KineticMolecular Theory of Gases Molecular Basis of Gas
Kinetic-Molecular Theory of Gases

Molecular Basis of Gas Pressure – N-Molecules : n n Now consider N-molecules, instead of just one, to be in the container. Our model claims that these molecules act independently of one another. The pressure required to confine each of these molecules is given by the expression: the sum of these pressure terms gives the net pressure as :

n n If is written to indicate an average value, we have (1) : A relation between the pressure and the speeds of the molecules, rather than component of the speeds is more convenient. The velocity vector can be resolved into three orthogonal components to give.

n n n For a large number of molecules moving in random directions : - And thus: Substitution of last Eq. in Eq. (1) gives the desired relation between P and V : -

n n n This important equation is as far as one can go to explain the basis for the pressure of a gas from the four kineticmolecular postulates set out earlier. An additional postulate must be added to reach a result that can be compared with the empirical ideal-gas laws. Derivation of PV = n. RT : The average kinetic energy of a molecule of a gas indicated by:

n n This quantity can be made to appear in the expression of Eq. Multiplication and division by 2 creates the expression: Now you can recognize the kinetic energy term, and write: This result for N molecules can be expressed in terms of the number of moles of molecules.

n n n The symbol N is used for the number implied by 1 mol, and n is the number of moles of molecules. Then : The PV expression of Eq. can now be written as: - If (KE) is used to denote the translational kinetic energy of 1 mol of molecules this equation becomes: - N = Avogadro’s number
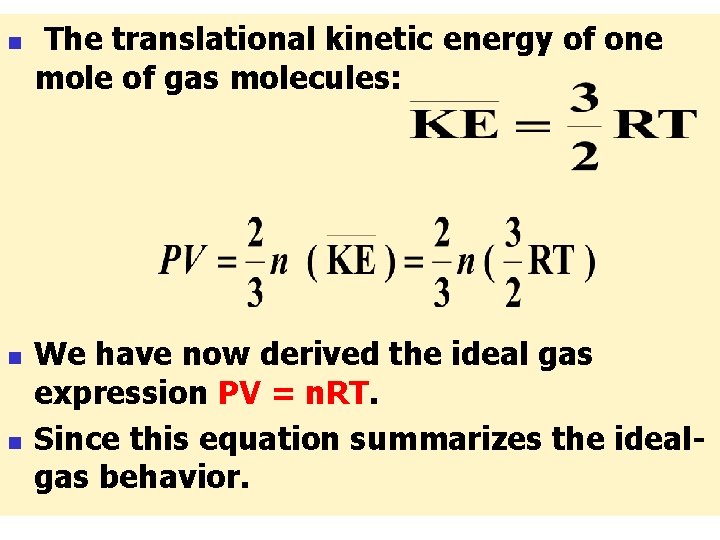
n n n The translational kinetic energy of one mole of gas molecules: We have now derived the ideal gas expression PV = n. RT. Since this equation summarizes the idealgas behavior.

Molecular energies and speeds n n The average translational kinetic energies and speeds of the molecules of a gas can be calculated. the kinetic energy of 1 mol of the molecules of a gas is equal to: R = 8. 3143 J. k-1 mol-1 The average translational energy of the individual molecules, depends on only the temperature of the gas.

n n None of the properties of the molecules not the atomic makeup, not the mass, not the shape need be considered. The average kinetic energy of gas molecules depends on only the temperature. Molecular Translational Energies: The translational kinetic energy of 1 mol of gas molecules at 25 o. C (298. 15 Ko) is: = 3718 J. mol-1 = 3. 718 k J. mol-1

n n n For dealing with the energies of individual atoms or molecules, it is convenient to introduce a constant k, called the Boltzmann constant, as: Notice that the Boltzmann constant (KB) is the gas constant per molecule.
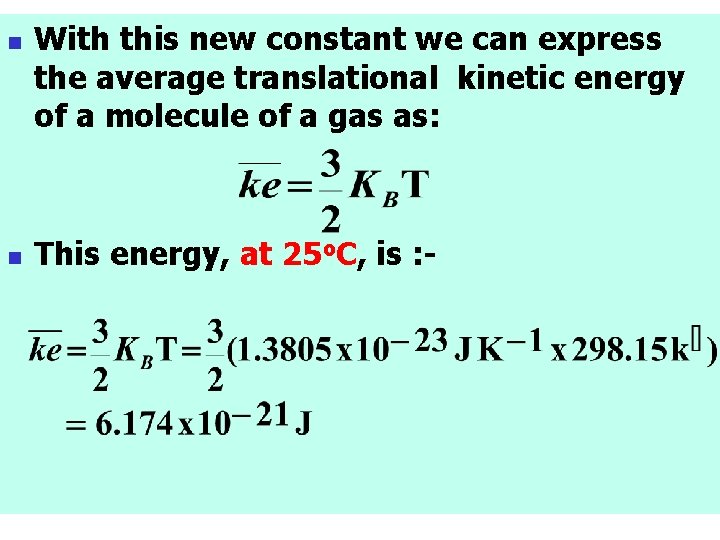
n n With this new constant we can express the average translational kinetic energy of a molecule of a gas as: This energy, at 25 o. C, is : -

Speeds of Molecules: - n Energies have broader applications in chemistry than do speeds. But at first, it is easier to appreciate speeds. Consider a gas that contains molecules of a particular mass. Molecular speed values can be obtained by writing the kinetic energy of mol of these molecules as: - n Where M is the mass of 1 mol of molecules. n

The cumbersome term is known as the root-mean square (rms) speed. n
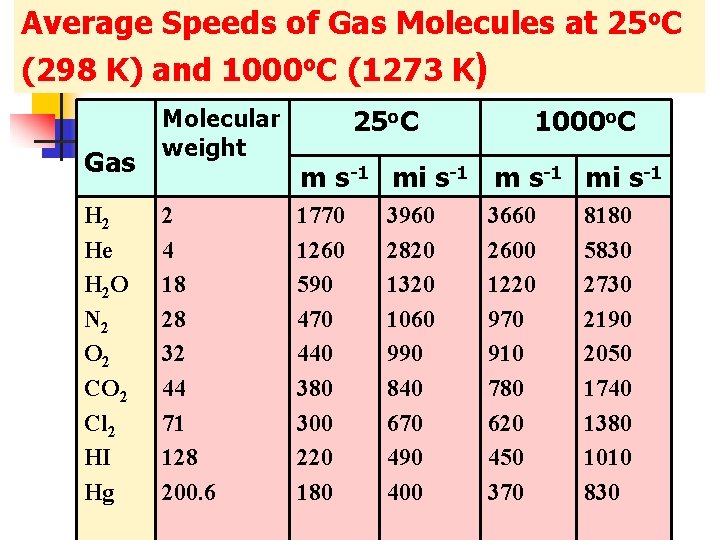
Average Speeds of Gas Molecules at 25 o. C (298 K) and 1000 o. C (1273 K) Gas H 2 He H 2 O N 2 O 2 Cl 2 HI Hg Molecular weight 2 4 18 28 32 44 71 128 200. 6 25 o. C 1000 o. C m s-1 mi s-1 1770 1260 590 470 440 380 300 220 180 3960 2820 1320 1060 990 840 670 490 400 3660 2600 1220 970 910 780 620 450 370 8180 5830 2730 2190 2050 1740 1380 1010 830

Example : Calculate the rms speed of nitrogen molecules at 25 o. C. n For nitrogen molecules (N 2): M has the value 28. 02 g or in the SI mass unit, 0. 02802 kg. The rms speed at 25 o. C is

Remember that the average molecular translational kinetic energy is the same for different molecules at any temperature, molecules with small mass must have greater average speeds than molecules with a large mass.

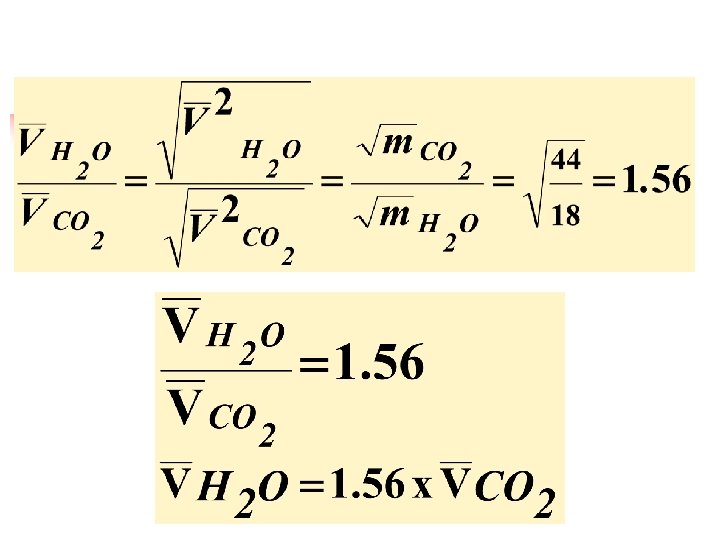
Comparison between molecular speeds and translational kinetic energy of two different gas molecules: n n At the same conditions of both pressure and temperature: Compare: (a) the average kinetic energies and (b) the average speeds of H 2 O molecules, which have relatively small masses, and CO 2 molecules, which have relatively large masses, in the air around you.

n n 1 - at the same conditions of T, P: Both H 2 O and CO 2 molecules have the average translational kinetic energy Due to the law: . 2 - The average kinetic energy is given by:

Degrees of Freedom : n n It is sometimes convenient to deal with the components of molecular speeds along three perpendicular direction. To do so, we use the relation: -

n n The components of the kinetic energy for motion along these directions are shown by multiplying by (½ m) to get : - Since the averages of the component energies are equal:

n n n it follows that : - The three perpendicular direction in which a velocity can be resolved are examples of degrees of freedom. We can say that the average molecular translational energy per degree of freedom is [ ½ k. BT ].
- Slides: 23