Kinetic Theory of Gases 1 PHYS116 A prof








- Slides: 12

Kinetic Theory of Gases -1 PHYS-116 A prof. Ramayya 11/29/2010 Momchil Velkovsky

Avogadro’s Number One mole is an amount of matter (atoms or molecules) equal to the number of atoms in a 12 g sample of carbon-12. The number of atoms or molecules in a mole is called Avogadro’s Number, NA.

Avogadro’s Number NA links our world (scale) with the atomic scale. Our world Atomic world n – number of moles N – number of molecules M – mass of a mole m – mass of a molecule n=N/NA, M=m*NA, Msample= n M = N m

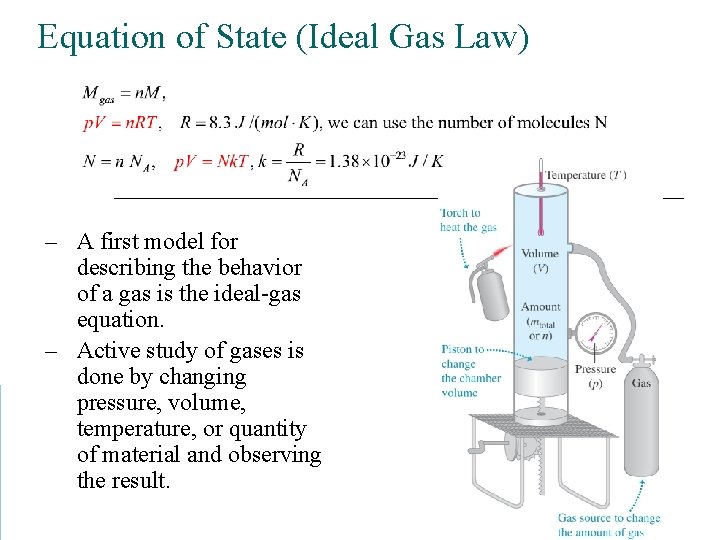
Equation of State (Ideal Gas Law) – A first model for describing the behavior of a gas is the ideal-gas equation. – Active study of gases is done by changing pressure, volume, temperature, or quantity of material and observing the result.

A quantity of an ideal gas is contained in a balloon. Initially the gas temperature is 27°C. You double the pressure on the balloon and change the temperature so that the balloon shrinks to one-quarter of its original volume. What is the new temperature of the gas? A. 54°C B. 27°C C. 13. 5°C D. – 123°C E. – 198°C

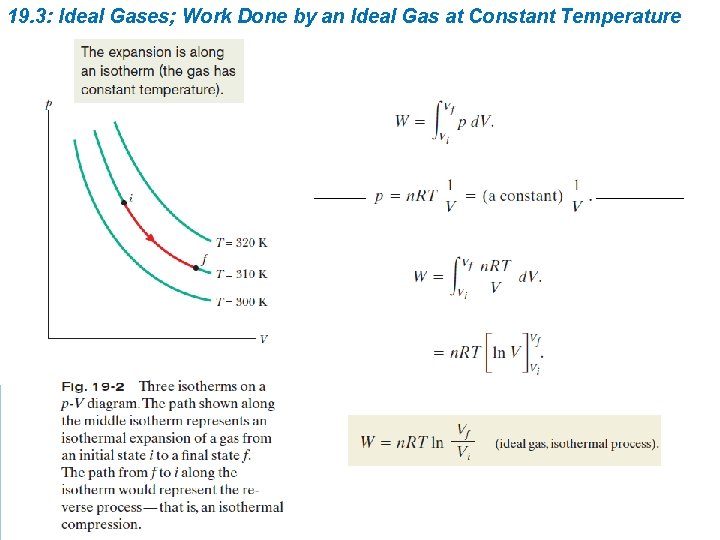
19. 3: Ideal Gases; Work Done by an Ideal Gas at Constant Temperature

19. 3: Ideal Gases; Work Done at Constant Volume and Constant Pressure W done by an ideal gas (or any other gas) during any process, such as a constantvolume process and a constant-pressure process. If the volume of the gas is constant, If, instead, the volume changes while the pressure p of the gas is held constant,

Atomic/molecular collisions and pressure

vrms and Total Kinetic Energy

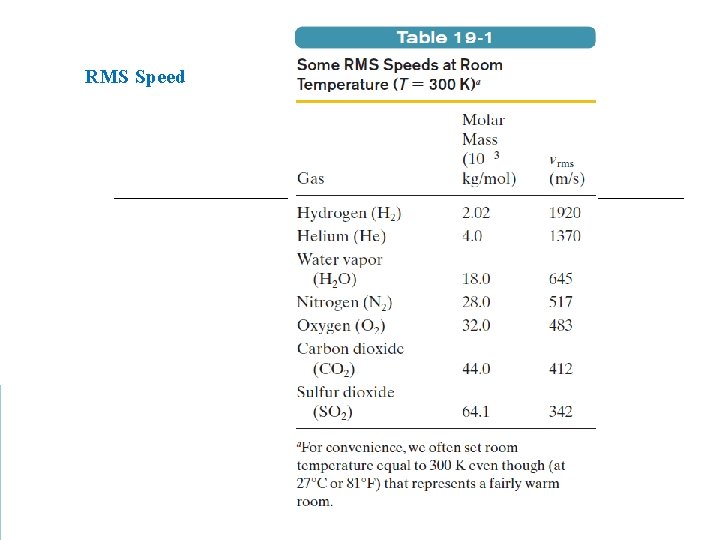
RMS Speed

This p. V–diagram shows three possible states of a certain amount of an ideal gas. Which state is at the highest temperature? A. state #1 B. state #2 C. state #3 D. Two of these are tied for highest temperature. E. All three of these are at the same temperature. p 3 2 1 O V

19. 6: Mean Free Path The mean free path, l, is the average distance traversed by a molecule between collisions. Taking into account that the other molecules move too gives a correction: