KINETIC THEORY OF GASES PRESENTED BY MR B

KINETIC THEORY OF GASES PRESENTED BY: MR. B. SESHA SAI, PGT, (PHY) K. V. EOI Kathmandu

CONTENTS ASSUMPTIONS RMS VELOCITY RELATION BETWEEN K. E. AND TEMP. BOYLE’S LAW CHARLE’S LAW GAY LUSSAC’S LAW PERFECT GAS EQUATION AVOGADRO’S LAW

CONTENTS ABSOLUTE ZERO TEMP. DERIVATION OF GAS PRESSURE DEGREES OF FREEDOM LAW OF EQUIPARTITION OF ENERGY Cp/Cv OF MONOATOMIC GAS Cp/Cv OF DIATOMIC GAS Cp/Cv OF TRIATOMIC(LINEAR) GAS Cp/Cv OF TRIATOMIC(NON-LINEAR) GAS

ASSUMPTIONS Ø Ø Ø All the molecules of a gas are identical as regards their shape and mass. The molecules of different gases are different. The gas molecules behave as rigid, elastic and smooth spheres. The size of gas molecule is extremely small as compared to intermolecular distance. The molecules of gas are in continuous random motion. They move with all possible velocities in all possible directions. They obey Newton’s Laws of Motion. The molecules collide with one another and also with the walls of the container in perfectly elastic manner. The number of molecules per unit volume of gas is very large.

Between two successive collisions molecule moves with uniform velocity. The distance between two successive collisions is called free path. The average distance traveled by molecule between successive collisions is called mean free path. Ø Their energy is wholly kinetic. Ø The collisions are instantaneous. The time elapsed in each collision is very small as compared to the time elapsed between two successive collisions. Ø

DERIVATION EXPRESSION OF GAS PRESSURE Consider a gas enclosed in a cubical vessel of side l having perfectly elastic walls. Volume of gas=l 3 No. of gas molecules=n Mass of each molecule=m Mass of gas M=mn Let O be the origin. Consider a molecule moving with velocity C 1 as in fig. u 1, v 1, w 1 , are reaction component of C 1 are along OX, OY OZ respectively. Ø

Ø Let the molecule strike the face A 1 of the vessel. Only the component u 1(perpendicular to A 1) is effective. Initial momentum of molecule along X-axis=mu 1 Collision is perfectly elastic. So K. E. is conserved. So molecule rebound with velocity =-u 1 Final momentum of molecule along X-axis=-mu 1 Change in momentum =-mu 1=-2 mu 1 After rebound the molecule strikes A 2, it rebounds with velocity u 1 and again collide against A 1. The molecule travels distance 2 l before colliding with face A 1 again. Time interval between successive collisions on face A 1 Ø Number of collision on A 1 by molecule in 1 second Ø Ø Ø Contd.

Contd. Ø Change in momentum of molecule in 1 sec= Ø From Newton’s IInd Law Force exerted on molecule by face A 1 = Ø From Newton’s IIIrd Law, Force exerted by molecule on face A 1 =f 1= Ø Ø The forces due to other molecules Ø Total force exerted by the n molecules on face A 1 Contd.

Contd. Ø Pressure on face A 1 Ø Let Px and Py be pressures exerted by gas on faces perpendicular to Y and Z axis.

A gas exerts the same pressure in all directions. Thus;

Where; C=Mean Square Velocity

RELATION BETWEEN K. E AND TEMPERATURE Ø Let us consider one mole of a gas. Let M and V be its mass and volume respectively. The pressure exerted by gas is given by: But, PV=RT

BOYLE’S LAW Which is Boyle’s Law.

BOYLE’S LAW GRAPH

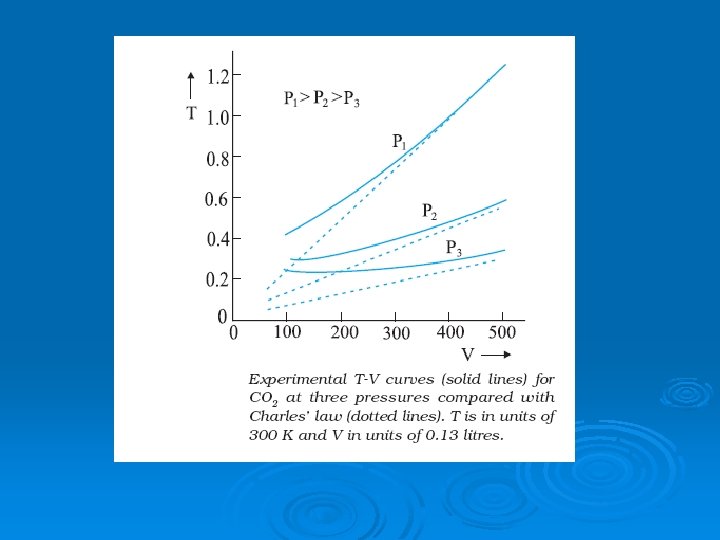
CHARLE’S LAW Ø The volume of given mass of gas is directly proportional to its absolute temperature provided the pressure remains constant. According to Kinetic theory of gases, At constant pressure for given mass of gas. Which is Charle’s Law.

GAY LUSSAC’S LAW The pressure P of a given mass of gas is directly proportional to its absolute temperature T, provided the volume V remains constant. According to Kinetic theory of gases, At constant volume for given mass of gas: Which is Gay Lussac’s Law.

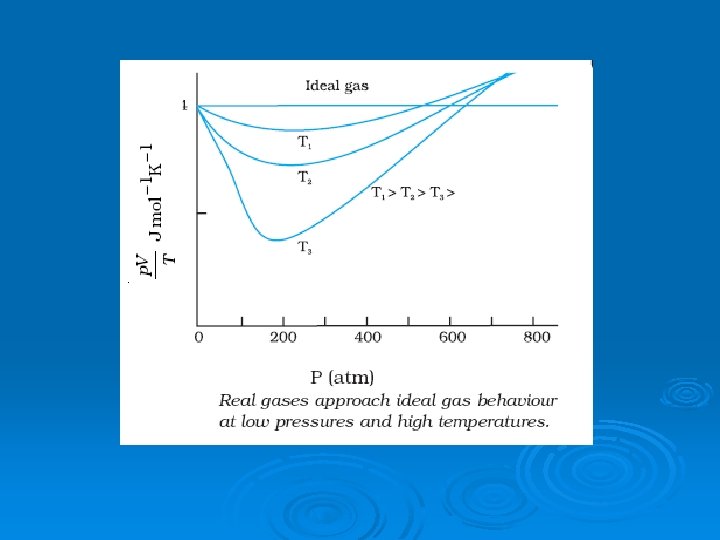
PERFECT GAS EQUATION Ø According to Kinetic theory of gases, Where R=universal gas constant.

AVOGADRO’S LAW Equal volumes of all gases at the same temperature and pressure contain equal number of molecules. Proof: Consider two gases having same temperature, pressure and volume. Let one gas contain n 1 molecules each of mass m 1. Let second gas contain n 2 contain molecules each of mass m 2. Let c 1 and c 2 be rms velocities of two gases. and Contd.

Contd. Since both gases are at equal temperature; Thus, average translational K. E. per molecule is same for each gas. Thus, from I and II; n 1= n 2 This proves the Avogadro’s Law.

ABSOLUTE ZERO OF TEMPERATURE Ø Absolute zero of temperature is that temperature at which the velocities of the gas molecule becomes zero. We know that; average translational K. E. per molecule is When T=0, c=0. The absolute zero for an ideal gas is -273 o. C.
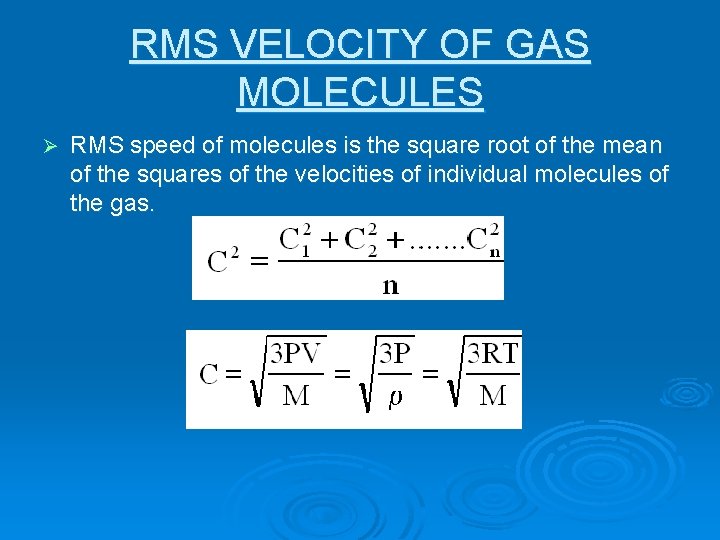
RMS VELOCITY OF GAS MOLECULES Ø RMS speed of molecules is the square root of the mean of the squares of the velocities of individual molecules of the gas.

DEGREE OF FREEDOM Ø The total number of co-ordinates or independent quantities required to completely specify the position and configuration of a dynamical system is called degree of freedom. If A be number of particles in system R is number of independent relations between them, then, degree of freedom (N)= 3 A-R. For monoatomic gas, N=3(1)-0=3 For diatomic gas, N=3(2)-1=5 For linear triatomic gas, N=3(3)-2=7 For non-linear triatomic gas, N=3(3)-3=6

LAW OF EQUIPARTITION OF ENERGY For a dynamical system in thermal equilibrium the energy of system is equally distributed among the various degrees of freedom and energy associated with each degree of freedom=1/2 KT. Ø This law holds good for all degrees of freedom whether translational, rotational or vibrational. Proof : The average translational K. E. of a gas molecule Ø But for gas;

Cp/Cv FOR MONOATOMIC GASES Ø Total energy associated with one monoatomic gas molecule (U)=(3/2)RT.

Cp/Cv FOR DIATOMIC GASES Ø Total energy associated with one diatomic gas molecule (U)=(5/2)RT.

Cp/Cv FOR TRIATOMIC(LINEAR) GASES Ø Total energy associated with one triatomic gas molecule (U)=(7/2)RT.

Cp/Cv FOR TRIATOMIC(NON-LINEAR) GASES Ø Total energy associated with one triatomic gas molecule (U)=(6/2)RT=3 RT.

- Slides: 30