (example) For example, consider the pair of reactions in which ethylene is oxidized either to ethylene oxide (desired) or to carbon dioxide (undesired): C 2 H 4+1/2 O 2 C 2 H 4+3 O 2 C 2 H 4 O 2 CO 2 + 2 H 2 O The moles (or molar flow rates) of each of the five species involved in these reactions can be expressed in terms of the feed values and extents of reaction: 131

Extent of Reaction (example) values of any two outlet amounts are given, the values of ξ 1 and ξ 2 may be determined from the corresponding two equations, and the remaining amounts may in turn be calculated from the remaining three equations. 132
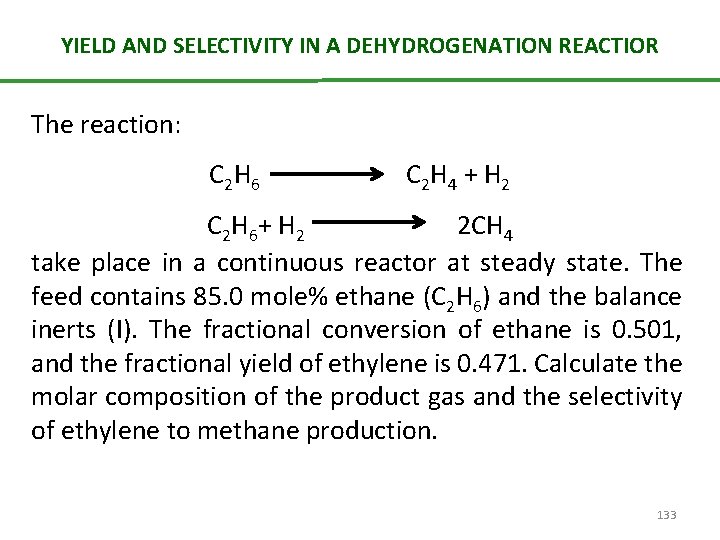
YIELD AND SELECTIVITY IN A DEHYDROGENATION REACTIOR The reaction: C 2 H 6 C 2 H 4 + H 2 C 2 H 6 + H 2 2 CH 4 take place in a continuous reactor at steady state. The feed contains 85. 0 mole% ethane (C 2 H 6) and the balance inerts (I). The fractional conversion of ethane is 0. 501, and the fractional yield of ethylene is 0. 471. Calculate the molar composition of the product gas and the selectivity of ethylene to methane production. 133
Solution Basis: 100 mol of feed 134

YIELD AND SELECTIVITY IN A DEHYDROGENATION REACTIOR Using the equation, ni = nio + ∑vijξj the outlet composition amounts in terms of extents of reaction are as follows: 135

YIELD AND SELECTIVITY IN A DEHYDROGENATION REACTIOR Etahne Conversion: If the fractional conversion of ethane is 0. 501, the fraction unconverted (and hence leaving the reactor) must be (1 -0. 501) = 42. 2 mol C 2 H 6 = 85 mol C 2 H 6 – ξ 1 - ξ 2 ------(1) Ethylene Yield: 136

YIELD AND SELECTIVITY IN A DEHYDROGENATION REACTIOR n 2 = 0. 471 (85 mol C 2 H 6) = 40 mol C 2 H 4 = ξ 1 Substituting 40 mol for ξ 1 in equation 1, we get ξ 2 as 2. 6 mol. Then Once we know the number of moles, the percentage composition of each species can be given as following: 137

YIELD AND SELECTIVITY IN A DEHYDROGENATION REACTIOR Product C 2 H 6 C 2 H 4 H 2 CH 4 I Composition (%) 30. 3 28. 6 26. 7 3. 7 10. 7 SELECTIVITY: = (40 mol C 2 H 4)/ (5. 2 mol CH 4) = 7. 7 mol C 2 H 4/mol CH 4 138
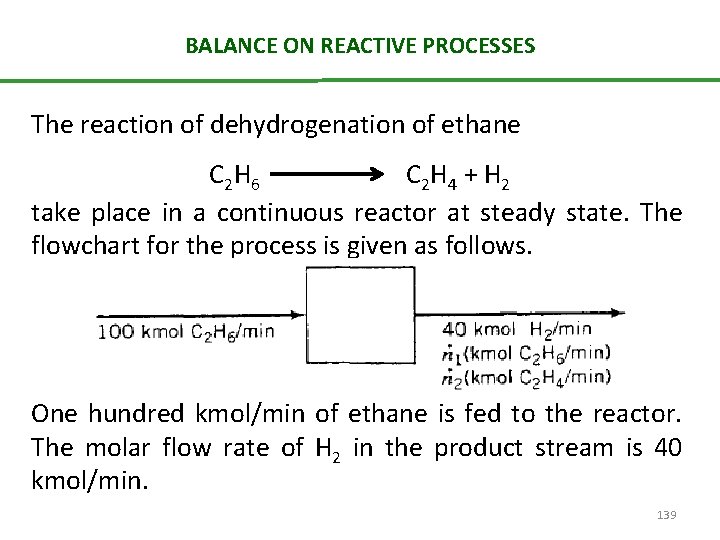
BALANCE ON REACTIVE PROCESSES The reaction of dehydrogenation of ethane C 2 H 6 C 2 H 4 + H 2 take place in a continuous reactor at steady state. The flowchart for the process is given as follows. One hundred kmol/min of ethane is fed to the reactor. The molar flow rate of H 2 in the product stream is 40 kmol/min. 139

BALANCE ON REACTIVE PROCESSES A number of different balances could be written on this process. 1. balances on total mass, 2. C 2 H 6 3. C 2 H 4 4. H 2 • The first has the simple form input = output • The species balance must include a generation term (for C 2 H 4 and H 2) or a consumption term (for C 2 H 6). • Notice, however, that balance equations may also be written for atomic carbon and atomic hydrogen, regardless of the molecular species in which the carbon 140 and hydrogen atoms happen to be found

BALANCE ON REACTIVE PROCESSES • Balances on atomic species can be written input=output, since atoms can neither be created (generation = 0) nor destroyed (consumption = 0) in a chemical reaction • Molecular hydrogen balance is different from the atomic hydrogen balance 141
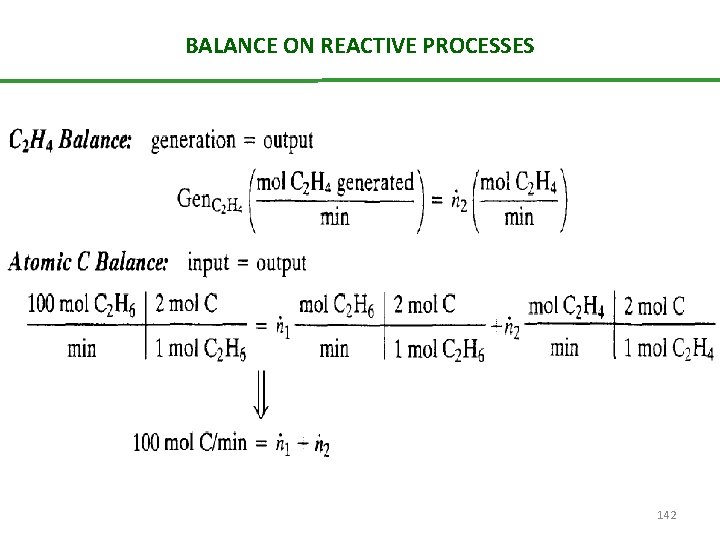
BALANCE ON REACTIVE PROCESSES 142
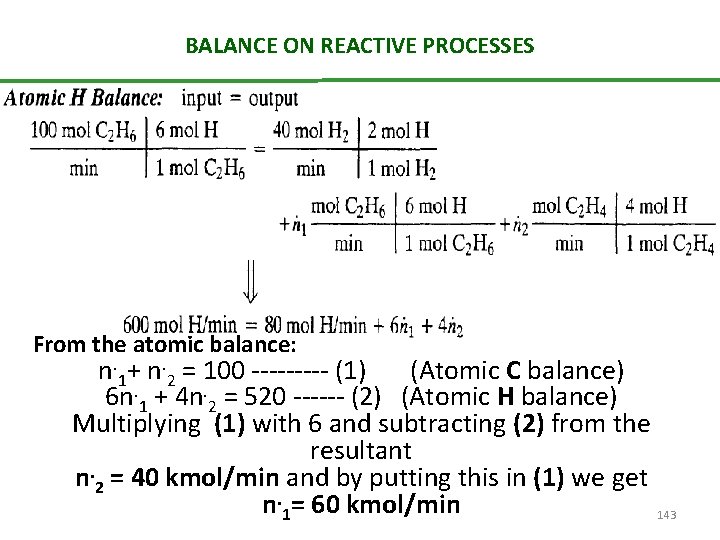
BALANCE ON REACTIVE PROCESSES From the atomic balance: n. 1+ n. 2 = 100 ----- (1) (Atomic C balance) 6 n. 1 + 4 n. 2 = 520 ------ (2) (Atomic H balance) Multiplying (1) with 6 and subtracting (2) from the resultant n. 2 = 40 kmol/min and by putting this in (1) we get n. 1= 60 kmol/min 143

BALANCE ON REACTIVE PROCESSES In general, systems that involve chemical reactions may be analyzed using (a) molecular species balances (the approach always used for nonreactive systems) (b) atomic species balances (c) extents of reaction Each approach leads to the same results, but anyone of them may be more convenient for a given calculation so 144

Bonus Question (+1) In general, systems that involve chemical reactions may be analyzed using (a) molecular species balances (the approach always used for nonreactive systems) (b) atomic species balances (c) extents of reaction Each approach leads to the same results, but anyone of them may be more convenient for a given calculation so 145

KINGDOM OF SAUDI ARABIA Ministry of Higher Education KING ABDULAZIZ UNIVERSITY Faculty of Engineering Rabigh Branch ﻛﻠﻴﺔ ﺍﻟﻬﻨﺪﺳﺔ ﺑﺮﺍﺑﻎ Introduction to Chemical Engineering “Ch. E 201” Lecture 6 Combustion Assis. Prof. Mohamed Ismail Bassyouni Syed Waheed ul Hasan 146 Dec. 2011

Combustion: The rapid reaction of a fuel with oxygen Important Fuel: Petroleum products, Coal, Gases like methane, LPG Partial Combustion: A combustion reaction in which CO is formed from a hydrocarbon is referred to as partial combustion or incomplete combustion of the hydrocarbon. 147

Composition of Stack Gases Composition on wet Basis The term composition on a wet basis is commonly used to denote the component mole fractions of a gas that contains water Composition on dry Basis Composition on a dry basis signifies the component mole fractions of the same gas without the water Gas analyzer for stack gases The product gas that leaves a combustion furnace is referred to as the stack gas or flue gas. Common techniques for analyzing stack gases provide compositions on a dry basis. 148

Flue or stack gas analyzer 149

Conversion from wet to dry basis A stack gas contains 60. 0 mole % N 2, 15. 0% CO 2, 10. 0% O 2, and the balance H 2 O. Calculate the molar composition of the gas on a dry basis. Solution: Basis: 100 mol wet gas Moles of N 2 = 60 Moles of CO 2 = 15 Moles of O 2 = 10 Total moles without considering water = 85 mol dry gas Composition of dry gas: fraction of nitrogen = 60 / 85 = 0. 706 Fraction of carbon dioxide = 16 / 85 = 0. 176 150 Fraction of oxygen = 10 / 85 = 0. 118

Conversion from dry to wet basis An Orsat analysis (a technique for stack analysis) yields the following dry basis composition: N 2 = 65 % CO 2 = 14 % CO = 11 % O 2 = 10 % A humidity measurement shows that the mole fraction of H 2 O in the stack gas is 0. 0700. Calculate the stack gas composition on a wet basis. 151
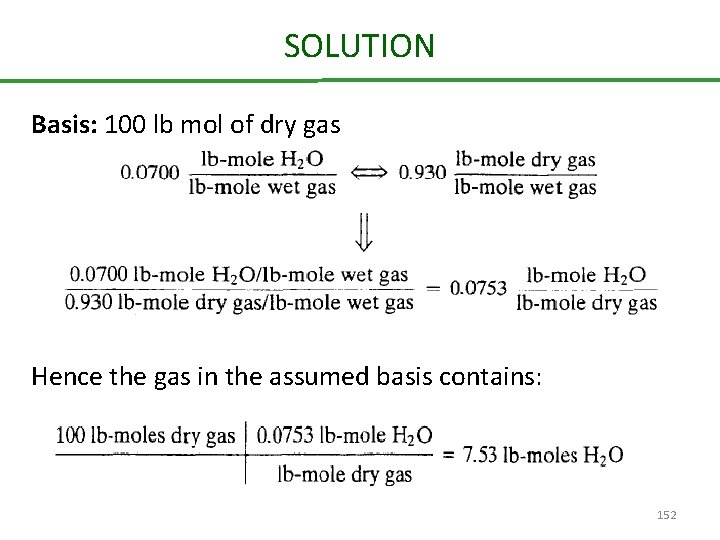
SOLUTION Basis: 100 lb mol of dry gas Hence the gas in the assumed basis contains: 152

Solution 153

Theoretical and Excess Reactant One hundred mol/h of butane (C 4 H 10 ) and 5000 mol/h of air are fed into a combustion reactor. Calculate the percent excess air. Solution: First, calculate theoretical air from the feed rate of fuel and the stoichiometric equation for complete combustion of butane 154
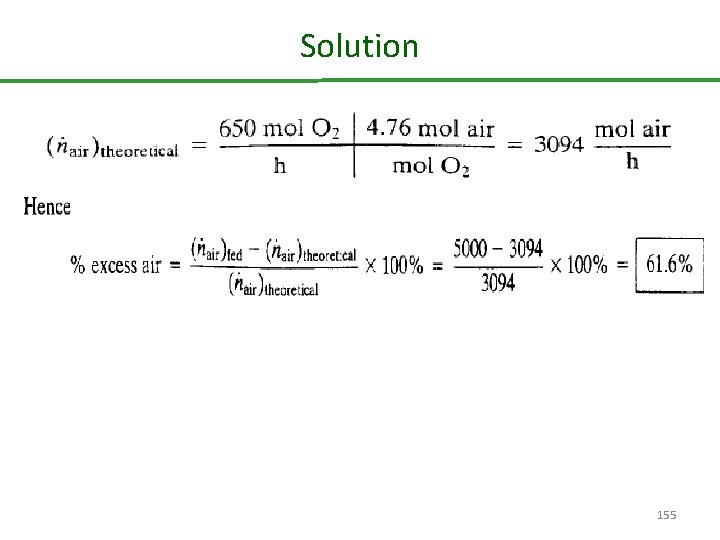
Solution 155

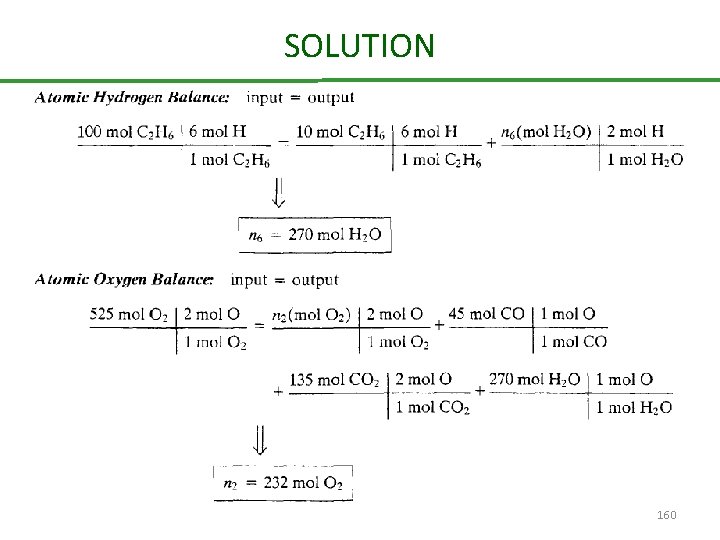
Combustion of ethane Ethane is burned with 50% excess air. The percentage conversion of the ethane is 90% of the ethane burned. 25% reacts to form CO and the balance reacts to form CO 2. Calculate the molar composition of the stack gas on a dry basis and the mole ratio of water to dry stack gas. 156
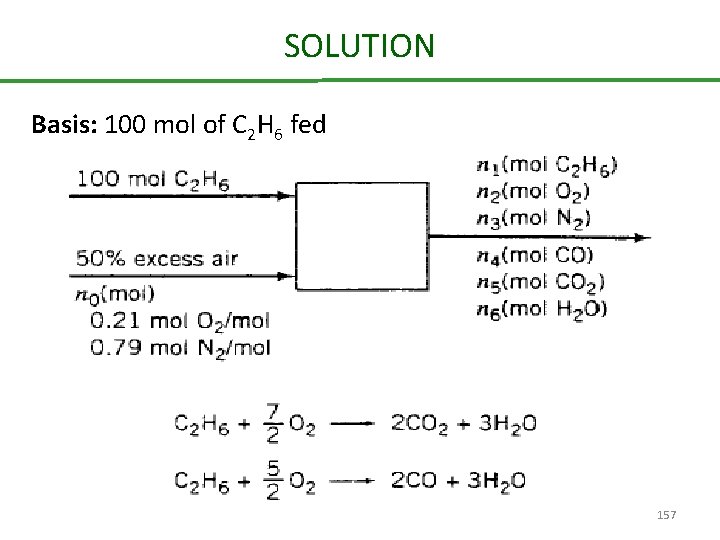
SOLUTION Basis: 100 mol of C 2 H 6 fed 157

SOLUTION 158
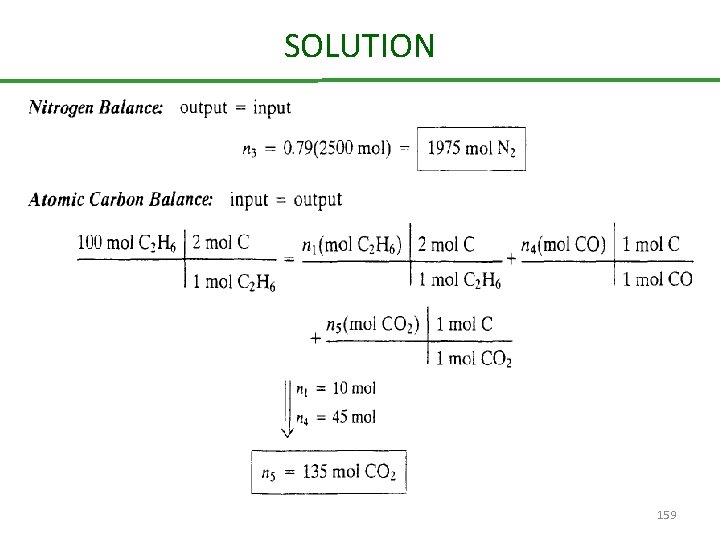
SOLUTION 159
SOLUTION 160

SOLUTION 161

SOLUTION 162
- Slides: 162