Kinetics Thermodynamics and Equilibrium Kinetics and Thermodynamics n

Kinetics , Thermodynamics and Equilibrium

Kinetics and Thermodynamics n n Kinetics: deals with rates of reactions (how quickly a reaction occurs) Thermodynamics: involves changes in energy that occur in reactions

Kinetics: Collision Theory n Frequency of collisions: n n more collisions = faster rate of reaction Effective collisions: must have: 1) proper orientation 2) enough energy

Factors Affecting Rate 1. Type of substance: n Ionic substances react faster: bonds require less energy to break Ag. NO 3 (aq)+Na. Cl(aq) Ag. Cl(s)+Na. NO 3 (aq) In solution ionic solids dissociate into ions: Ag+ NO 3 Na+ Cln Covalent react more slowly: bonds require more energy to break H 2 (g)+I 2 (g) 2 HI (g) Bonds must be broken then be reformed. (takes more time)

Factors Affecting Rate 2. Temperature increase n Average kinetic energy increases and the number of collisions increases. Reactants have more energy when colliding. This increases rate.

Factors Affecting Rate 3. Concentration increase n Increases rate due to the fact that more particles are in a given volume, which creates more collisions.

Factors Affecting Rate 4. Surface Area Increase n Increases rate due to increased reactant interaction or collisions (powder vs. lump)

Factors Affecting Rate 5. Pressure Increases n Increases the rate of reactions involving gases only As pressure Volume so: spaces between molecules frequency of effective collisions

Factors Affecting Rate 6. Catalyst: substance that increases rate of reaction, provides a shorter or alternate pathway by lowering the activation energy of the reaction. n Catalysts remain unchanged during the reaction and can be reused. n Activation energy: amount of energy required to “start” a reaction

Quick Review – Factors that affect reactions n n n Ionic solutions have faster reactions than molecule compounds. (bonding) Temp. Rate conc. rate surface area rate Pressure rate, P rate Catalysts speed up reactions.

Potential Energy Diagrams n Graphs heat energy during the course of a reaction.


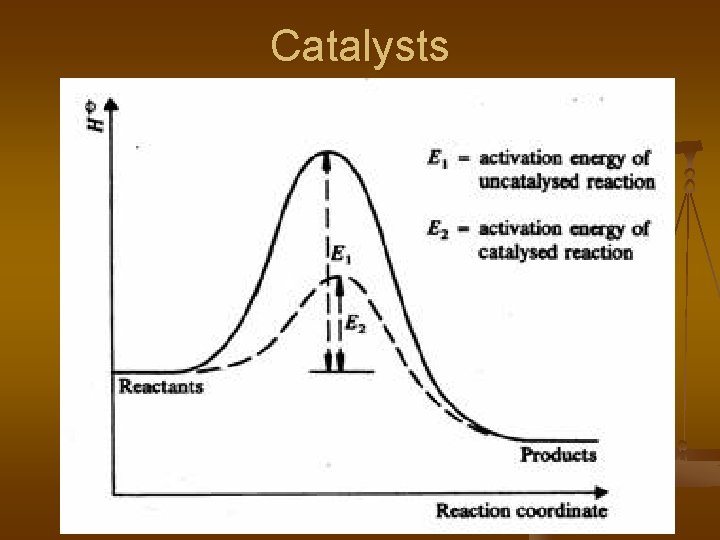
Catalysts

Thermodynamics n n Heat content (Enthalpy): amount of heat absorbed or released in a chemical reaction Enthalpy (ΔH = Hproducts – Hreactants)

ΔH = Hproducts – Hreactants n n ΔH is positive when the reaction is endothermic. Heat of products are greater than reactants ΔH is negative when the reaction is exothermic. Heat of reactants were greater than the products

Entropy (ΔS) n n n Definition: randomness, disorder in a sample of matter Gases have high entropy Solids have low entropy

Increasing ΔS n n n Phase change from s l g Mixing gases Dissolving a substance

Spontaneous Reactions n n Nature favors low energy (more stable) and high entropy Reactions are spontaneous when heat (ΔH) decreases and entropy (ΔS) increases ΔH = (-) ΔS= (+)

Analogy: Your Bedroom n n n You like to have low enthalpy (low energy) when it comes to household chores. As a result, your room tends to have high entropy (very messy, disorderly). This is what nature prefers: low enthalpy and high entropy.

Stability of Products and H n n n Help determine if a reaction is spontaneous Products tend toward Lower energy (-ΔH) Products tend toward more randomness (+ΔS) Products of exothermic reactions are usually more stable. Result in lower amounts of heat. The more negative the H, the more stable the product is. Gas products result in increased Entropy.

Chemical Equilibrium

Reversible Reactions n n Most chemical reactions are able to proceed in both directions under the appropriate conditions. Example: Fe 3 O 4 (s) + 4 H 2 (g) ↔ 3 Fe(s) + 4 H 2 O(g)

Reversible Reactions cont. n n In a closed system, as products are produced they will react in the reverse reaction until the rates of the forward and reverse reactions are equal. Ratefwd = Raterev This is called chemical equilibrium.

Equilibrium n n Equilibrium is dynamic condition where rates of opposing processes are equal. Types of Equilibrium: Phase equilibrium n Solution Equilibrium n Chemical Equilibrium n

Phase Equilibrium n n n Rate of one phase change is equal to the rate of the opposing phase change. Occurs when two phases exist at the same temperature. Example: Ratemelting = Ratefreezing H 2 O (s) H 2 O (l)

Solution Equilibrium n Rate of dissolving = rate of crystallization n Occurs in saturated solutions

Chemical Equilibrium n n n Rateforward reaction = Ratereverse reaction Concentration of reactants and products are constant NOT necessarily equal. [reactants] and [products] is constant.

The Concept of Equilibrium n n As a system approaches equilibrium, both the forward and reverse reactions are occurring. At equilibrium, the forward and reverse reactions are proceeding at the same rate.

Le Chatelier’s Principle n n Whenever stress is applied to a reaction at equilibrium, the reaction will shift its point of equilibrium to offset the stress. Stresses include: n Temperature, pressure, changes in reactant or product concentrations

Example: The Haber Process N 2 (g) + 3 H 2 (g) 2 NH 3 (g) + heat a) b) c) d) e) f) g) h) [N 2] [H 2] [NH 3] pressure temperature

Example: The Haber Process N 2 (g) + 3 H 2 (g) 2 NH 3 (g) + heat a) b) c) d) e) f) g) h) [N 2] [H 2] [NH 3] pressure temperature shift towards products (right) shift towards reactants (left) shift towards products (right)

Equilibrium shifts due to stresses: n n n Concentration increase shift away from increase Concentration decrease shift toward decrease pressure shifts in direction of fewer gas molecules. pressure shifts in direction of more gas molecules temperature favors endothermic reaction n n Shift away from heat temperature favors exothermic reaction n Shift towards heat

Effect of Catalyst: n n Addition of catalysts changes the rate of both the forward and reverse reactions. There is no change in concentrations but equilibrium is reached more rapidly.

Reactions that go to completion: n n n Equilibrium is not reached if one of the products is withdrawn as quickly as it is produced and no new reactants are added. Reaction continues until reactants are used up. Products are removed if: n n Gases in liquid solution Insoluble products (precipitate)

The Haber Process n Application of Le. Chatelier’s Principle N 2 (g) + 3 H 2 (g) 2 NH 3 (g) + 92 k. J increase pressure Shift decrease Temp Shift remove NH 3 add N 2 and H 2 Shift ****Maximum yields of NH 3 occurs under high pressures, low temperatures and by constantly removing NH 3 and adding N 2 & H 2
- Slides: 36