Kinetics of EDTA degradation induced by dioxygen activation

Kinetics of EDTA degradation induced by dioxygen activation in the Fe(0)/air/water (ZEA) system Frank Cheng* Tina Noradoun University of Idaho Chemistry Department Moscow, ID 83843 -2343 ifcheng@uidaho. edu 208 -885 -6387 3/15/05 Cheng & Noradoun, University of Idaho

The search for a “green” oxidant n Problems with chlorine based bleaching methods. n Oxygen is the ultimate green oxidant. n Oxygen is kinetically stable 3/15/05 Cheng & Noradoun, University of Idaho 2
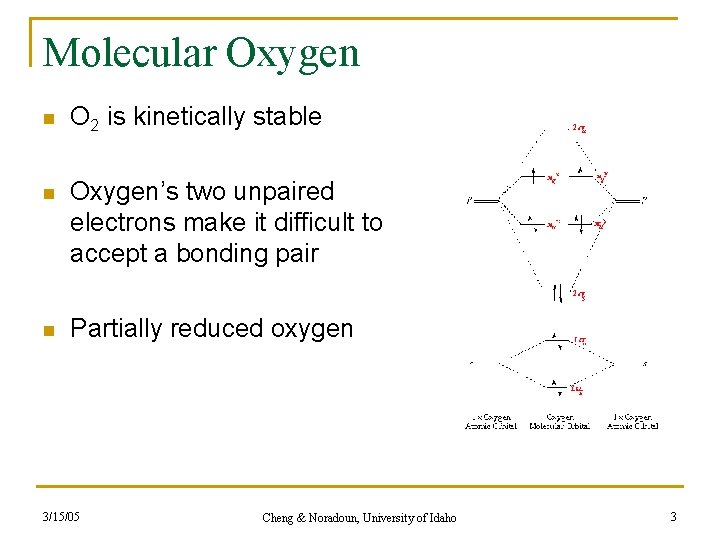
Molecular Oxygen n O 2 is kinetically stable n Oxygen’s two unpaired electrons make it difficult to accept a bonding pair n Partially reduced oxygen 3/15/05 Cheng & Noradoun, University of Idaho 3

Reactive Oxygen Forms • O=O • b. o. = 2 120 kcal/mol +e- • O-O • b. o. = 1. 5 80 kcal/mol G +4 e+4 H+ +2 e+2 H+ HO-OH b. o. = 1 50 kcal/mol 2 H 2 O 3/15/05 Cheng & Noradoun, University of Idaho 4

The Fenton Reaction H 2 O 2 + e- HO • + HOFe(II) Fe(III) + e. Fe(II) + H 2 O 2 Fe(III) + HO • + HO- H. J. H. Fenton. J. Chem. Soc. 1894, 65, 889. F. Haber and J. J. Weiss. Proc. Roy. Soc. London, Ser. A. 1934, 147, 332. 3/15/05 Cheng & Noradoun, University of Idaho 5
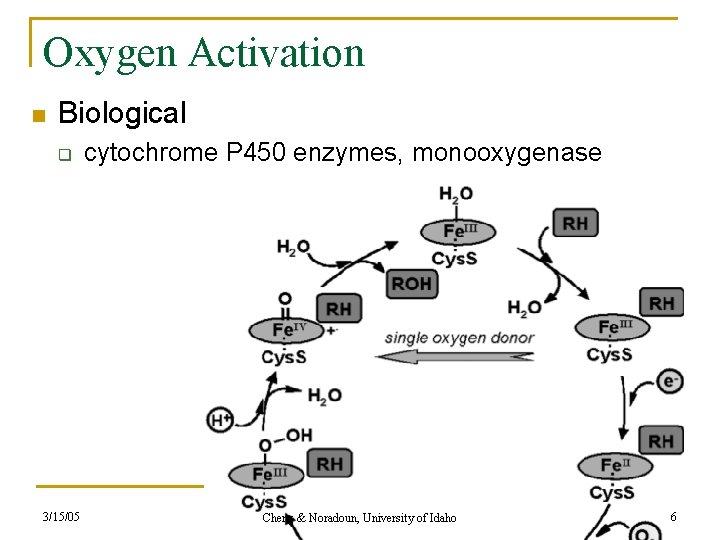
Oxygen Activation n Biological q 3/15/05 cytochrome P 450 enzymes, monooxygenase Cheng & Noradoun, University of Idaho 6

Molecular Oxygen as an Oxidant §Most attractive oxidant for green oxidations is O 2 from air. Diagram showing reaction oxygen intermediates between O 2 and H 2 O. H+ left out for simplicity 3/15/05 Cheng & Noradoun, University of Idaho 7
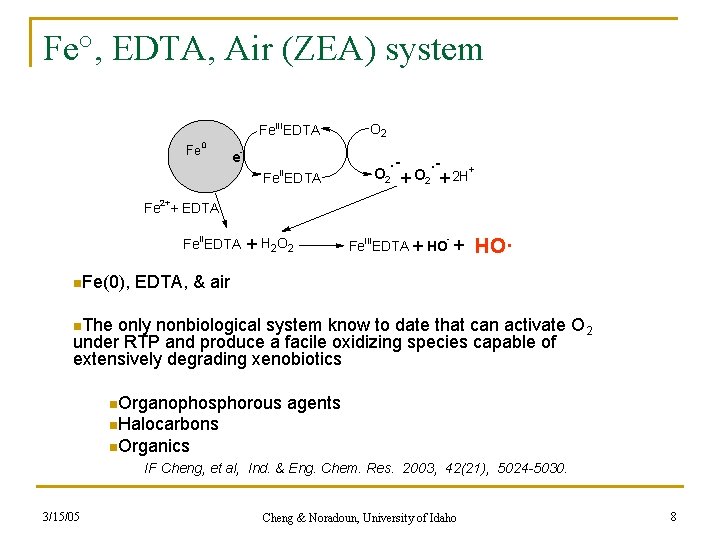
Fe°, EDTA, Air (ZEA) system Fe. IIIEDTA Fe 0 e- O 2 . - II Fe EDTA O 2 . - + O 2 + + 2 H Fe 2++ EDTA Fe. IIEDTA n. Fe(0), + H 2 O 2 - Fe. IIIEDTA + HO· EDTA, & air n. The only nonbiological system know to date that can activate O 2 under RTP and produce a facile oxidizing species capable of extensively degrading xenobiotics n. Organophosphorous agents n. Halocarbons n. Organics IF Cheng, et al, Ind. & Eng. Chem. Res. 2003, 42(21), 5024 -5030. 3/15/05 Cheng & Noradoun, University of Idaho 8

Outline n Introduction q Environmental Impact of EDTA q n General Reaction Scheme q n 3/15/05 EDTA q Chlorinated phenols q Organophosphorus and UXO compounds Mechanisms q n Zero-valent iron/EDTA/air (ZEA) system Degradation Kinetics and Reaction Products q n The RTP Dioxygen Activation Rate-limiting Step Conclusions Cheng & Noradoun, University of Idaho 9

EDTA • Finds widespread use in food, cosmetic and pharmaceutical preparations • Newer uses – manufacture of textiles and in paper-pulp bleaching q. Swedish pulping industry where the use of EDTA has increased from 2, 000 metric tons to about 8, 000 metric tons per year from 1990 to 2000. Ekland, B; Bruno, E; Lithner, G. ; Hans, B. ; “Use of Etheylenediaminetetraacetic acid in Pulp Mills and Effects on Metal Mobility and Primary Products”; Environ. Toxicol. Chem. ; 2002; 21(5), 1040 -1051. 3/15/05 Cheng & Noradoun, University of Idaho 10

EDTA n Control of metal ion activities, Fe, Mn, Zn, Cu, Mg, Ca -Corrosion -Catalysis -“Green” bleaching - H 2 O 2 q 3/15/05 Sillanpaa, M; Pirkanniemi, K. ; Sorokin, A. ; “Degradative hydrogen peroxide oxidation of chelates catalysed by metallophthalocyanines”; The Science of the Total Environment; 2003; 307, 11 -18. Cheng & Noradoun, University of Idaho 11

Concerns about EDTA q q q Many industrial chelating agents are not degradable by methods currently found in wastewater treatment facilities Not readily biodegradable Considerable quantities of EDTA pass through wastewater treatment facilities in the form of Fe. IIIEDTA, as high as 18µM. Sillanpaa, Mika; Orma, Marjatt; Ramo, Jaakko; Oikair; “The importance of ligand speciation in environmental research: a case study”; The Science of the Total Environment; 2001; 267, 2331. Sillanpaa, M; Pirkanniemi, K. ; “Recent Developments in Chelate Degradation”; Environmental Technology, 2001, 22, 791. Kari, F. G. ; Giger W; Modeling the Photochemical Degradation of Ethylenediaminetetraacetate in the River Glatt; Environmental Science and Technology; 1995, 29, 2814. Nirel, P. et. al. ; Method for EDTA Speciation Deteremination: Application to Sewage Treatment Plant Effluents; Wat. Res; 1998; 32, 3615. Kari, F. G. and Giger W. ; Speciation and fate of EDTA in municipal wasterwater treatment. Wat. Res. , 1996, 30, 122 -134. 3/15/05 Cheng & Noradoun, University of Idaho 12

Concerns about EDTA n Questions regarding the ability to mobilize metals in the environment. q q q n Currently not being monitored or treated at waste water treatment facilities Concern for heavy metal mobility and longer bioavailability of metals to aquatic plants and animals Stable in aquatic environment EDTA is anthropogenic and long-lived. 3/15/05 Cheng & Noradoun, University of Idaho 13

Goals • The destruction or neutralization of EDTA (xenobiotics) • Search for in situ conditions that will aid in the reduction in the release of EDTA in emerging green chemistries. Inexpensive & Safe Processes. • Room Temperature and Pressure Conditions (RTP) • Common Reagents – Long Term Storage • No Specialized Catalysts • System that may be incorporated into existing water treatment systems 3/15/05 Cheng & Noradoun, University of Idaho 14

Overall Goals of Our Green Oxidation Program § The destruction or neutralization of xenobiotics, including nerve agents and chlorinated pesticides using green oxidation chemistry. § Focus on non-biological oxygen activation to eliminate the need for tricky enzyme based systems 3/15/05 Cheng & Noradoun, University of Idaho 15

Oxygen activation system n The ZEA system uses only zero-valent iron, EDTA and air 3/15/05 n The only nonbiological system know to date that can activate O 2 under RTP and produce a facile oxidizing species capable of extensively degrading xenobiotics n Industrial and Engineering Chemistry Research 2003, 42, 5024 -5030 Cheng & Noradoun, University of Idaho 16
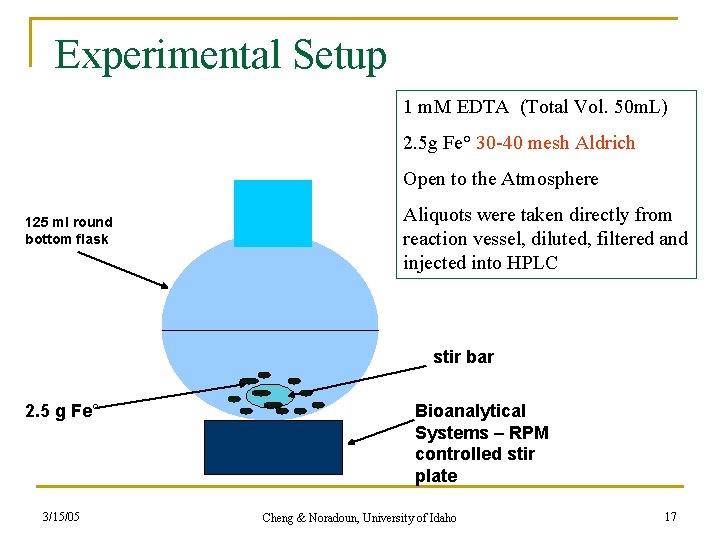
Experimental Setup 1 m. M EDTA (Total Vol. 50 m. L) 2. 5 g Fe° 30 -40 mesh Aldrich Open to the Atmosphere 125 ml round bottom flask Aliquots were taken directly from reaction vessel, diluted, filtered and injected into HPLC stir bar 2. 5 g Fe° 3/15/05 Bioanalytical Systems – RPM controlled stir plate Cheng & Noradoun, University of Idaho 17
Evidence for Production of Reactive Oxygen Species n ROS q n O 2 - • , H 2 O 2, HO. , Fe. IV=O, etc. Two Analyses were performed q q 3/15/05 Thiobarbituric acid-reactive substances (TBARS) assay Addition of known radical scavenger, 1 -butanol Cheng & Noradoun, University of Idaho 18
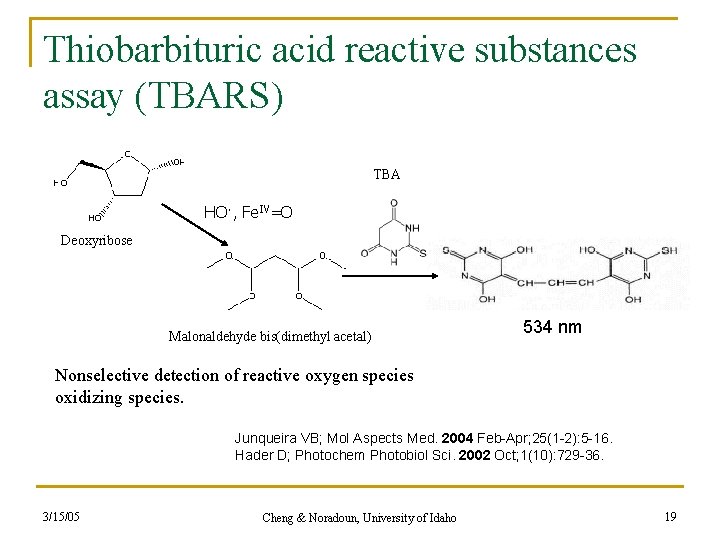
Thiobarbituric acid reactive substances assay (TBARS) TBA HO·, Fe. IV=O Deoxyribose Malonaldehyde bis(dimethyl acetal) 534 nm Nonselective detection of reactive oxygen species oxidizing species. Junqueira VB; Mol Aspects Med. 2004 Feb-Apr; 25(1 -2): 5 -16. Hader D; Photochem Photobiol Sci. 2002 Oct; 1(10): 729 -36. 3/15/05 Cheng & Noradoun, University of Idaho 19
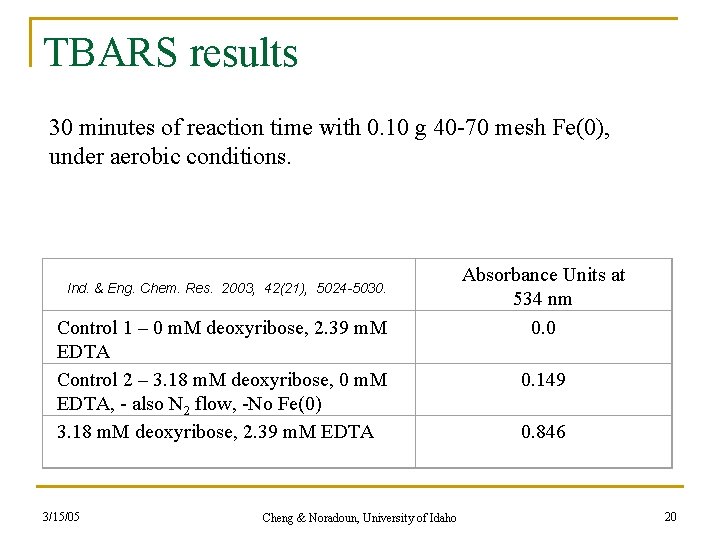
TBARS results 30 minutes of reaction time with 0. 10 g 40 -70 mesh Fe(0), under aerobic conditions. Ind. & Eng. Chem. Res. 2003, 42(21), 5024 -5030. Control 1 – 0 m. M deoxyribose, 2. 39 m. M EDTA Control 2 – 3. 18 m. M deoxyribose, 0 m. M EDTA, - also N 2 flow, -No Fe(0) 3. 18 m. M deoxyribose, 2. 39 m. M EDTA 3/15/05 Cheng & Noradoun, University of Idaho Absorbance Units at 534 nm 0. 0 0. 149 0. 846 20
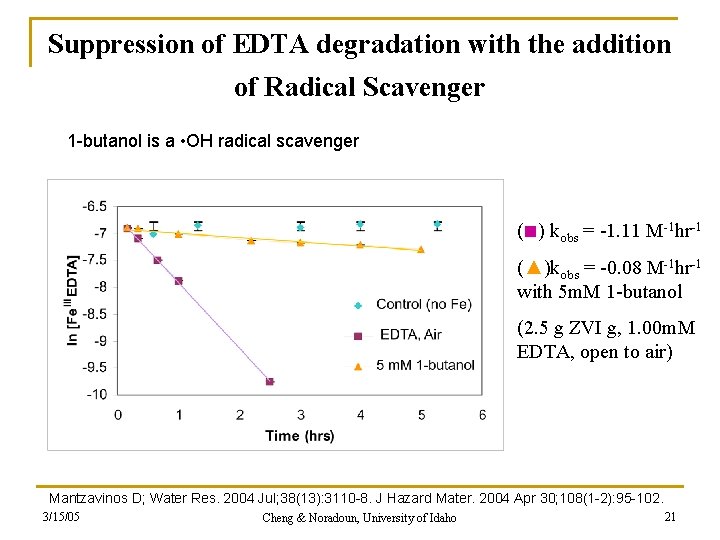
Suppression of EDTA degradation with the addition of Radical Scavenger 1 -butanol is a • OH radical scavenger (■) kobs = -1. 11 M-1 hr-1 (▲)kobs = -0. 08 M-1 hr-1 with 5 m. M 1 -butanol (2. 5 g ZVI g, 1. 00 m. M EDTA, open to air) Mantzavinos D; Water Res. 2004 Jul; 38(13): 3110 -8. J Hazard Mater. 2004 Apr 30; 108(1 -2): 95 -102. 3/15/05 Cheng & Noradoun, University of Idaho 21

Summary of ZEA system and O 2 n n n Both TBARS and butanol tests indicate that ZEA system is able to produce facile oxidant from air at RTP Form of abiotic O 2 activation at RTP Identity of oxidant isn’t clear q q 3/15/05 HO· Fe. IV=O Cheng & Noradoun, University of Idaho 22
Degradation of EDTA by ZEA reaction 1 m. M EDTA (50 m. L, aqueous) 2. 5 g Fe° + air 3/15/05 Cheng & Noradoun, University of Idaho 23

Products of ZEA System n None of the products of EDTA are significant metal chelation agents. n All are more easily biodegraded. n The ZEA system has proven successful at the degradation of other organic xenobiotics. n n n 3/15/05 Halocarbons Organophosphorus Organics Cheng & Noradoun, University of Idaho 24

Carbon Balance - Total Organic Carbon*, ESI-MS**, HPLC# Trapping of the volatile gases using Tenax® showed no volatile organic carbon of molecular weight C 4 and above released from the system during the course of the reaction 3/15/05 Cheng & Noradoun, University of Idaho 25
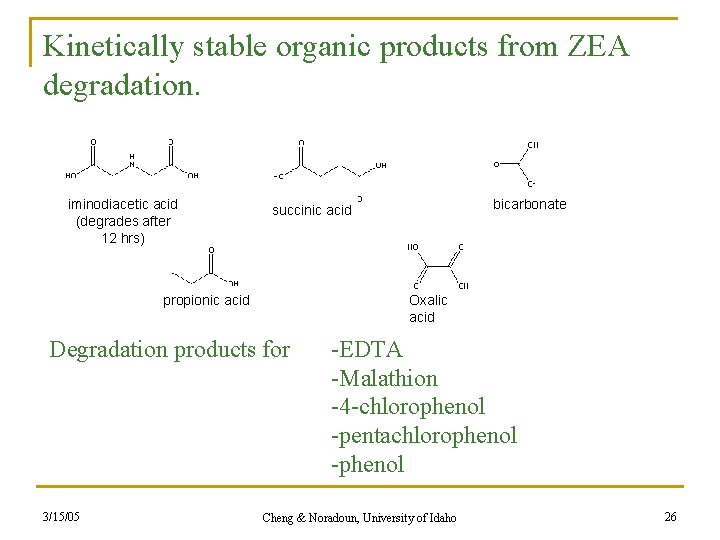
Kinetically stable organic products from ZEA degradation. iminodiacetic acid (degrades after 12 hrs) propionic acid Oxalic acid Degradation products for 3/15/05 bicarbonate succinic acid -EDTA -Malathion -4 -chlorophenol -pentachlorophenol -phenol Cheng & Noradoun, University of Idaho 26

Summary of EDTA degradation n EDTA is degraded by the ZEA system to LWM acids and inorganic carbons All products are more biodegradable than EDTA RTP O 2 activation n Next: Kinetic studies n n 3/15/05 Cheng & Noradoun, University of Idaho 27

Overall Scheme (simplified) Metal dissolution: n Fe(0) Fe 2+ + 2 e- (1) Complex formation n Fe 2+ + EDTA Fe. IIEDTA (2) Homogeneous O 2 activation: n 2 Fe. IIEDTA + O 2 + 2 H+ 2 Fe. IIIEDTA + H 2 O 2 (3) Fenton Reaction n Fe. IIEDTA + H 2 O 2 Fe. IIIEDTA + OH • + OH- (4) EDTA degradation: n OH • + Fe. EDTA Fe 2+/3+ + EDTA* (5) § EDTA* = damaged EDTA Redox Cycling: n Fe. IIIEDTA + e- Fe. IIEDTA 3/15/05 Cheng & Noradoun, University of Idaho (6) 28

Kinetic Parameters Examined Effects of § EDTA concentration § § Fe° mass (surface area) § § Omitted Temperature § 3/15/05 Omitted Rate of mixing § § Possible Scale-up Rate-limiting Step Cheng & Noradoun, University of Idaho 29
![Kinetics of EDTA degradation [EDTA]initial = 1 m. M kobs = 1. 22 /M Kinetics of EDTA degradation [EDTA]initial = 1 m. M kobs = 1. 22 /M](http://slidetodoc.com/presentation_image_h2/01d2525af297205fe60011b0e100177f/image-30.jpg)
Kinetics of EDTA degradation [EDTA]initial = 1 m. M kobs = 1. 22 /M hr 1 m. M EDTA, 2. 5 g Fe° and air (▲), control in the absence of iron (■) 3/15/05 Pseudo-first order plot showing linearity for EDTA degradation from 10 min-2. 5 hrs. Cheng & Noradoun, University of Idaho 30
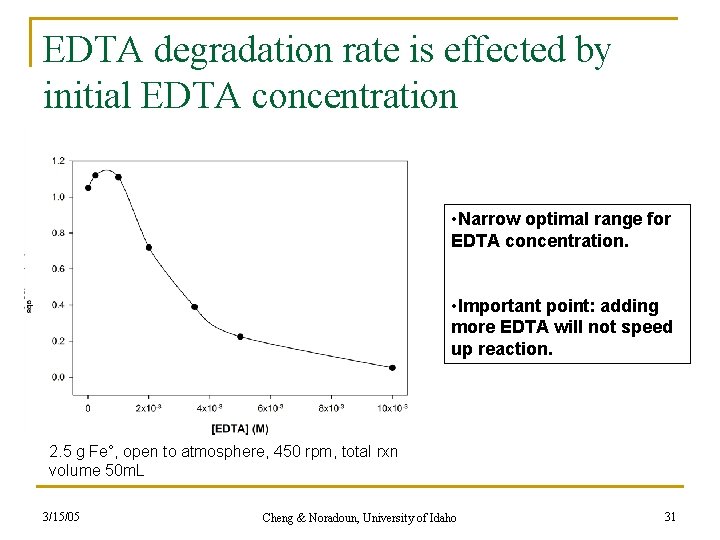
EDTA degradation rate is effected by initial EDTA concentration • Narrow optimal range for EDTA concentration. • Important point: adding more EDTA will not speed up reaction. 2. 5 g Fe°, open to atmosphere, 450 rpm, total rxn volume 50 m. L 3/15/05 Cheng & Noradoun, University of Idaho 31
![How does [EDTA]i effect the ZEA reaction rate? 3/15/05 n Corrosion Rate of Fe. How does [EDTA]i effect the ZEA reaction rate? 3/15/05 n Corrosion Rate of Fe.](http://slidetodoc.com/presentation_image_h2/01d2525af297205fe60011b0e100177f/image-32.jpg)
How does [EDTA]i effect the ZEA reaction rate? 3/15/05 n Corrosion Rate of Fe. IIIEDTA reduction by Fe(0). n Rate of Fenton Reaction n O 2 activation Cheng & Noradoun, University of Idaho 32

Corrosion studies n Electrochemical Studies - Tafel Analysis n i 0 is the exchange current which is the rate of the corrosion. 3/15/05 Cheng & Noradoun, University of Idaho 33
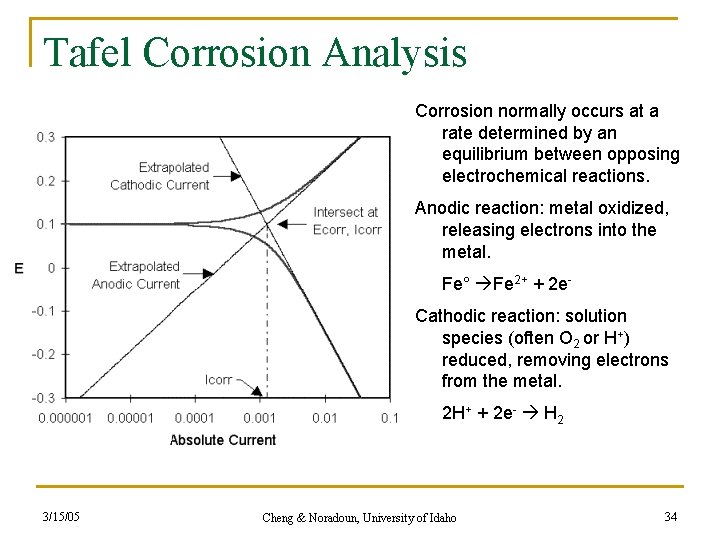
Tafel Corrosion Analysis Corrosion normally occurs at a rate determined by an equilibrium between opposing electrochemical reactions. Anodic reaction: metal oxidized, releasing electrons into the metal. Fe° Fe 2+ + 2 e. Cathodic reaction: solution species (often O 2 or H+) reduced, removing electrons from the metal. 2 H+ + 2 e- H 2 3/15/05 Cheng & Noradoun, University of Idaho 34

Corrosion Cell • Working Electrode: Fe° (99%), 3/8" diameter by 1/2" length (surface area 5. 22 x 10 -4 m 2) • Counter Electrode: high density graphite rod • Reference: Standard Calomel Electrode (SCE), glass luggin capillary • 1 liter glass cell • Polished working electrode with 600 grit sandpaper between sample runs • Used 50 m. M KNO 3 as electrolyte in all samples 3/15/05 Cheng & Noradoun, University of Idaho 35
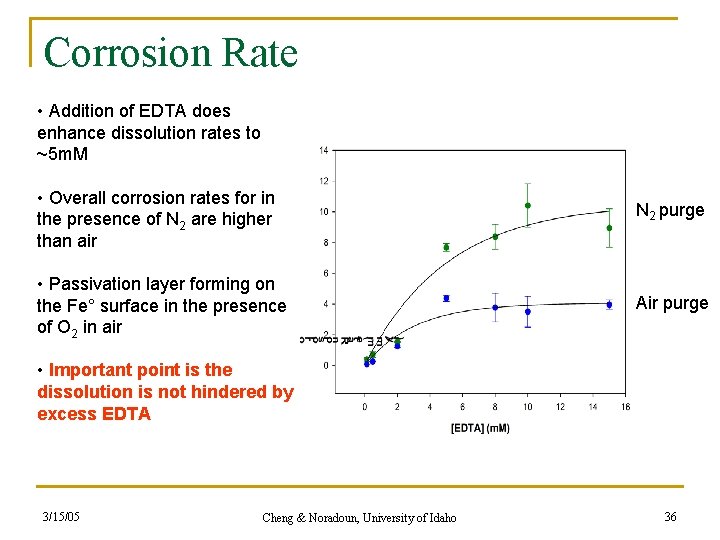
Corrosion Rate • Addition of EDTA does enhance dissolution rates to ~5 m. M • Overall corrosion rates for in the presence of N 2 are higher than air N 2 purge • Passivation layer forming on the Fe° surface in the presence of O 2 in air Air purge • Important point is the dissolution is not hindered by excess EDTA 3/15/05 Cheng & Noradoun, University of Idaho 36
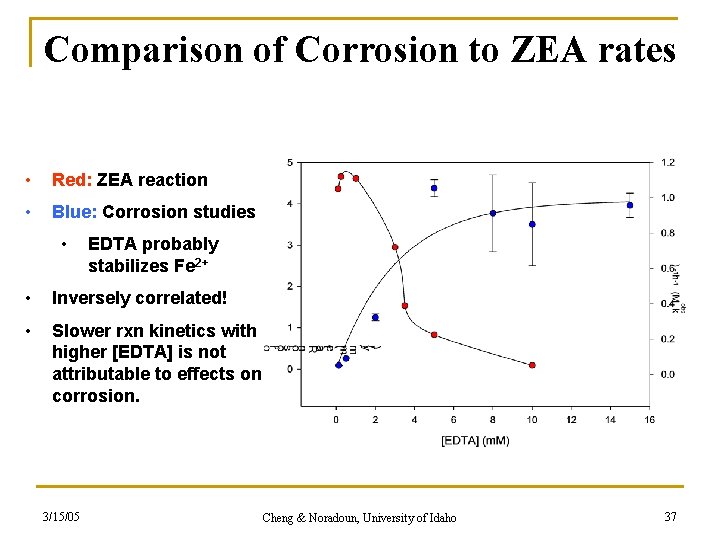
Comparison of Corrosion to ZEA rates • Red: ZEA reaction • Blue: Corrosion studies • EDTA probably stabilizes Fe 2+ • Inversely correlated! • Slower rxn kinetics with higher [EDTA] is not attributable to effects on corrosion. 3/15/05 Cheng & Noradoun, University of Idaho 37
![Other Possibilities for effects of [EDTA] on ZEA reaction rates n Kinetic barriers q Other Possibilities for effects of [EDTA] on ZEA reaction rates n Kinetic barriers q](http://slidetodoc.com/presentation_image_h2/01d2525af297205fe60011b0e100177f/image-38.jpg)
Other Possibilities for effects of [EDTA] on ZEA reaction rates n Kinetic barriers q Electrochemical q q Fenton Rxn q q Fe. IIEDTA + H 2 O 2 Fe. IIIEDTA +HO- + HO· Oxygen Reduction/Activation q q 3/15/05 Fe. IIIEDTA + e- = Fe. IIEDTA + O 2 Fe. IIIEDTA + O 2. Other routes Cheng & Noradoun, University of Idaho 38
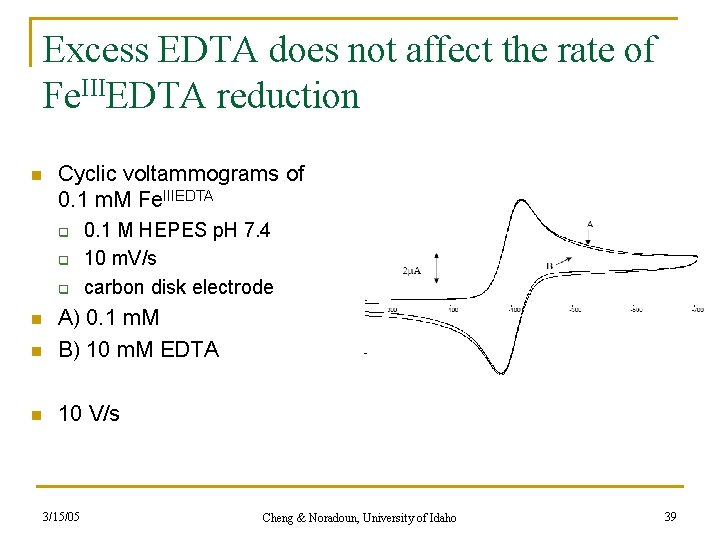
Excess EDTA does not affect the rate of Fe. IIIEDTA reduction n Cyclic voltammograms of 0. 1 m. M Fe. IIIEDTA q q q 0. 1 M HEPES p. H 7. 4 10 m. V/s carbon disk electrode n A) 0. 1 m. M B) 10 m. M EDTA n 10 V/s n 3/15/05 Cheng & Noradoun, University of Idaho 39

Other Possibilities n Kinetic barriers q Electrochemical q q Fenton Rxn q q Fe. IIEDTA + H 2 O 2 Fe. IIIEDTA +HO- + HO· Oxygen Reduction q q 3/15/05 Fe. IIIEDTA + e- = Fe. IIEDTA + O 2 Fe. IIIEDTA + O 2. Other routes Cheng & Noradoun, University of Idaho 40
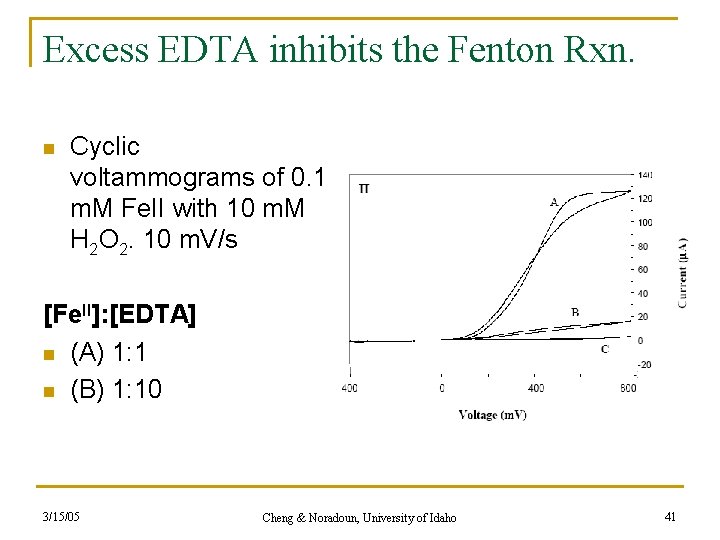
Excess EDTA inhibits the Fenton Rxn. n Cyclic voltammograms of 0. 1 m. M Fe. II with 10 m. M H 2 O 2. 10 m. V/s [Fe. II]: [EDTA] n (A) 1: 1 n (B) 1: 10 3/15/05 Cheng & Noradoun, University of Idaho 41
![[Fe. III]: [EDTA] Fenton Rxn trends §EDTA may act as an anti- or pro-oxidant. [Fe. III]: [EDTA] Fenton Rxn trends §EDTA may act as an anti- or pro-oxidant.](http://slidetodoc.com/presentation_image_h2/01d2525af297205fe60011b0e100177f/image-42.jpg)
[Fe. III]: [EDTA] Fenton Rxn trends §EDTA may act as an anti- or pro-oxidant. §Highly dependent on [EDTA]i 3/15/05 Cheng & Noradoun, University of Idaho 42

Other Possibilities n Kinetic barriers q Electrochemical q ü q Fenton Rxn Fe. IIEDTA + H 2 O 2 Fe. IIIEDTA +HO- + HO· HO Oxygen Reduction (future work) q q 3/15/05 Fe. IIIEDTA + e- = Fe. IIEDTA + O 2 Fe. IIIEDTA + O 2. Other routes Cheng & Noradoun, University of Idaho 43

Temperature Experiments n An Arrhenius plot - activation energy n May allow us to determine rate-limiting step q q q 3/15/05 Metal Dissolution Complex Formation Homogeneous O 2 Activation Fenton Rxn EDTA oxidation Heterogeneous Redox Cycling Cheng & Noradoun, University of Idaho 44
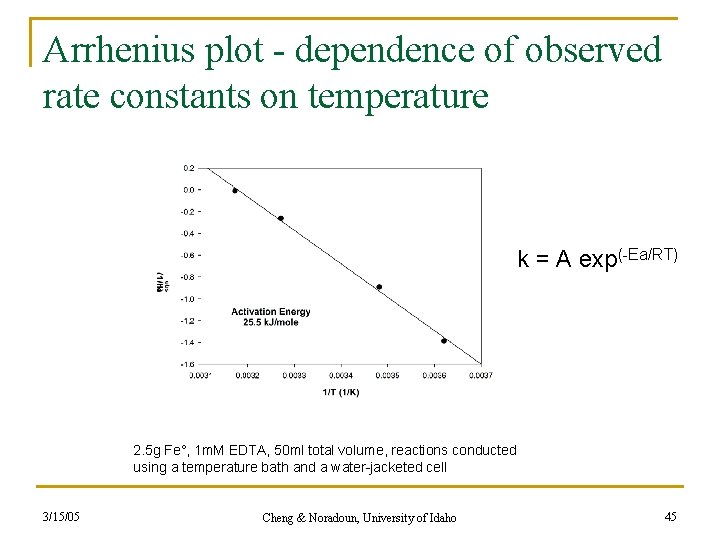
Arrhenius plot - dependence of observed rate constants on temperature k = A exp(-Ea/RT) 2. 5 g Fe°, 1 m. M EDTA, 50 ml total volume, reactions conducted using a temperature bath and a water-jacketed cell 3/15/05 Cheng & Noradoun, University of Idaho 45

Summary of Arrhenius Studies • This study: Ea = 25. 5 k. J/mol • Under vigorous stirring; Mass transport limited region • This activation energy includes all steps. • How may this determine which of the 6 processed is rate-limiting? • Comparison with literature regarding: • O 2 reduction by Fe. IIEDTA 3/15/05 Cheng & Noradoun, University of Idaho 46

Overall Scheme (simplified) Metal dissolution: n Fe(0) Fe 2+ + 2 e- (1) Complex formation n Fe 2+ + EDTA Fe. IIEDTA (2) Homogeneous O 2 activation: n 2 Fe. IIEDTA + O 2 + 2 H+ 2 Fe. IIIEDTA + H 2 O 2 (3) Fenton Reaction n Fe. IIEDTA + H 2 O 2 Fe. IIIEDTA + OH • + OH- (4) EDTA degradation: n OH • + Fe. EDTA Fe 2+/3+ + EDTA* (5) § EDTA* = damaged EDTA Redox Cycling: n Fe. IIIEDTA + e- Fe. IIEDTA 3/15/05 Cheng & Noradoun, University of Idaho (6) 47

Comparison to kinetics of homogeneous O 2 reduction by Fe. IIEDTA in literature R. Van Eldik; Inorg. Chem. ; 1990, 29, 1705 -1711. [Fe. IIEDTA] = 0. 02 M p. H=5 Fe. IIEDTA + O 2 Fe. IIEDTAO 2 Fe. IIIEDTA + O 2 Fe. IIEDTAO 2 + H+ Fe. IIIEDTA + HO 2 q q k 1 = 107/Ms k 2 = 102/Ms k 3 = 1010/Ms Rate limiting step is the activation of oxygen at the iron coordination site Activation energy, 33. 9 k. J/mol* 3/15/05 Cheng & Noradoun, University of Idaho 48

Summary of Van Eldik Study Fe. IIEDTAH(H 2 O) + O 2 Fe. IIEDTAH(O 2) + H 2 O Fe. IIEDTAH(O 2) Fe. IIIEDTAH(O 2 -) + Fe. IIEDTAH(H 2 O) Fe. IIIEDTAH(O 22 -)Fe. IIIEDTAH + H 2 O + 2 H+ 2 Fe. IIIEDTAH(H 2 O) + H 2 O 2 2 Fe. IIEDTAH(H 2 O) + H 2 O 2 2 Fe. IIIEDTAH(H 2 O) + H 2 O *Proposes H 2 O 2 as intermediate *Saw no evidence of H 2 O 2 or Fenton Rxn Van Eldik, R. Inorg. Chem, 1997, 36, 4115 -4120 3/15/05 Cheng & Noradoun, University of Idaho 49

Comparison to kinetics of homogeneous O 2 reduction by Fe. IIEDTA in literature Beenackers, A. ; Ing. Eng. Chem. Res. 1992, 32, 2580 [Fe. IIEDTA]=0. 10 M q q p. H=7. 5 2 Fe. IIEDTA + O 2 + H+ 2 Fe. IIIEDTA + H 2 O 2 (overall) Activation energy, 27. 2 k. J/mol 3/15/05 Cheng & Noradoun, University of Idaho 50
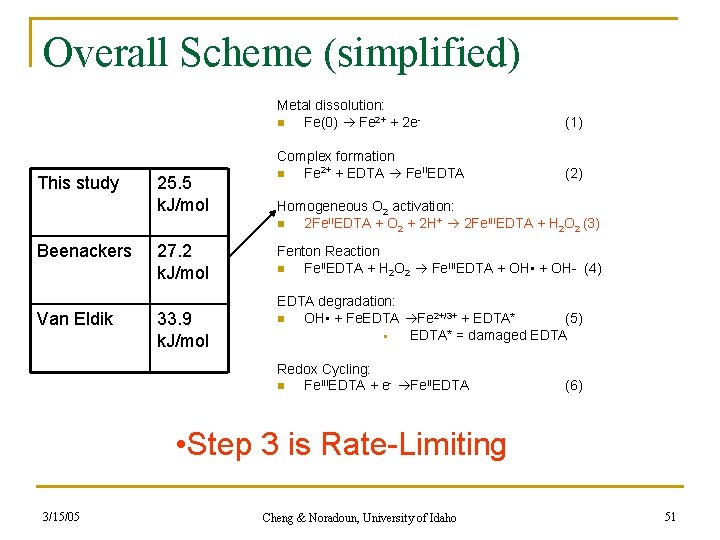
Overall Scheme (simplified) This study Beenackers Van Eldik 25. 5 k. J/mol 27. 2 k. J/mol 33. 9 k. J/mol Metal dissolution: n Fe(0) Fe 2+ + 2 e- (1) Complex formation n Fe 2+ + EDTA Fe. IIEDTA (2) Homogeneous O 2 activation: n 2 Fe. IIEDTA + O 2 + 2 H+ 2 Fe. IIIEDTA + H 2 O 2 (3) Fenton Reaction n Fe. IIEDTA + H 2 O 2 Fe. IIIEDTA + OH • + OH- (4) EDTA degradation: n OH • + Fe. EDTA Fe 2+/3+ + EDTA* (5) § EDTA* = damaged EDTA Redox Cycling: n Fe. IIIEDTA + e- Fe. IIEDTA (6) • Step 3 is Rate-Limiting 3/15/05 Cheng & Noradoun, University of Idaho 51

Conclusions n This system is a viable option for the destruction of a variety of pollutants and has a strong possibility for scale up. n The only system known to date that can obtain non-biological Oxygen Activation at room temperature and pressure to produce reactive oxygen species that are capable of fully degrading pollutants to LMW carboxylates and inorganic forms n Due to the duality of EDTA acting as both a pro-oxidant and antioxidant, controlling the [EDTA] is imperative to the success of the process. n Rate-limiting steps is (are) oxygen activation n Fe. IIEDTA may provide insights into biological oxygen activation n This study may provide insights as to how EDTA release can be controlled q Future studies – how many oxidative hits & redox cycles required to degrade EDTA to inorganic forms n Search for more suitable reducing agents 3/15/05 Cheng & Noradoun, University of Idaho 52

Acknowledgments Thank You! Christina Noradoun Funding n NSF BES-0328827 n UI Foundation Seed Grant q q n NIH EPRI Dr. Malcolm and Mrs. Renfrew q 3/15/05 Renfrew Scholarships Cheng & Noradoun, University of Idaho 53
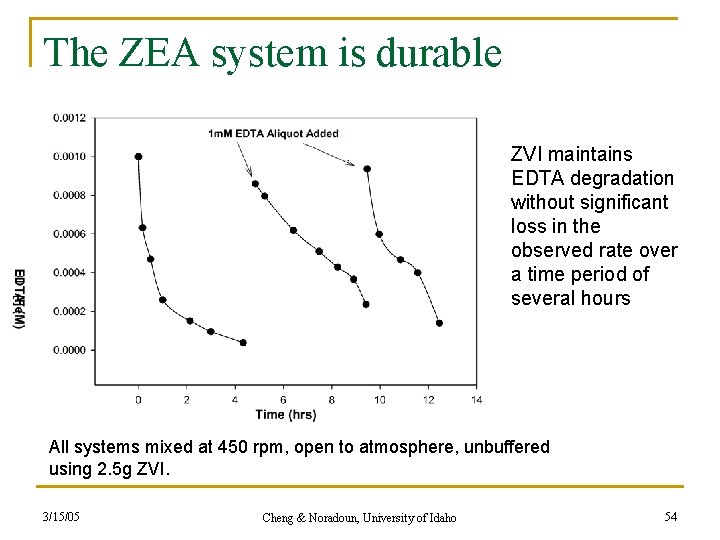
The ZEA system is durable ZVI maintains EDTA degradation without significant loss in the observed rate over a time period of several hours All systems mixed at 450 rpm, open to atmosphere, unbuffered using 2. 5 g ZVI. 3/15/05 Cheng & Noradoun, University of Idaho 54

Comparison Studies n Auto-oxidation of Fe. II to Fe. III by O 2 in aqueous solutions q q Significantly enhanced by EDTA Fe. II: EDTA ratios were important n n 1: 1 ratios were reported as optimal 1: 20 ratios showed a significant decrease in the autoxidation process R. Van Eldik; Inorg. Chem. ; 1990, 29, 1705 -1711. (* 0. 02 M [Fe(EDTA)]) 3/15/05 Cheng & Noradoun, University of Idaho 55
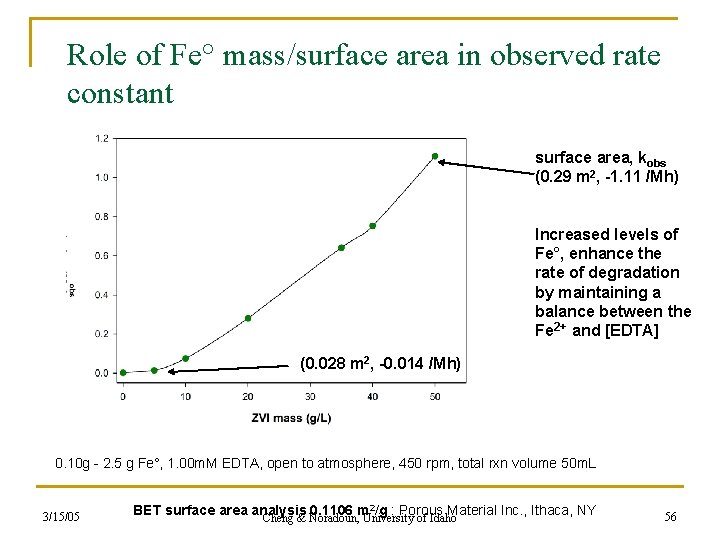
Role of Fe° mass/surface area in observed rate constant surface area, kobs (0. 29 m 2, -1. 11 /Mh) Increased levels of Fe°, enhance the rate of degradation by maintaining a balance between the Fe 2+ and [EDTA] (0. 028 m 2, -0. 014 /Mh) 0. 10 g - 2. 5 g Fe°, 1. 00 m. M EDTA, open to atmosphere, 450 rpm, total rxn volume 50 m. L 3/15/05 BET surface area analysis 0. 1106 m 2/g : Porous Material Inc. , Ithaca, NY Cheng & Noradoun, University of Idaho 56

Maintaining proper Fe°to EDTA ratios n n Interactions between EDTA and Fe 2+ are important factor controlling the degradation rates Due to the duality of EDTA acting as both a pro-oxidant and antioxidant, controlling the [EDTA] is imperative to the success of the process. Rate-limiting step n 1. Surface chemistry : Reduction of Fe. II/III at the iron surface inhibited by excess EDTA n 2. Solution chemistry: High Fe. II/III: EDTA ratios inhibiting Fenton reactivity. 3/15/05 Cheng & Noradoun, University of Idaho 57

General Model Mass Transport-limited Kinetics 1) mass transport of Fe. IIIEDTA to the Fe° surface 2) Fe. IIIEDTA + e- Fe. IIEDTA 3) mass transport of Fe. IIEDTA to the bulk soln. “A common criterion for detecting mass transport-limited kinetics is variation in reaction rate with intensity of mixing. Rates that are controlled by chemical reaction step should not be affected, where as aggressive mixing usually accelerates diffusioncontrolled rates by reducing the thickness of the diffusion layer. ” ES&T. 1994, 28 2045 -2053. 3/15/05 Leah Matheson and Paul Tratnyek; Cheng & Noradoun, University of Idaho 58
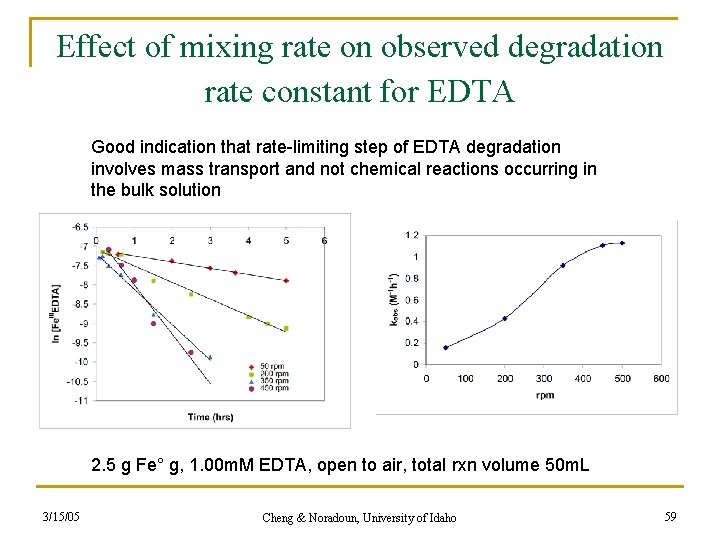
Effect of mixing rate on observed degradation rate constant for EDTA Good indication that rate-limiting step of EDTA degradation involves mass transport and not chemical reactions occurring in the bulk solution 2. 5 g Fe° g, 1. 00 m. M EDTA, open to air, total rxn volume 50 m. L 3/15/05 Cheng & Noradoun, University of Idaho 59
n If reaction is mass transport controlled rate limiting step likely: q n n n Fe. II/IIIEDTA reduction at iron surface Can not rule out the heterogeneous O 2 activation Mass transport of oxygen from the bulk solution to the reacting iron surface is enhanced by the fluid flow. Typical bulk oxygen concentrations at room temperature in aqueous solutions are 0. 25 m. M (8 ppm). 3/15/05 Cheng & Noradoun, University of Idaho 60

HPLC conditions for Fe. IIIEDTA detection EDTA non-extractable using organic solvent must use direct aqueous injection EDTA alone not absorb, however Fe. IIIEDTA complex does at 258 nm n n n n Mobile phase: 0. 02 M formate buffer, p. H 3. 3 Containing: TBA-Br (0. 001 M) and acetonitrile (8%) Flow rate: 1 ml/min Temp: ambient temp UV = 258 nm + Sample volume 20µL Column RP-C 18 TBA-Br 3/15/05 Nowack et. al. ; Anal. Chem. 1996, 68, 561 Cheng & Noradoun, University of Idaho 61

Organophosphorous Nerve Agents and Nitrated Explosive Surrogates TNT surrogate, nitrobenzene (985 ppm) was decomposed in 24 hours. VX surrogate, malathion (49 ppm) was consumed in 4 hours, to give diethyl succinate. Malathion was the only pollutant to give a byproduct detectable by GC-FID. 3/15/05 Cheng & Noradoun, University of Idaho 62
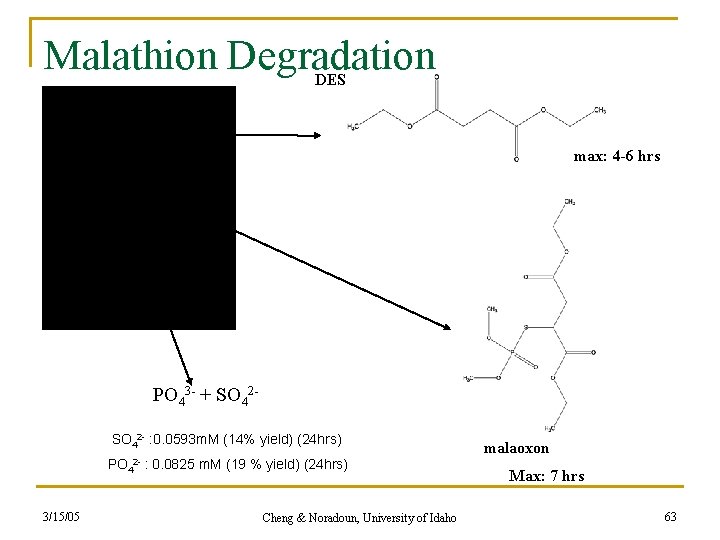
Malathion Degradation DES malathion max: 4 -6 hrs PO 43 - + SO 42 - : 0. 0593 m. M (14% yield) (24 hrs) PO 42 - 3/15/05 : 0. 0825 m. M (19 % yield) (24 hrs) Cheng & Noradoun, University of Idaho malaoxon Max: 7 hrs 63
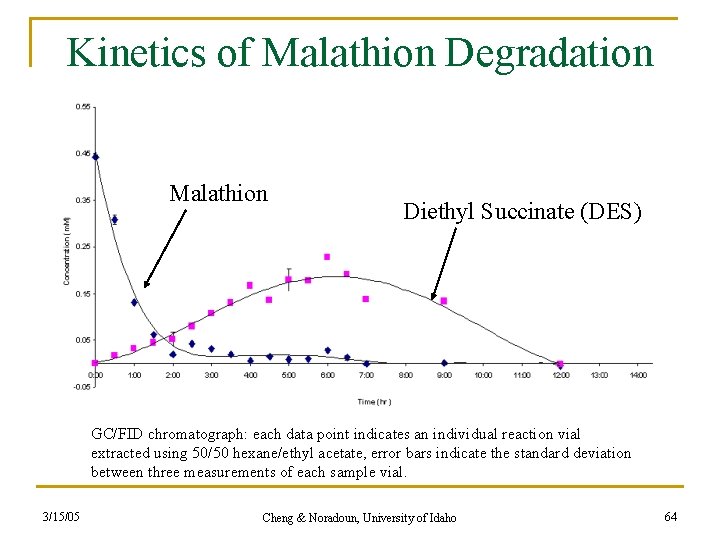
Kinetics of Malathion Degradation Malathion Diethyl Succinate (DES) GC/FID chromatograph: each data point indicates an individual reaction vial extracted using 50/50 hexane/ethyl acetate, error bars indicate the standard deviation between three measurements of each sample vial. 3/15/05 Cheng & Noradoun, University of Idaho 64
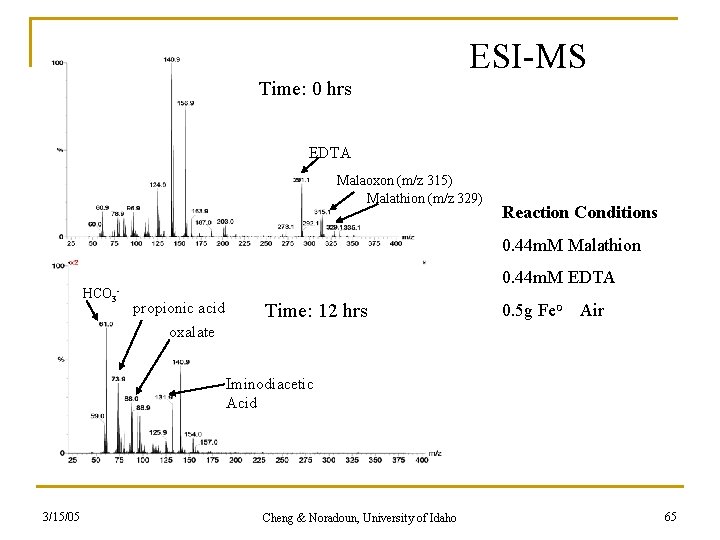
ESI-MS Time: 0 hrs EDTA Malaoxon (m/z 315) Malathion (m/z 329) Reaction Conditions 0. 44 m. M Malathion HCO 3 0. 44 m. M EDTA - propionic acid oxalate Time: 12 hrs 0. 5 g Fe. O Air Iminodiacetic Acid 3/15/05 Cheng & Noradoun, University of Idaho 65
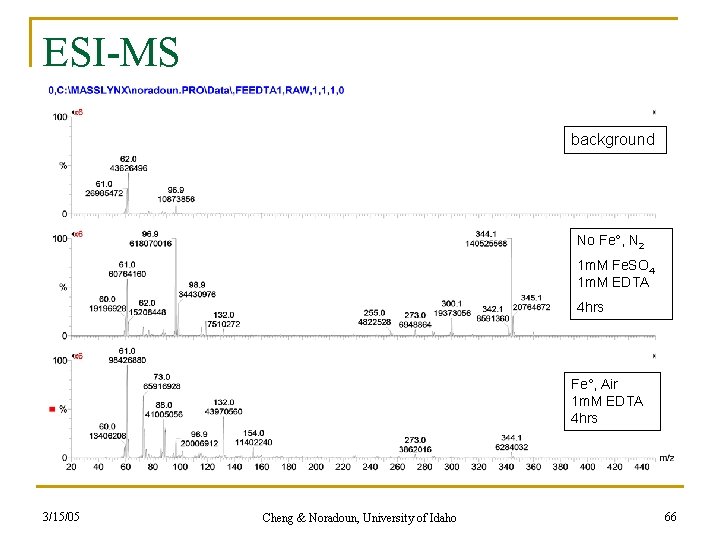
ESI-MS background No Fe°, N 2 1 m. M Fe. SO 4 1 m. M EDTA 4 hrs Fe°, Air 1 m. M EDTA 4 hrs 3/15/05 Cheng & Noradoun, University of Idaho 66
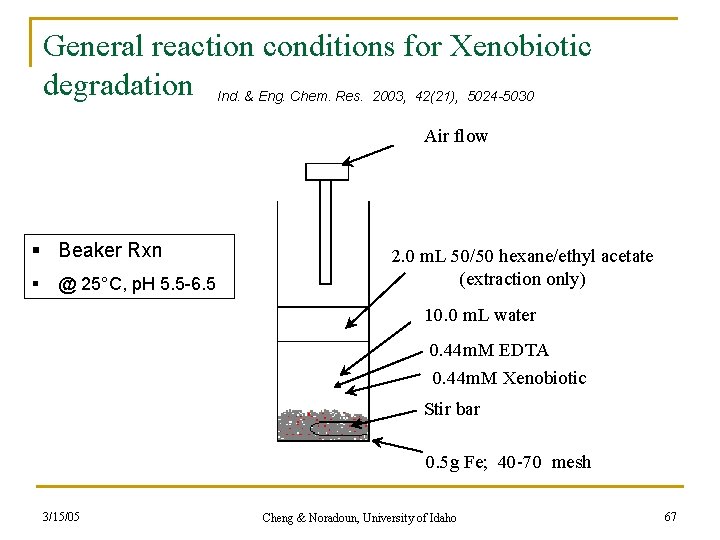
General reaction conditions for Xenobiotic degradation Ind. & Eng. Chem. Res. 2003, 42(21), 5024 -5030 Air flow § Beaker Rxn § @ 25°C, p. H 5. 5 -6. 5 2. 0 m. L 50/50 hexane/ethyl acetate (extraction only) 10. 0 m. L water 0. 44 m. M EDTA 0. 44 m. M Xenobiotic Stir bar 0. 5 g Fe; 40 -70 mesh 3/15/05 Cheng & Noradoun, University of Idaho 67
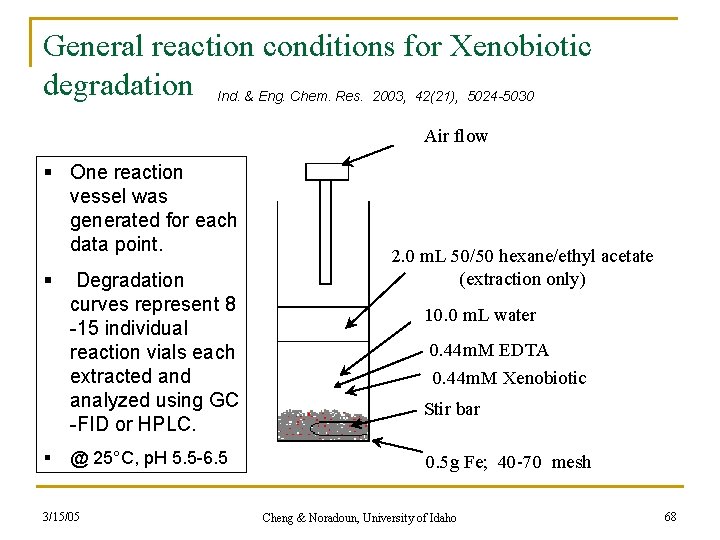
General reaction conditions for Xenobiotic degradation Ind. & Eng. Chem. Res. 2003, 42(21), 5024 -5030 Air flow § One reaction vessel was generated for each data point. § § Degradation curves represent 8 -15 individual reaction vials each extracted analyzed using GC -FID or HPLC. @ 25°C, p. H 5. 5 -6. 5 3/15/05 2. 0 m. L 50/50 hexane/ethyl acetate (extraction only) 10. 0 m. L water 0. 44 m. M EDTA 0. 44 m. M Xenobiotic Stir bar 0. 5 g Fe; 40 -70 mesh Cheng & Noradoun, University of Idaho 68
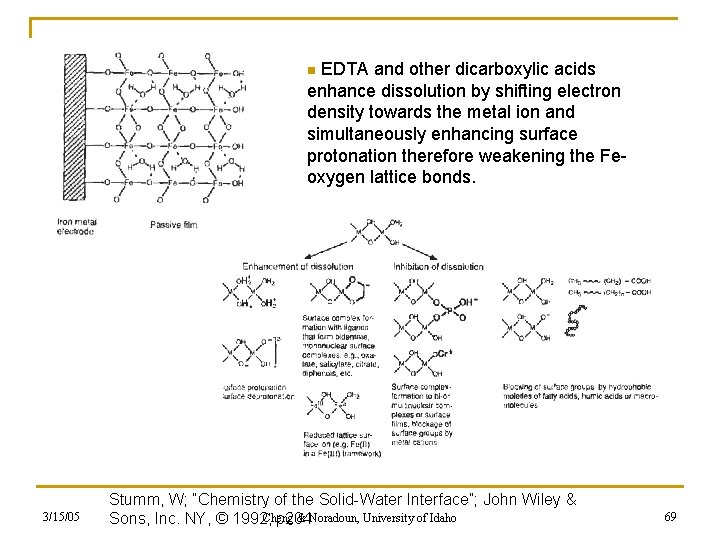
EDTA and other dicarboxylic acids enhance dissolution by shifting electron density towards the metal ion and simultaneously enhancing surface protonation therefore weakening the Feoxygen lattice bonds. n 3/15/05 Stumm, W; “Chemistry of the Solid-Water Interface”; John Wiley & Cheng & Noradoun, University of Idaho Sons, Inc. NY, © 1992, p 204 69
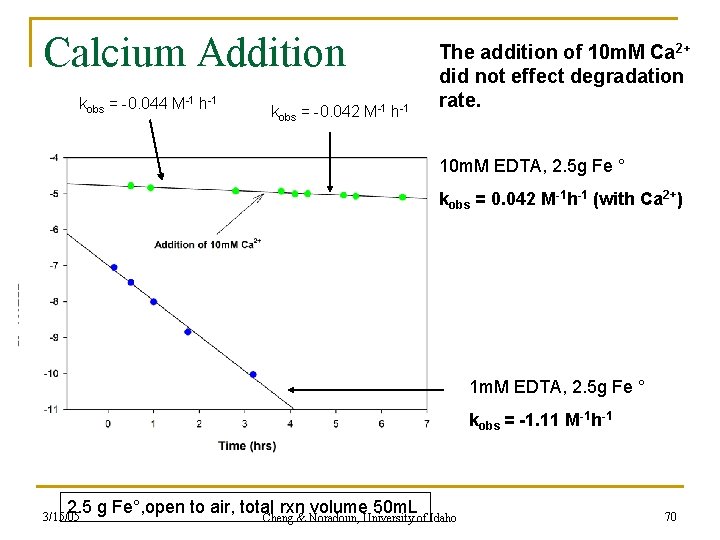
Calcium Addition kobs = -0. 044 M-1 h-1 kobs = -0. 042 M-1 h-1 The addition of 10 m. M Ca 2+ did not effect degradation rate. 10 m. M EDTA, 2. 5 g Fe ° kobs = 0. 042 M-1 h-1 (with Ca 2+) 1 m. M EDTA, 2. 5 g Fe ° kobs = -1. 11 M-1 h-1 2. 5 g Fe°, open to air, total rxn volume 50 m. L Cheng & Noradoun, University of Idaho 3/15/05 70

Calcium Addition cont. n Ca 2+ addition had no overall effect on the rate of degradation n The added Ca 2+ also did not help sequester excess EDTA in solution n Therefore there was no improvement of Fenton Reactivity with the Ca 2+ addition n Alternative way of examining the problem was to hold EDTA concentration constant and vary amount of Fe° present 3/15/05 Cheng & Noradoun, University of Idaho 71
- Slides: 71