Kilimanjaro Clinical Research Institute KCRI PANDORA Laboratory quality

Kilimanjaro Clinical Research Institute (KCRI) PANDORA: Laboratory quality control in low and middle income countries Quality management - Auditing 22 October 2020 Presented by Davis Kuchaka

Quality Management - Auditing • Understand the concept of Auditing in Laboratory Quality management Learning objectives • Understand the kinds of auditing in laboratories • Understand the areas to be audited in the laboratories

What is an Audit The ISO 9000: 2005 definition “a systematic, independent and documented process for obtaining evidence and evaluating it objectively to determine the extent to which required criteria are fulfilled”

What is an Audit A GCP (ICH-GCP 1. 6) definition “a systematic and independent examination of trialrelated activities and documents to determine whether the evaluated trial-related activities were conducted and the data recorded, analysed and accurately reported according to the protocol, sponsor’s SOPs, GCP and the applicable regulatory requirements”

Why perform an audit? For the purpose of accreditation, certification or licensure – External audit Performance of the laboratory and whether it is in compliance with policy requirements Internal audit • conducted by groups or agencies from outside the laboratories • staff working in one area of the lab conduct audits on another area of the same laboratory

An audit, allows the laboratory to understand how well it is performing when compared to a benchmark or standard. This information about its performance is important for: • planning and implementing the quality system • monitoring effectiveness of the quality system • correcting any deficiencies that are identified • working toward continuous improvement
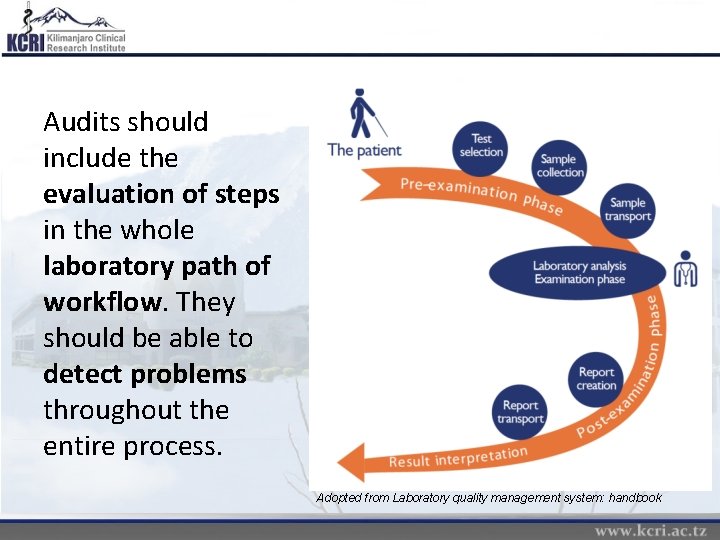
Audits should include the evaluation of steps in the whole laboratory path of workflow. They should be able to detect problems throughout the entire process. Adopted from Laboratory quality management system: handbook

Auditing The value of a well-designed audit is that it will reveal weaknesses in the pre-exam, exam and post-exam phases During audits, information is gathered about: • processes and operating procedures • staff competence and training • equipment • environment • handling of samples • quality control and verification of results • recording and reporting practices

• Audit findings are compared with the laboratory’s internal policies and to a standard or external benchmark. • Any breakdown in the system or departure from procedures will be identified and form a basis for continuous improvement process

Sample forms and documents

• • • Laboratory readiness checklist Continuous QC improvement form Contamination rates Reagent QC forms Recording and processing samples Equipment maintenance and calibration schedule

Resources • TBA Myco. Lab QC Manual version 2, 2 Mar 2019 • Laboratory Quality Management: Handbook • Global Health Training Centre

Acknowledgement
- Slides: 13