Kinetics and Equilibrium Unit VI I Kinetics A

Kinetics and Equilibrium Unit VI

I Kinetics A. Kinetics is the study of the rates of reactions and reaction mechanisms Rate § Speed of a reaction Mechanism § Steps involved in a reaction § §

B. Role of Energy in Kinetics Activation Energy (Ea) 1. § Amount of energy needed to start up a reaction Heat of Reaction ( H) 2. Measures the difference between the potential energy of the products and reactants in a reaction § Hreaction = Hproducts –Hreactants Reference Table I § HNO = + 66. 4 k. J 2 § HNH = - 91. 8 k. J 3 § §

C. Types of Reactions 1. Exothermic § § 2. Reactions that release energy § Energy is a product of the reaction § N 2 + 3 H 2→ 2 NH 3 + 91. 8 k. J H is negative § “Feels hot” because it releases energy to you Endothermic § § Reactions that absorb energy § Energy is a reactant in the reaction § N 2 + O 2 + 182. 6 k. J → 2 NO H is positive § “Feels cold” because it absorbs energy from you

D. Collision Theory A reaction occurs when particles collide with sufficient energy and proper alignment l More collisions, faster rate l

E. Factors Affecting the Rate of Reactions 1. Concentration § § § 2. Temperature § § § 3. Increasing concentration increases number of molecules Increases collisions Increases rate Increasing temperature increases speed of molecules Increases collisions Increases rate Surface Area § § § Increasing surface area increases contact with reactants Increases collisions Increases rate

4. Pressure Increase pressure decreases volume of a gas; less space § Increases collisions § Increases rate § 5. Nature of Reactant Ionic bonds break into pieces which increase concentration § Increases collisions § Increases rate § Covalent bonds do not breakdown ; react slower § 6. Catalyst Changes the mechanism of the reaction § Lowers activation energy making reaction occur easier § More collisions can occur § Rate increases §
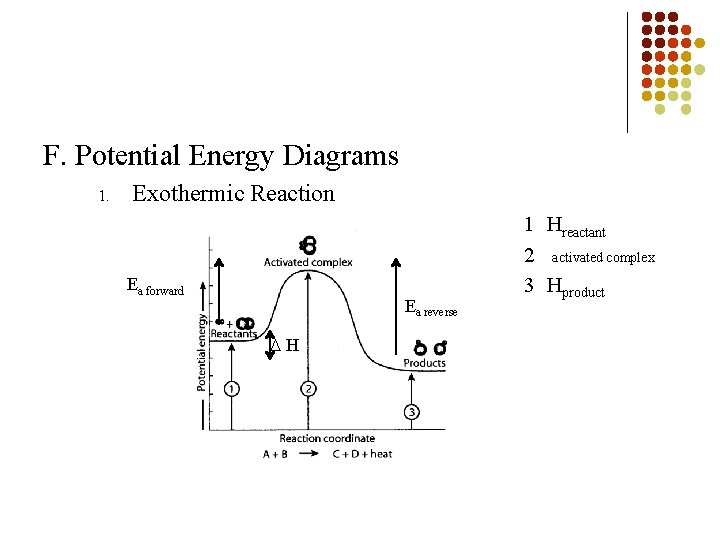
F. Potential Energy Diagrams 1. Exothermic Reaction Ea forward Ea reverse H 1 Hreactant 2 activated complex 3 Hproduct
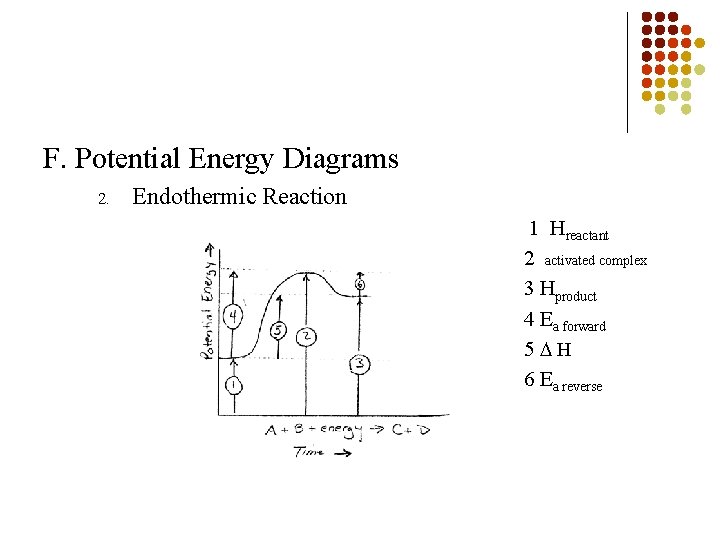
F. Potential Energy Diagrams 2. Endothermic Reaction 1 Hreactant 2 activated complex 3 Hproduct 4 Ea forward 5 H 6 Ea reverse

II Equilibrium is the balance between the rates of two opposing reactions • Example of two opposing reactions: H 2 O(s) → H 2 O(l) H 2 O(s) ← H 2 O(l) melting freezing written in equilibrium notation H 2 O(s) ↔ H 2 O(l) phase change » forward direction—melt » reverse direction—freeze At equilibrium, • • Rates are equal Concentrations are constant

A. Types of Equilibrium Reactions 1. Phase Equilibrium -any phase changes 2. Solution Equilibrium -balance between dissolved and undissolved solute --saturated solution Na. Cl(s) ↔ Na. Cl(aq) § forward direction—dissolve § reverse direction—precipitate 3. Chemical Equilibrium -balance between forward and reverse reactions

B. Le Chatelier’s Principle systems that are at equilibrium are stable and want to remain at equilibrium • • when equilibrium reactions are “stressed”, they will “shift” to establish equilibrium stresses include: o o concentration changes temperature changes pressure changes or volume changes (gases only) add a catalyst

1. Concentration Changes When changing concentration, use the teetertauter technique • Given the reaction: 2 NO 2 ↔ N 2 O 4 What will happen to [N 2 O 4 ] if [NO 2 ] is increased? ([ – – – ] represents concentration) Tilt left Shift right Makes more N 2 O 4

2. Temperature Changes When changing temperature, l l l Use the teeter-tauter technique OR If temp increases, shift away from heat If temp decreases, shift toward heat Given the reaction: 2 NO 2 + 300 k. J ↔ N 2 O 4 What will happen to [N 2 O 4 ] if the temperature is increased? Tilt left Shift right Makes more N 2 O 4 Heat (300 k. J) is on the left Equilibrium shifts away from the left side Shifts right N 2 O 4 increases

3. Pressure Changes Volume Changes When pressure changes occur, only gases will be effected (g) – • Count gases in the system » An increase in pressure (decrease in volume) causes a shift toward the smallest side of the reaction Given the reaction: 2 NO 2 (g) ↔ N 2 O 4 (g) What will happen to [N 2 O 4 ] if the pressure is increased? – Count gases – – – 2 gases left side………. 1 gas right side Shifts right (fewer gases) Makes more N 2 O 4

4. Addition of a Catalyst When a catalyst is added to a system at equilibrium, both forward and reverse reactions increase rate There is no effect on the equilibrium l No shift will occur l

Test yourself l Given the reaction: N 2(g) + 3 H 2 (g) ↔ 2 NH 3 (g) + 80 k. J l What will happen to the concentration of NH 3 if l l H 2 increases? Temperature increases? Pressure increases? A catalyst is added?

III Enthalpy and Entropy There are TWO factors which determine if a reaction will occur spontaneously or not Enthalpy (ΔH) A. l The natural tendency is to change to a lower energy state l Exothermic direction is preferred Entropy (ΔS) B. l Entropy measures randomness or disorder l Greater disorder (messy), higher entropy § § l (solid) lowest entropy (liquid) (aqueous) (gas) highest entropy High entropy is preferred

Spontaneous or Nonspontaneous? C(s) + O 2(g) ↔CO 2(g) + 120 k. J ∆H = -120 k. J Exothermic ∆ S solid/gas to gas only Increase in entropy Spontaneous

Spontaneous or Nonspontaneous? N 2 (g) + 4 H 2(g) + Cl 2(g) + 93 k. J ↔ 2 NH 4 Cl(s) ∆H = +93 k. J Endothermic ∆ S gas to solid Decrease in entropy Nonspontaneous

Spontaneous or Nonspontaneous? 2 H 2(g) + O 2(g) ↔ 2 H 2 O(l) + 80 k. J ∆H = -80 k. J Exothermic ∆ S gas to liquid Decrease in entropy ? ? ? ? ? “Free Energy” needs to be used
- Slides: 21