KINETICS POTENTIAL ENERGY OF REACTION GRAPH SLIDE1 100

- Slides: 7

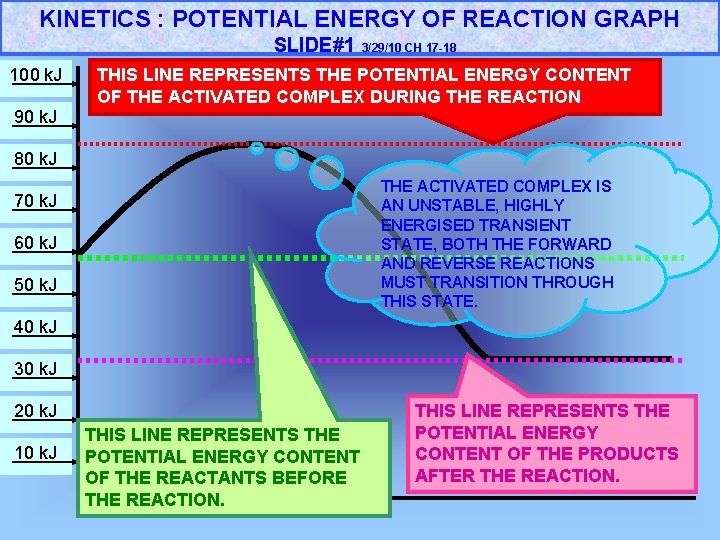
KINETICS : POTENTIAL ENERGY OF REACTION GRAPH SLIDE#1 100 k. J 3/29/10 CH 17 -18 THIS LINE REPRESENTS THE POTENTIAL ENERGY CONTENT OF THE ACTIVATED COMPLEX DURING THE REACTION. 90 k. J 80 k. J THE ACTIVATED COMPLEX IS AN UNSTABLE, HIGHLY ENERGISED TRANSIENT STATE, BOTH THE FORWARD AND REVERSE REACTIONS MUST TRANSITION THROUGH THIS STATE. 70 k. J 60 k. J 50 k. J 40 k. J 30 k. J 20 k. J 10 k. J THIS LINE REPRESENTS THE POTENTIAL ENERGY CONTENT OF THE REACTANTS BEFORE THE REACTION. THIS LINE REPRESENTS THE POTENTIAL ENERGY CONTENT OF THE PRODUCTS AFTER THE REACTION.

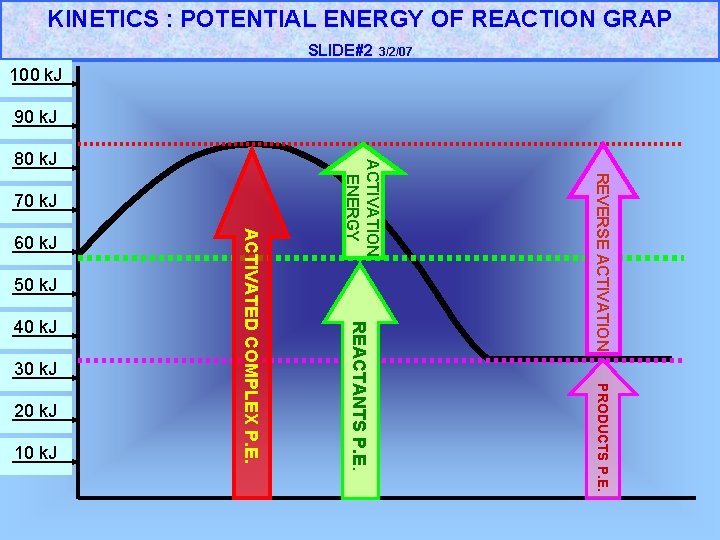
KINETICS : POTENTIAL ENERGY OF REACTION GRAP SLIDE#2 3/2/07 100 k. J 90 k. J 50 k. J 30 k. J 10 k. J PRODUCTS P. E. 20 k. J REACTANTS P. E. 40 k. J ACTIVATED COMPLEX P. E. 60 k. J REVERSE ACTIVATION 70 k. J ACTIVATION ENERGY 80 k. J

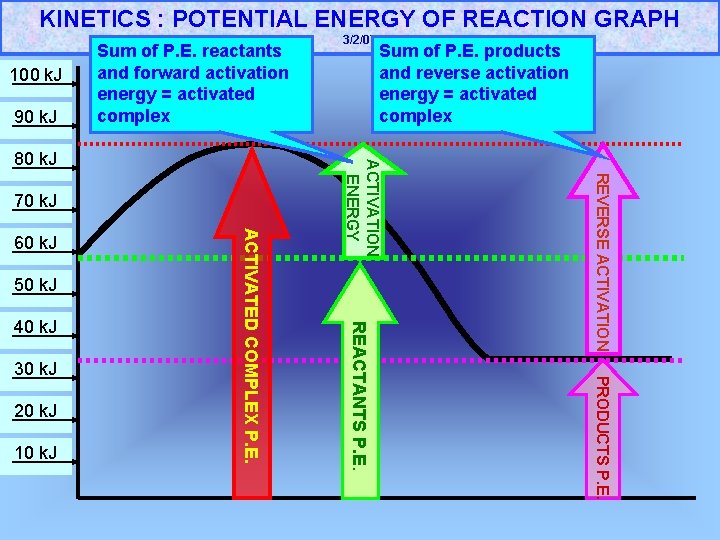
KINETICS : POTENTIAL ENERGY OF REACTION GRAPH 100 k. J 90 k. J Sum of P. E. reactants and forward activation energy = activated complex 50 k. J 20 k. J 10 k. J PRODUCTS P. E. 30 k. J REACTANTS P. E. 40 k. J ACTIVATED COMPLEX P. E. 60 k. J REVERSE ACTIVATION 70 k. J Sum of P. E. products and reverse activation energy = activated complex ACTIVATION ENERGY 80 k. J 3/2/07

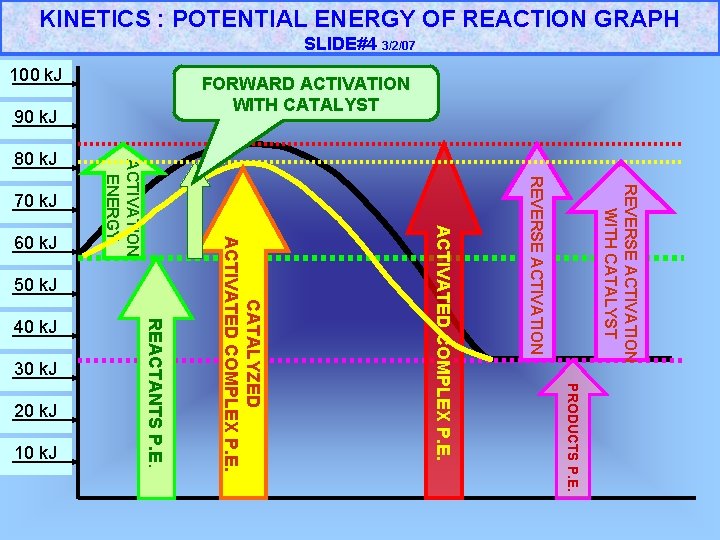
KINETICS : POTENTIAL ENERGY OF REACTION GRAPH SLIDE#4 3/2/07 90 k. J REVERSE ACTIVATION WITH CATALYST 50 k. J 60 k. J 40 k. J 30 k. J PRODUCTS P. E. 20 k. J 10 k. J REACTANTS P. E. CATALYZED ACTIVATED COMPLEX P. E. REVERSE ACTIVATION 70 k. J ACTIVATION ENERGY 80 k. J FORWARD ACTIVATION WITH CATALYST 100 k. J

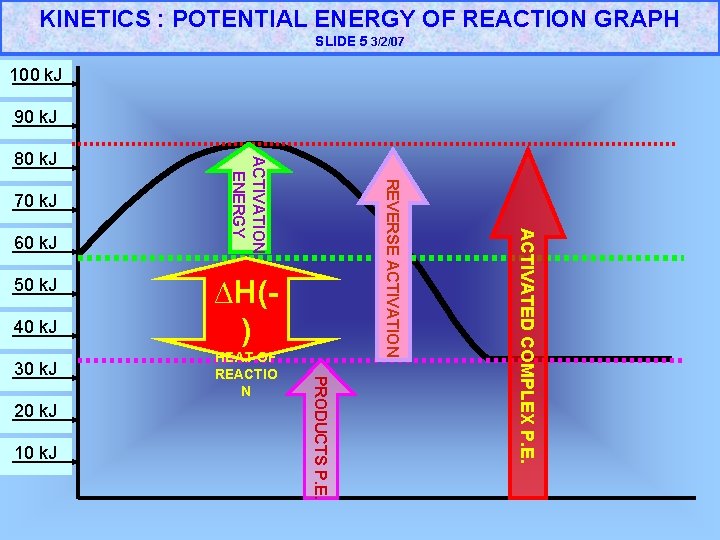
KINETICS : POTENTIAL ENERGY OF REACTION GRAPH SLIDE 5 3/2/07 100 k. J 90 k. J 50 k. J 40 k. J 20 k. J 10 k. J HEAT OF REACTIO N PRODUCTS P. E. 30 k. J ∆H() ACTIVATED COMPLEX P. E. 60 k. J REVERSE ACTIVATION 70 k. J ACTIVATION ENERGY 80 k. J

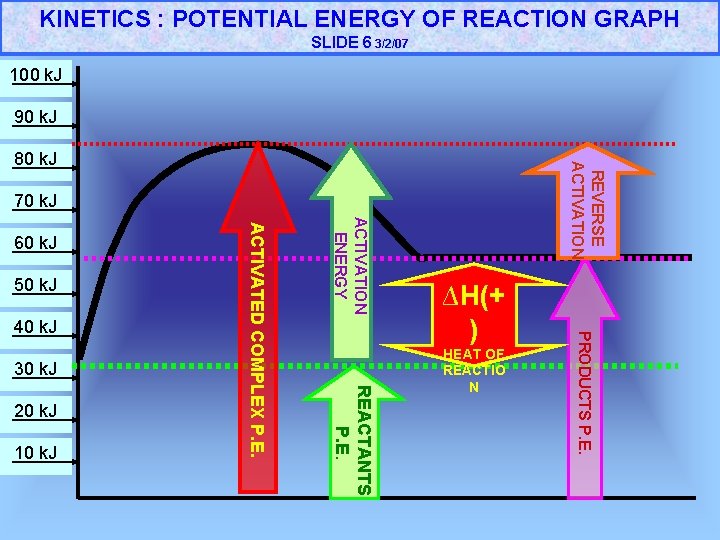
KINETICS : POTENTIAL ENERGY OF REACTION GRAPH SLIDE 6 3/2/07 100 k. J 90 k. J REVERSE ACTIVATION 80 k. J 70 k. J 30 k. J 10 k. J REACTANTS P. E. 20 k. J ∆H(+ ) HEAT OF REACTIO N PRODUCTS P. E. 40 k. J ACTIVATION ENERGY 50 k. J ACTIVATED COMPLEX P. E. 60 k. J

U DO IT NOW – PRACTICE IN GROUPS 1) What is the value of ∆H (k. J) in the reaction shown in the slide #1, show draw the curve and show arrows. 2) What is the value of THE FORWARD AND REVERSE ACTIVATION ENERGY (k. J) IN SLIDE #1, show arrow addition equalities. 3) What is the value of THE ACTIVATED COMPLEX ENERGY LEVEL (k. J) IN SLIDE #1, show arrow addition equalities. 4) What is the value energy difference between the activation energy and catalyzed activation energy IN SLIDE #4.