Kinetics Kinetics is the branch of chemistry concerned

Kinetics

“Kinetics” is the branch of chemistry concerned with the rate or speed at which chemical reactions occur.

In general, increasing the number of effective collisions between reacting particles at will increase the speed at which the reaction occurs.

There are 6 factors that influence the number of effective collisions and, therefore, the rate or speed of a reaction.

#1 - Bond Type Ionic solutions usually react much more rapidly than covalent solutions. Why?

#2 - Concentration The higher the concentration of reacting particles, the faster a reaction will occur.

#3 - Pressure (gases only) The higher the pressure is, the faster a reaction will occur.

#4 - Temperature The higher the temperature is, the faster a reaction will occur.

#5 - Surface Area The greater the surface area is, the faster a reaction will occur.

#6 - Adding a Catalyst Adding a catalyst lowers the activation energy of a reaction. The reaction will occur more rapidly.

A “Potential Energy Diagram” can be used to show the changes in energy as a chemical reaction occurs.

“Endothermic” reactions must absorb energy in order to occur. Heat Energy is on the left…reactant side!!

In “endothermic” reactions, the potential energy of the reactants is smaller than the potential energy of the products.
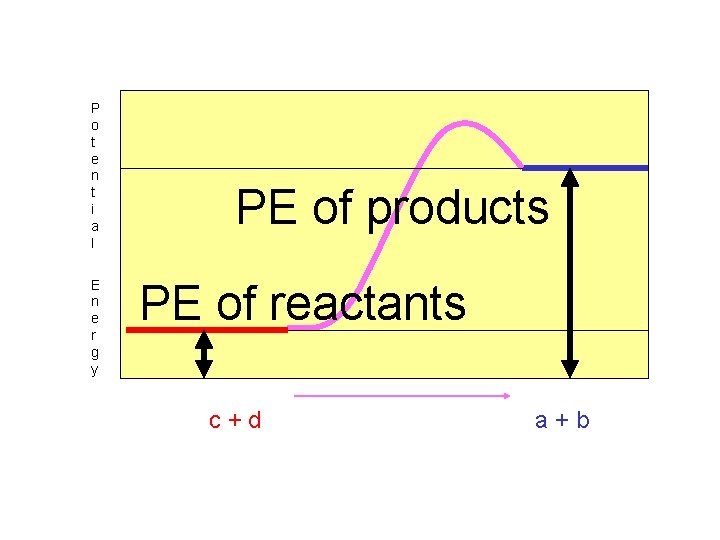
P o t e n t i a l E n e r g y PE of products PE of reactants c+d a+b

The net change in potential energy between reactants and products is called “Heat of Reaction, H, or Enthalpy”.

For endothermic reactions, H is positive. (See Table I)
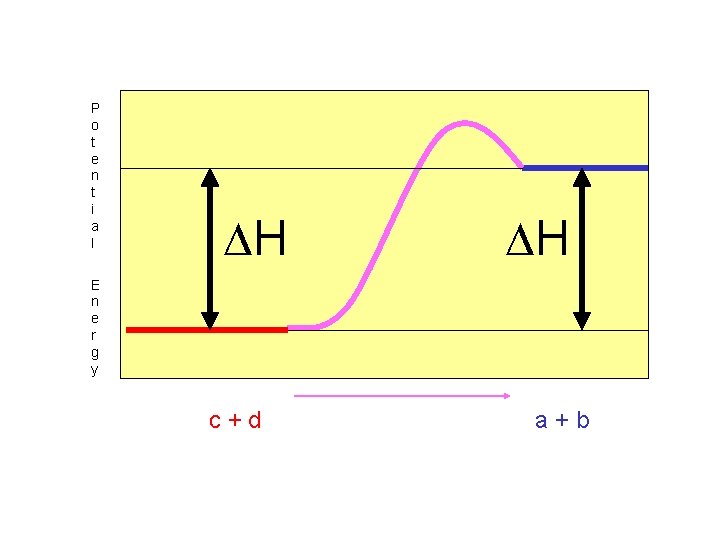
P o t e n t i a l H H E n e r g y c+d a+b

The energy needed to start any reaction (to get over the bump) is called “Activation Energy”.
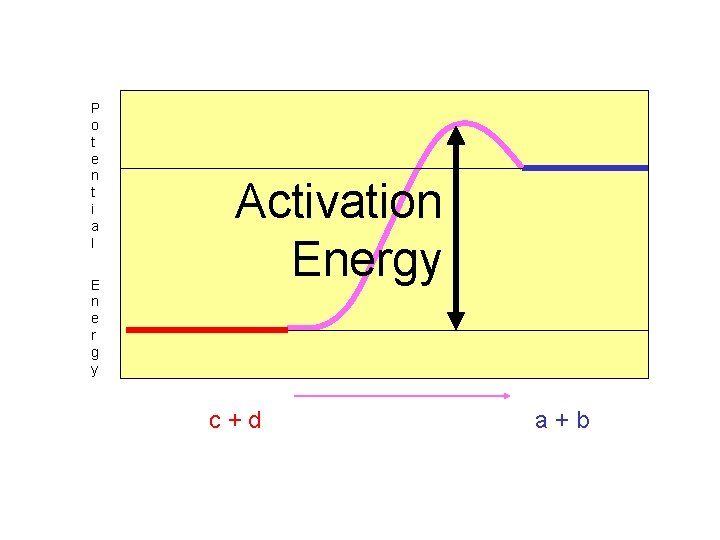
P o t e n t i a l E n e r g y Activation Energy c+d a+b

The “Potential Energy of the Activated Complex” represents the total potential energy of the activated complex.
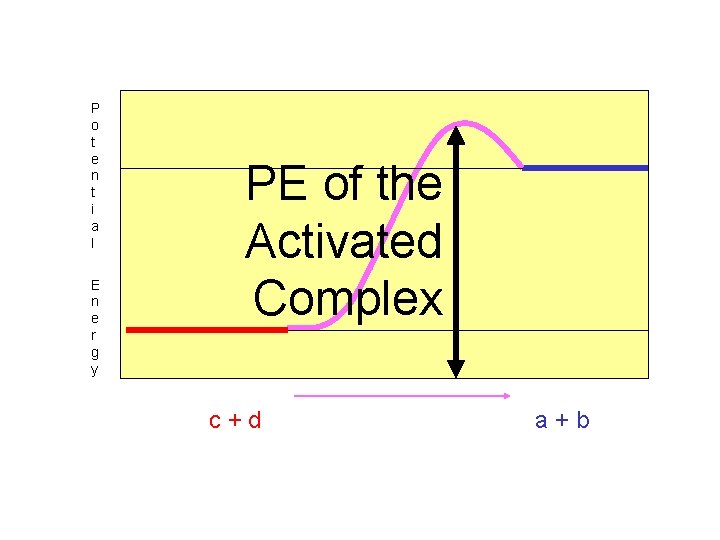
P o t e n t i a l E n e r g y PE of the Activated Complex c+d a+b

Activation Energy with a Catalyst • Lowers the activation energy • Creates an alternate pathway for the reaction

“Exothermic” reactions release energy when they occur. Heat energy is on the right side…products!!

In “exothermic” reactions, the potential energy of the reactants is greater than the potential energy of the products.
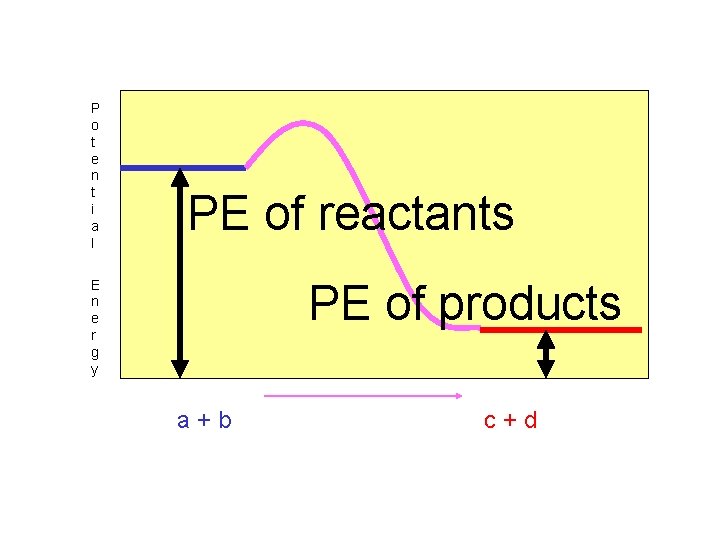
P o t e n t i a l PE of reactants PE of products E n e r g y a+b c+d

H remains the same. Only the sign of H changes! For exothermic reactions, H is negative. (See Table I)
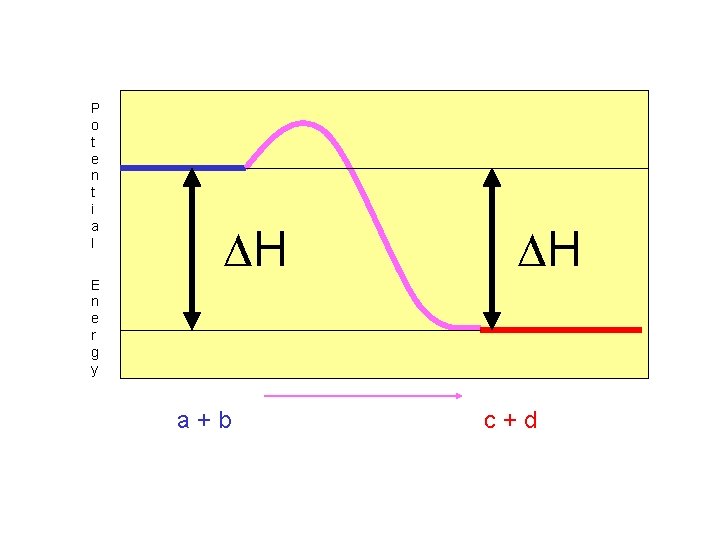
P o t e n t i a l H H E n e r g y a+b c+d

The “Activation Energy” is always smaller for the exothermic reaction. Why?
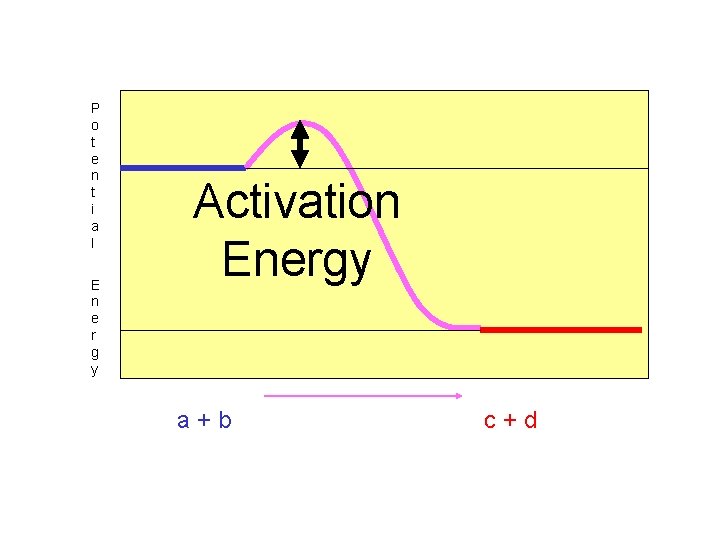
P o t e n t i a l E n e r g y Activation Energy a+b c+d

The “Potential Energy of the Activated Complex” is still the same value.
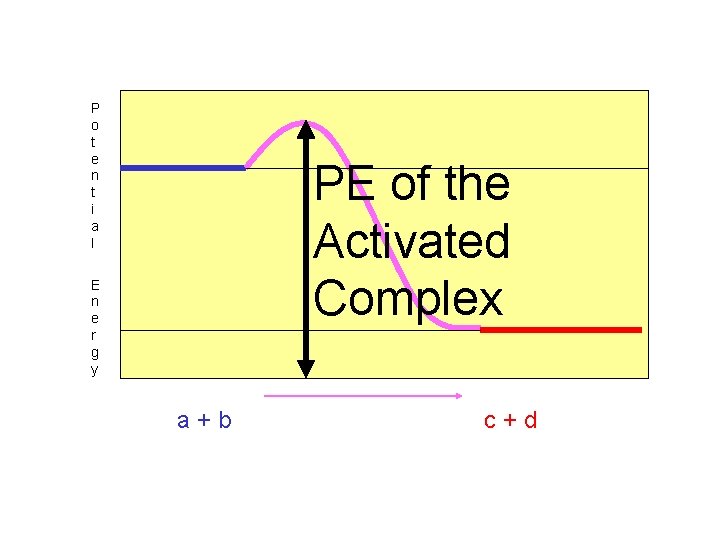
P o t e n t i a l PE of the Activated Complex E n e r g y a+b c+d

the End
- Slides: 32