Kinetic Theory or life as a molecule N






















- Slides: 40

Kinetic Theory (or life as a molecule) N 2 O 2 N 2 Prof. Fred Remer University of North Dakota N 2 O 2 Kinetic Theory H 2 O N 2

Objective • Be able to define temperature and pressure • Be able to perform simple calculations using the Ideal Gas Law Prof. Fred Remer University of North Dakota Kinetic Theory

Reading • Wallace and Hobbs, pp. 64, 74 • Bohren and Albrecht – pp. 1 -30 Prof. Fred Remer University of North Dakota Kinetic Theory

Kinetic Theory • Once Upon A Time There Was A Molecule Prof. Fred Remer University of North Dakota Kinetic Theory

Kinetic Theory • The molecule had no size or internal structure, but it was a happy molecule. Her name was Point Mass. m = mass Prof. Fred Remer University of North Dakota Kinetic Theory

Kinetic Theory • One day, Point Mass decided to move. He only moved in one direction. He moved a a constant speed. x - direction velocity = vx Prof. Fred Remer University of North Dakota Kinetic Theory

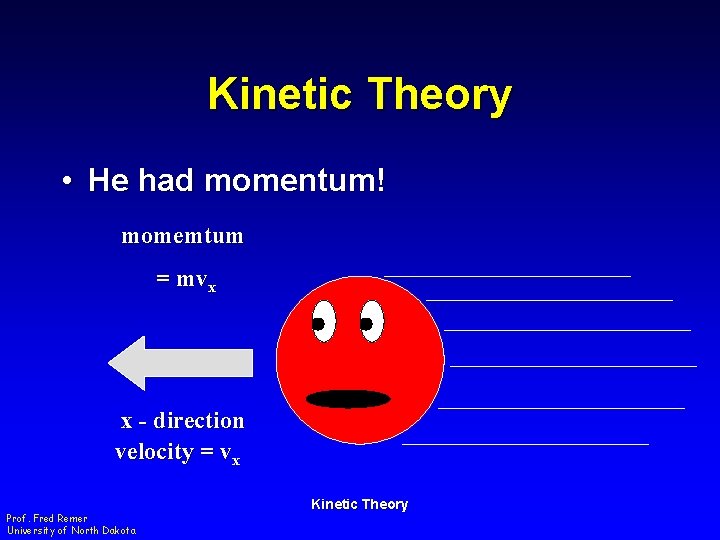
Kinetic Theory • He had momentum! momemtum = mvx x - direction velocity = vx Prof. Fred Remer University of North Dakota Kinetic Theory

Kinetic Theory • He had so much momentum, he could not slow down when he saw the wall! Prof. Fred Remer University of North Dakota Kinetic Theory

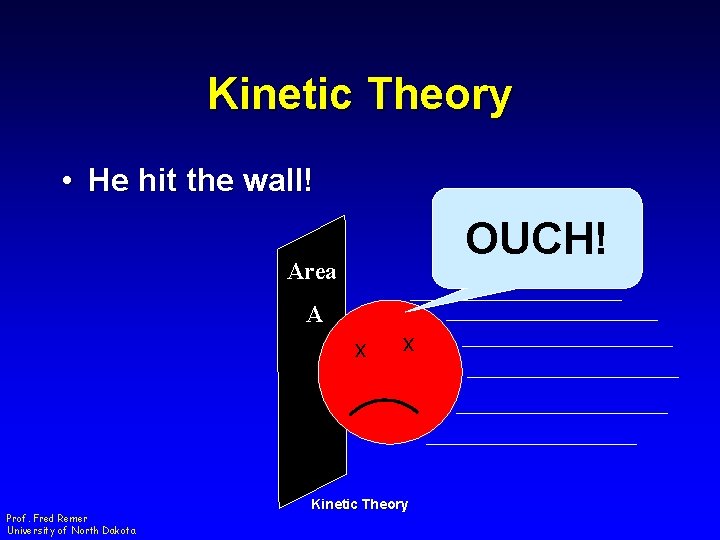
Kinetic Theory • He hit the wall! OUCH! Area A x Prof. Fred Remer University of North Dakota x Kinetic Theory

Kinetic Theory • But much to his surprise, he rebounded! His collision was perfectly elastic! No energy was lost in the collision. Prof. Fred Remer University of North Dakota Kinetic Theory

Kinetic Theory • He had the same momentum leaving the wall as he had before the collision, but in the opposite direction. momentum Prof. Fred Remer University of North Dakota Kinetic Theory

Kinetic Theory • His change in momentum was Change in Momentum Prof. Fred Remer University of North Dakota Kinetic Theory

Kinetic Theory • The force exerted on Point Mass by the wall was or Prof. Fred Remer University of North Dakota Kinetic Theory

Kinetic Theory • Point Mass had other friends who are molecules identical to himself. Prof. Fred Remer University of North Dakota Kinetic Theory

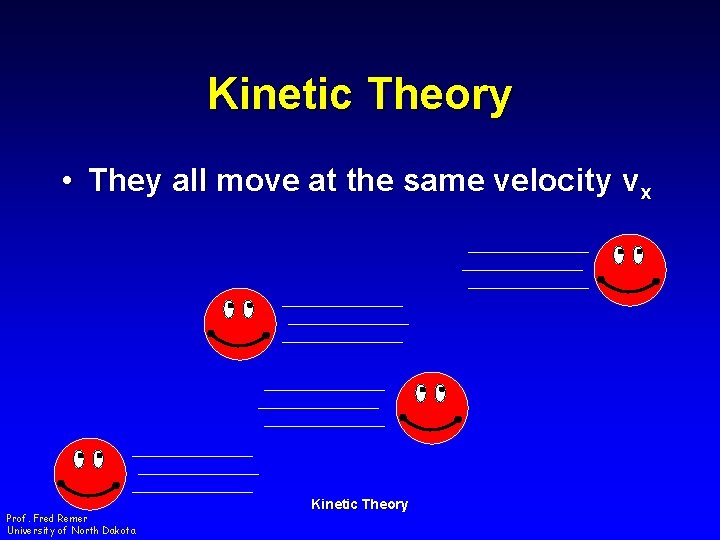
Kinetic Theory • They all move at the same velocity vx Prof. Fred Remer University of North Dakota Kinetic Theory

Kinetic Theory • The molecules do not interact between themselves. . . Prof. Fred Remer University of North Dakota Kinetic Theory

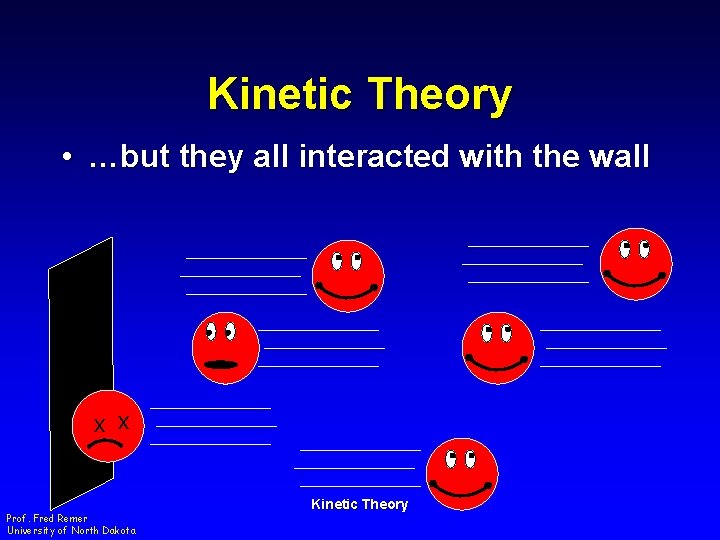
Kinetic Theory • …but they all interacted with the wall x x Prof. Fred Remer University of North Dakota Kinetic Theory

Kinetic Theory • The number of molecules (N) in a given volume (V) is the number density (n) V = Volume N = # of molecules n = number density Prof. Fred Remer University of North Dakota Kinetic Theory

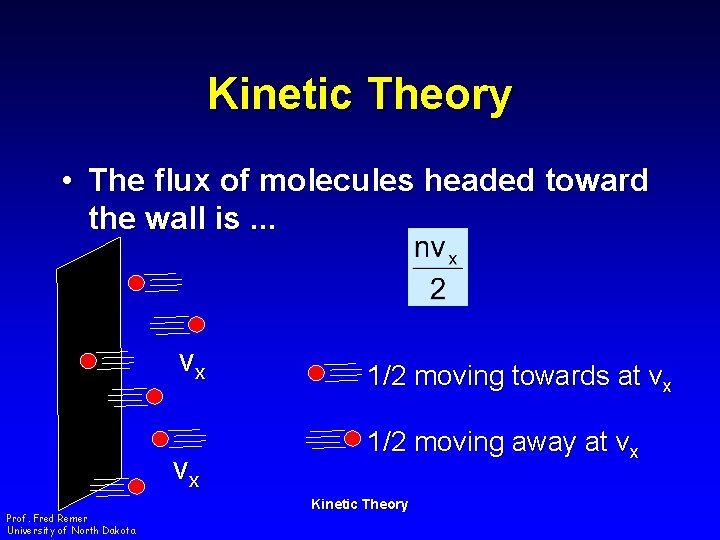
Kinetic Theory • The flux of molecules headed toward the wall is. . . vx vx Prof. Fred Remer University of North Dakota 1/2 moving towards at vx 1/2 moving away at vx Kinetic Theory

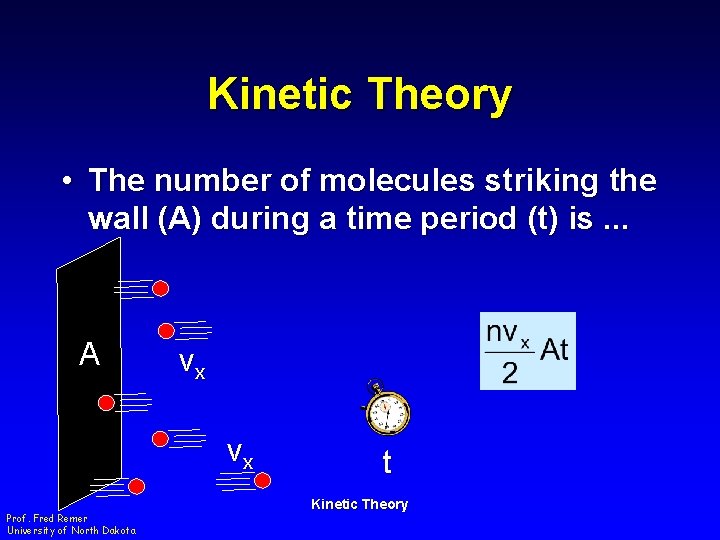
Kinetic Theory • The number of molecules striking the wall (A) during a time period (t) is. . . A vx vx Prof. Fred Remer University of North Dakota t Kinetic Theory

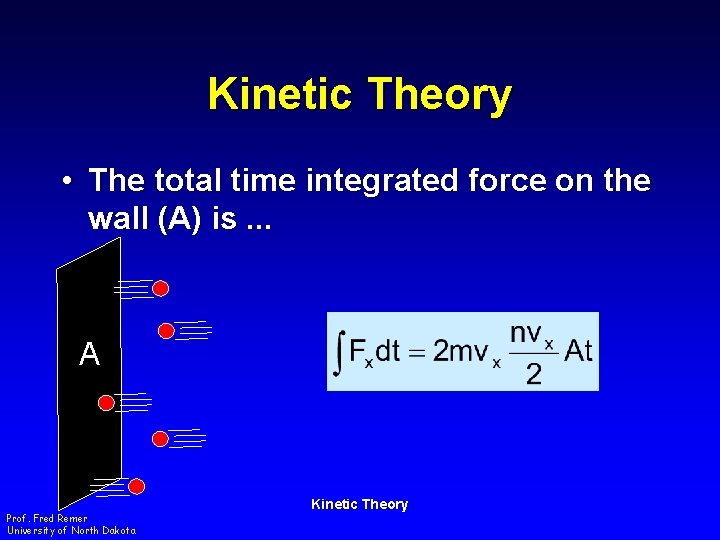
Kinetic Theory • The total time integrated force on the wall (A) is. . . A Prof. Fred Remer University of North Dakota Kinetic Theory

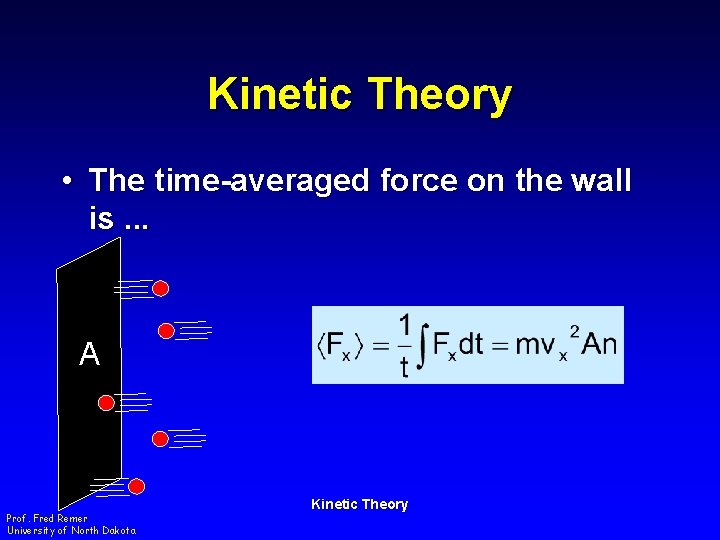
Kinetic Theory • The time-averaged force on the wall is. . . A Prof. Fred Remer University of North Dakota Kinetic Theory

Kinetic Theory • The average force per unit area is. . . A Prof. Fred Remer University of North Dakota Kinetic Theory

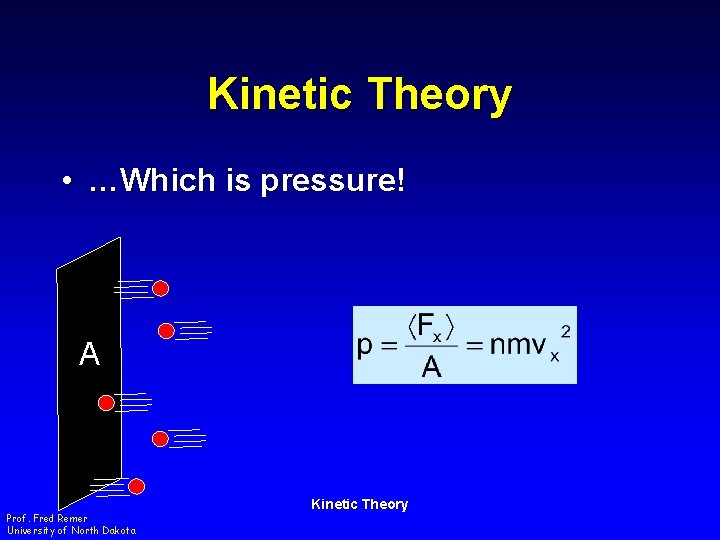
Kinetic Theory • …Which is pressure! A Prof. Fred Remer University of North Dakota Kinetic Theory

Kinetic Theory • Lets modify one assumption. The molecules are moving at different speeds. Prof. Fred Remer University of North Dakota Kinetic Theory

Kinetic Theory • Let’s replace vx 2 with an average. Prof. Fred Remer University of North Dakota Kinetic Theory

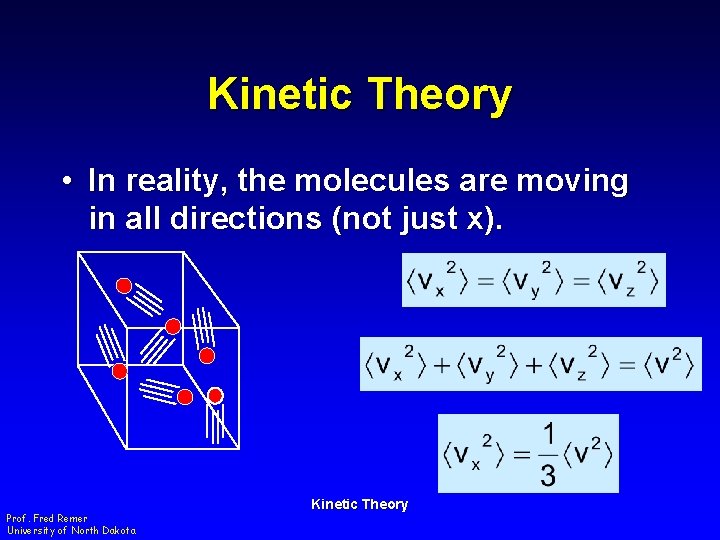
Kinetic Theory • In reality, the molecules are moving in all directions (not just x). Prof. Fred Remer University of North Dakota Kinetic Theory

Kinetic Theory • Substitute back into the equation Prof. Fred Remer University of North Dakota Kinetic Theory

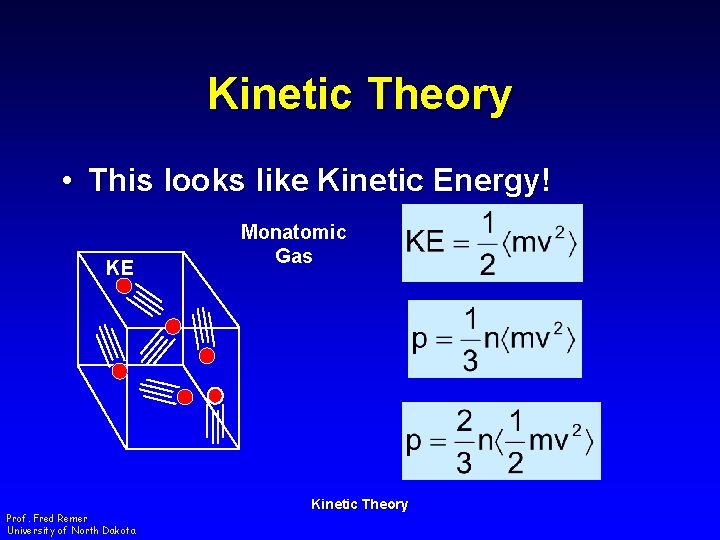
Kinetic Theory • This looks like Kinetic Energy! KE Prof. Fred Remer University of North Dakota Monatomic Gas Kinetic Theory

Kinetic Theory • Definition of Temperature where k = Boltzmann Constant = 1. 38 x 10 -23 J/K – Temperature is a measure of the average KE of the molecules! Prof. Fred Remer University of North Dakota Kinetic Theory

Kinetic Theory • Substitute temperature into pressure Prof. Fred Remer University of North Dakota Kinetic Theory

Kinetic Theory • Ideal Gas Law where. . . or Prof. Fred Remer University of North Dakota p = pressure V = volume N = number of molecules T = temperature k = Boltzman Constant Kinetic Theory

Kinetic Theory • Monatomic Molecules – Energy Is a Result of Atom’s Motion Only Prof. Fred Remer University of North Dakota Kinetic Theory

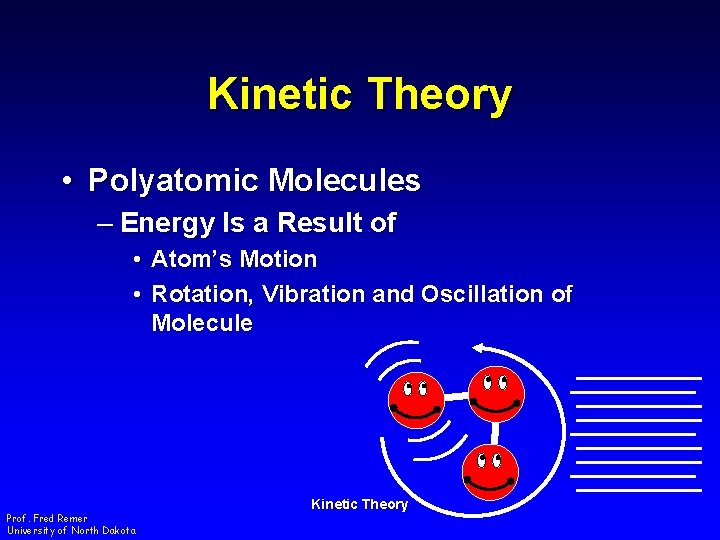
Kinetic Theory • Polyatomic Molecules – Energy Is a Result of • Atom’s Motion • Rotation, Vibration and Oscillation of Molecule Prof. Fred Remer University of North Dakota Kinetic Theory

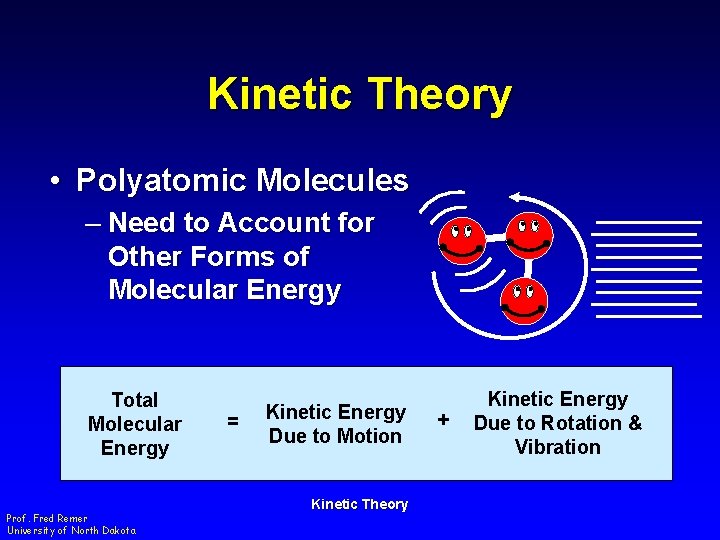
Kinetic Theory • Polyatomic Molecules – Need to Account for Other Forms of Molecular Energy Total Molecular Energy Prof. Fred Remer University of North Dakota = Kinetic Energy Due to Motion Kinetic Theory + Kinetic Energy Due to Rotation & Vibration

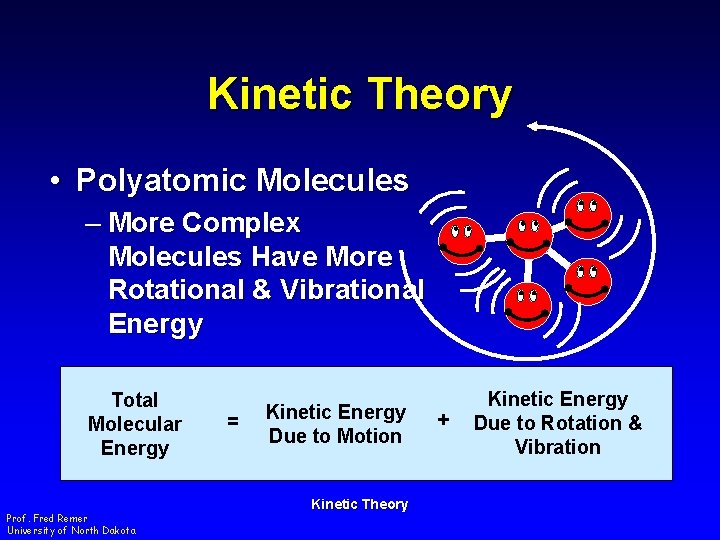
Kinetic Theory • Polyatomic Molecules – More Complex Molecules Have More Rotational & Vibrational Energy Total Molecular Energy Prof. Fred Remer University of North Dakota = Kinetic Energy Due to Motion Kinetic Theory + Kinetic Energy Due to Rotation & Vibration

Kinetic Theory • Polyatomic Molecules – More Complex Molecules Have More Rotational & Vibrational Energy Total Molecular Energy Prof. Fred Remer University of North Dakota = Kinetic Energy Due to Motion Kinetic Theory + Kinetic Energy Due to Rotation & Vibration

Kinetic Theory • Polyatomic Molecules – Low Pressure • Approximates Ideal Gas – High Pressure • Deviates More Total Molecular Energy Prof. Fred Remer University of North Dakota = Kinetic Energy Due to Motion Kinetic Theory + Kinetic Energy Due to Rotation & Vibration

Kinetic Theory • Summary – pressure is a measure of the total kinetic energy of molecules, the force per unit area of these molecules – temperature is proportional to the average kinetic energy of molecules – from this kinetic theory viewpoint, we can derive the perfect gas law: Prof. Fred Remer University of North Dakota Kinetic Theory

Kinetic Theory • We will return to the perfect gas law from a macroscopic point of view and derive exactly the same relationship: where. . . or Prof. Fred Remer University of North Dakota p = pressure V = volume, n = number of moles N = number of molecules T = temperature k = Boltzmann constant m = mass, M = molecular weight Kinetic Theory