Kinetic Molecular Theory and the Combined Gas Law

Kinetic Molecular Theory and the Combined Gas Law

The Kinetic Molecular Theory of Gases “Ideal Gas Laws”

An “Ideal Gas” will obey or follow the Gas Laws perfectly under all conditions. Ideal Gases must meet 4 criteria:

Criteria #1 The gas particles must be in continuous, random motion. (This is true!)

Criteria #2 There must be no net loss of energy when gas particles collide. (This is true!)

Criteria #3 The volume of the gas particles is not significant to the overall volume of the gas. (This one is shakey!)

Is the gas particle volume significant?

Is the gas particle volume significant?

Is the gas particle volume significant?

Criteria #4 There are NO forces of attraction between the gas particles. (This is NOT true!)

Since Criteria #3 and #4 are NOT met, there is no such thing as an “Ideal Gas” or a perfect gas.

A “Real Gas” will follow the gas laws perfectly under most conditions. Gases we are familiar with are “Real Gases”.

The “Real Gas” that is closest to being an “Ideal Gas” is Helium (He). Hydrogen (H 2) is second.

Real Gases do NOT follow the Gas Laws very well when the pressure is high and the temperature is low. Why?

High pressure and low temperature will change a gas into a liquid. Liquids do NOT follow the Gas Laws!

The Combined Gas Law

Volume measures the amount of space a sample occupies. We will use liters (L) or milliliters (m. L) to measure volume.

Temperature measures the average speed of the particles in a sample. We will measure temperature in Kelvin (K) when using the Gas Laws.

Pressure measures the collisions that occur between particles in a sample. We will use 2 units to measure pressure.

atmospheres = atm kilopascals = k. Pa

1 atm = 101. 3 k. Pa

Standard temperature and pressure (STP) for a gas (Table A) is: º OC or 273 K 1 atm or 101. 3 k. Pa

Boyle’s Law states that the pressure of a gas sample will vary indirectly with its volume, if the temperature is constant.

In plain English, this means when the pressure increases, the volume must decrease and visa/versa.

PV = k (constant) or P 1 V 1 = P 2 V 2
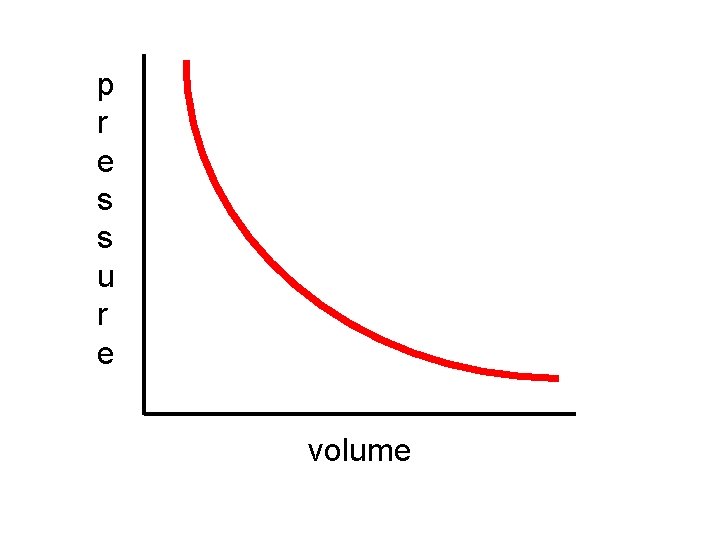
p r e s s u r e volume

Charles’s Law states that the volume of a gas sample will vary directly with its absolute (K) temperature, if the pressure is constant.

In plain English, this means when the volume increases, the Kelvin temperature also increases and visa/versa.

V = k (constant) T or V 1 = V 2 T 1 T 2
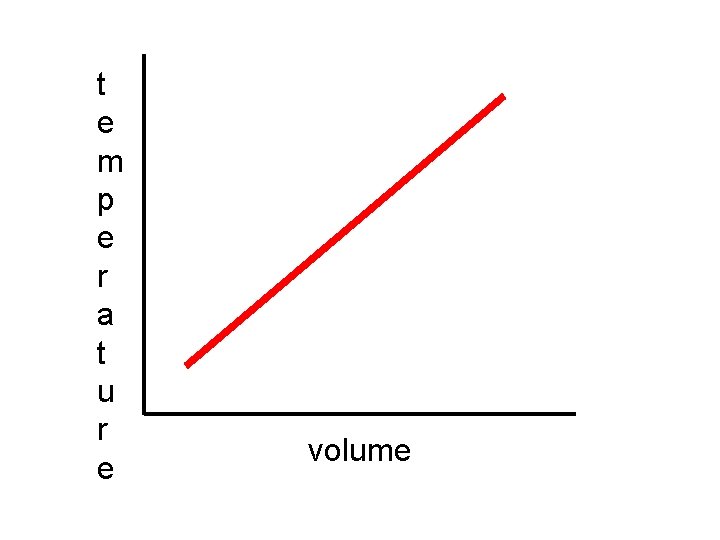
t e m p e r a t u r e volume

There are 3 problems that arise when using just Boyle’s or Charles’s Law.

Problem #1 What if the pressure and the temperature of a gas sample change and the volume is constant?
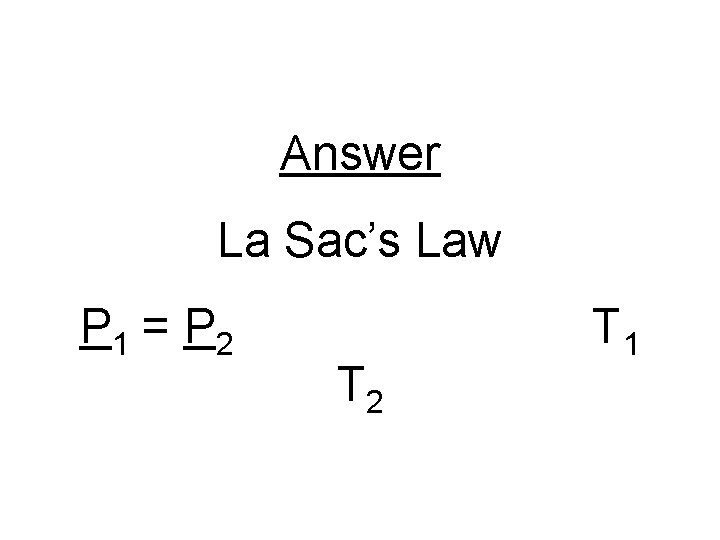
Answer La Sac’s Law P 1 = P 2 T 1

Problem #2 How can you remember who’s law to use?

Problem #3 What if the pressure, volume, and temperature all change (nothing is constant) ?

Answer the Combined Gas Law

The Combined Gas Law combines Boyle’s, Charles’s, and La Sac’s Laws into 1 easy to remember formula!

The Combined Gas Law P 1 V 1 = P 2 V 2 T 1 T 2 temperature must be in K (Kelvin)! (See Table T)

the End
- Slides: 39