Kidney By Dr Abdel Aziz M Hussein Lecturer


Kidney By Dr. Abdel Aziz M. Hussein Lecturer of Medical Physiology


Test Yourself The cortex of the kidney contains the • • • a. hilus. b. glomeruli. c. perirenal fat. d. renal pyramids. e. renal pelvis.

• A layer of fibrous connective tissue that surrounds each kidney is the • a. hilum. • b. renal pelvis. • c. renal sinus. • d. renal capsule. • e. perirenal fat.

Renal Blood Flow
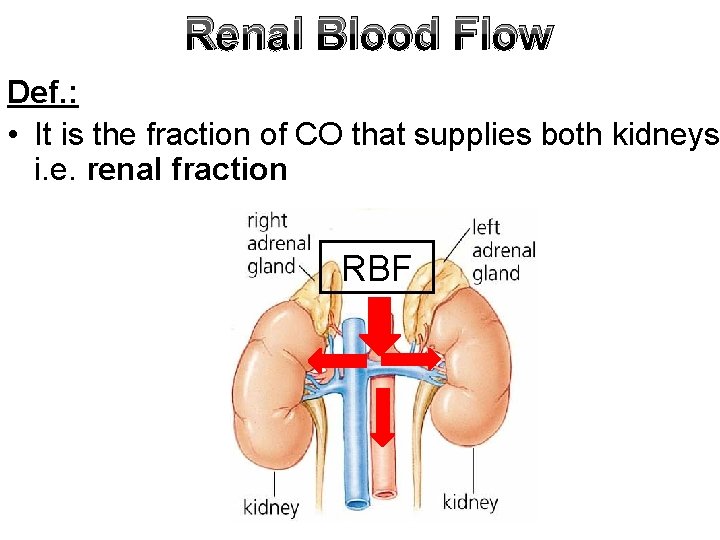
Renal Blood Flow Def. : • It is the fraction of CO that supplies both kidneys i. e. renal fraction RBF
Renal Blood Flow Value: • ¼ CO or 1200 ml/min or 4 ml/ 1 gm kidney tissues Significance: • Is high to ensure high GFR NOT to supply excess O 2 for excess metabolism
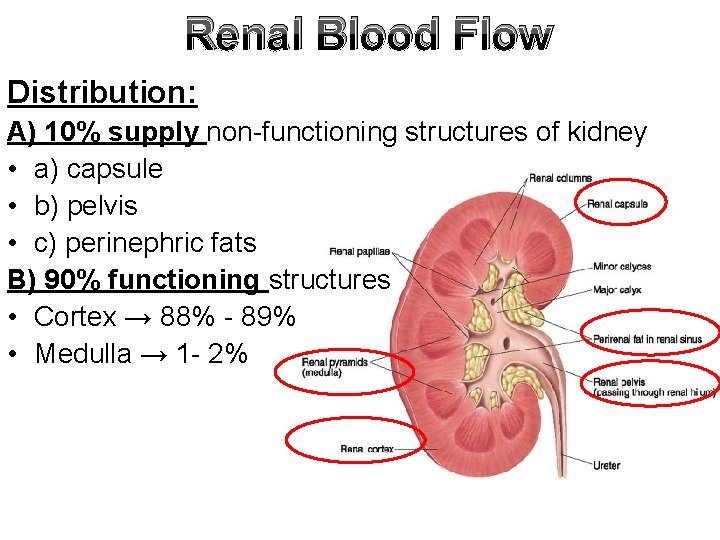
Renal Blood Flow Distribution: A) 10% supply non-functioning structures of kidney • a) capsule • b) pelvis • c) perinephric fats B) 90% functioning structures • Cortex → 88% - 89% • Medulla → 1 - 2%
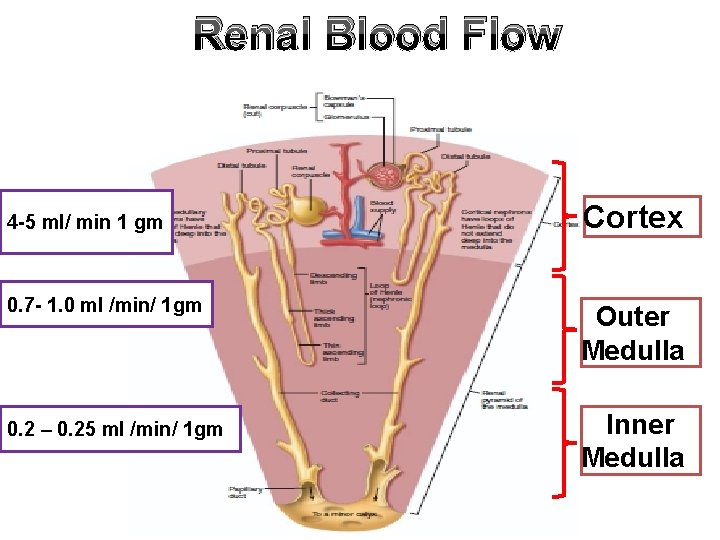
Renal Blood Flow 4 -5 ml/ min 1 gm Cortex 0. 7 - 1. 0 ml /min/ 1 gm Outer Medulla 0. 2 – 0. 25 ml /min/ 1 gm Inner Medulla
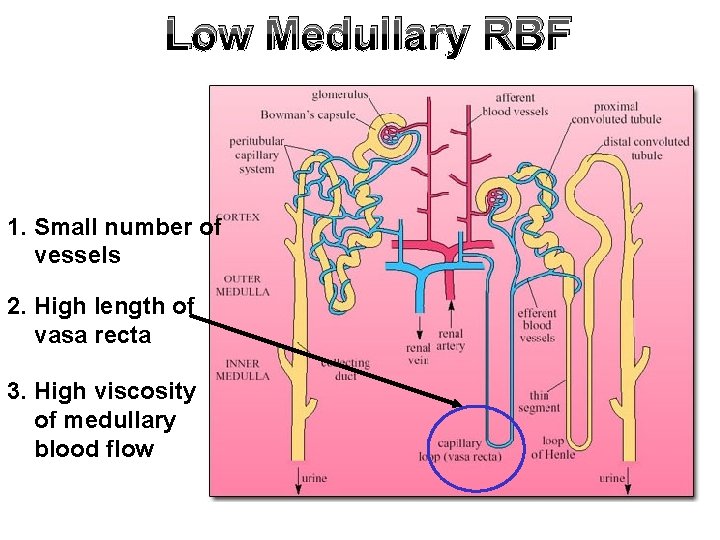
Low Medullary RBF 1. Small number of vessels 2. High length of vasa recta 3. High viscosity of medullary blood flow

Regulation of Renal Blood Flow
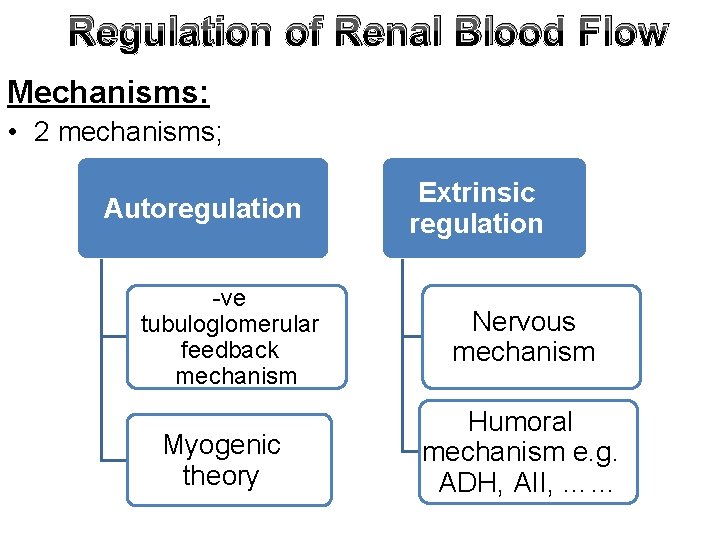
Regulation of Renal Blood Flow Mechanisms: • 2 mechanisms; Autoregulation -ve tubuloglomerular feedback mechanism Myogenic theory Extrinsic regulation Nervous mechanism Humoral mechanism e. g. ADH, AII, ……

Autoregulation of RBF Def. , : • It is the ability of the kidney to keep its RBF and GFR at nearly normal levels in the face of reasonable changes of ABP Range of Autoregulation: • From 80 – 180 mm. Hg • is not perfect 100% since RBF and GFR show changes by less than 10% with previous range of ABP
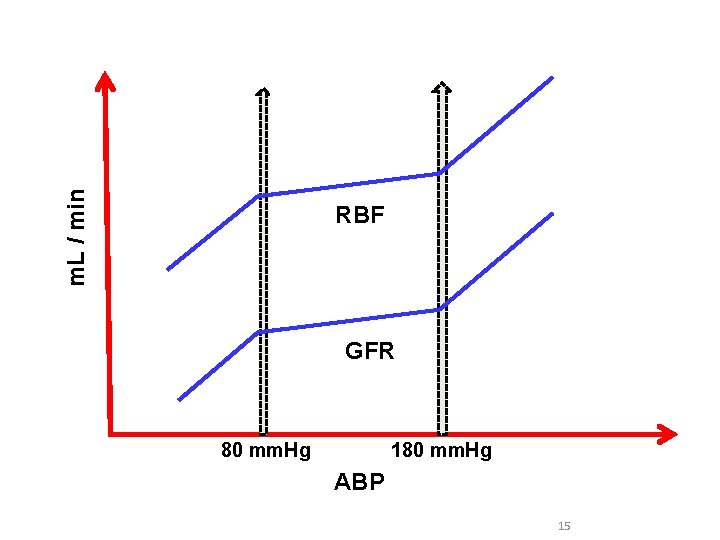
m. L / min RBF GFR 80 mm. Hg 180 mm. Hg ABP 15

Autoregulation of RBF Significance: • To minimize effect of change of ABP on GFR and hence on Na+ and water excretion or helps to decouple renal regulation of salt and water excretion from fluctuation of ABP Mechanism of Autoregulation: • Mainly by variation of afferent arteriolar resistance • Two theories; 1. Myogenic theory 2. Tubuloglomerular –ve feedback theory
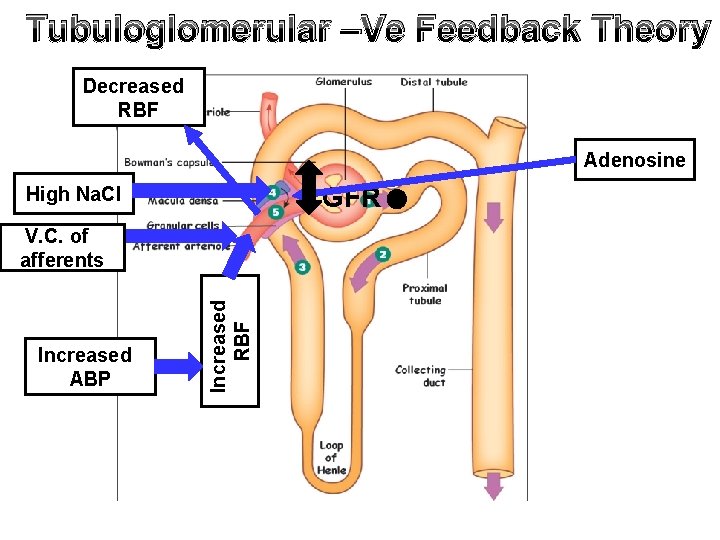
Tubuloglomerular –Ve Feedback Theory Decreased RBF Adenosine GFR High Na. Cl Increased ABP Increased RBF V. C. of afferents
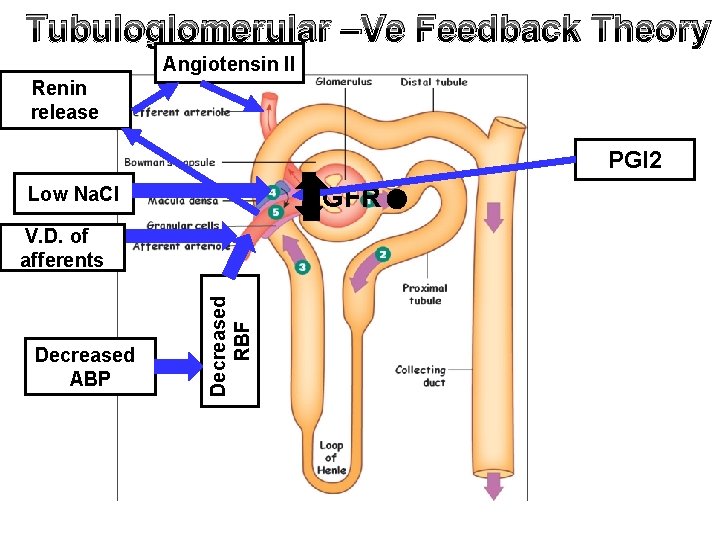
Tubuloglomerular –Ve Feedback Theory Angiotensin II Renin release PGI 2 GFR Low Na. Cl Decreased ABP Decreased RBF V. D. of afferents
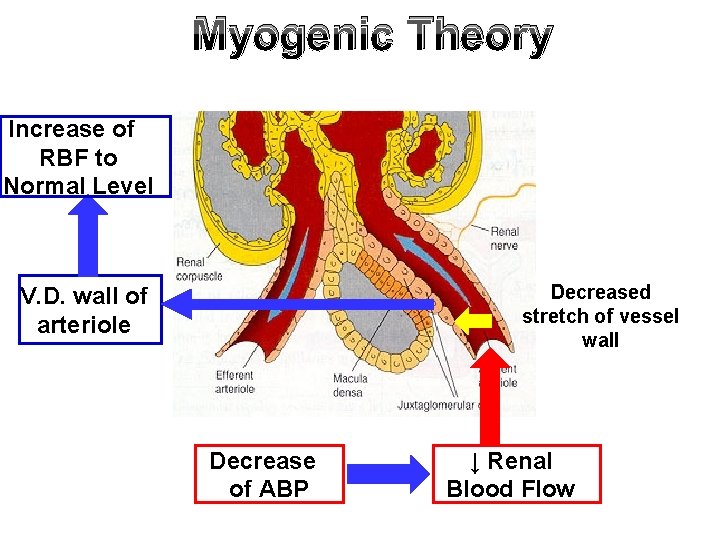
Myogenic Theory Increase of RBF to Normal Level Decreased stretch of vessel wall V. D. wall of arteriole Decrease of ABP ↓ Renal Blood Flow
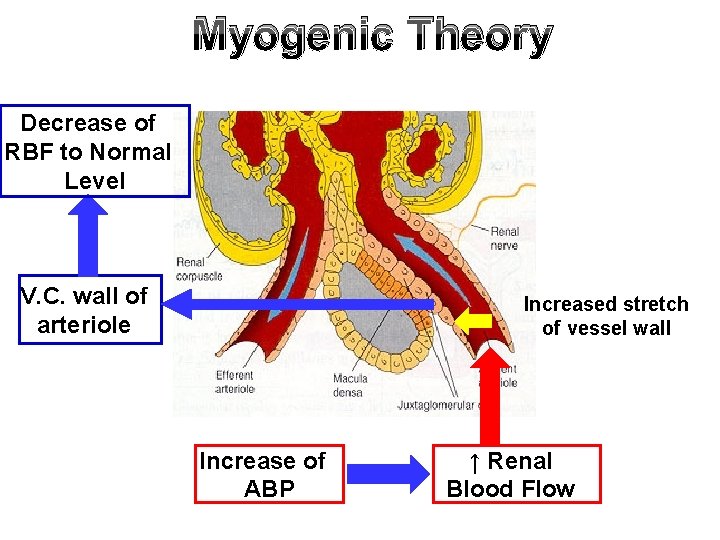
Myogenic Theory Decrease of RBF to Normal Level V. C. wall of arteriole Increased stretch of vessel wall Increase of ABP ↑ Renal Blood Flow

Extrinsic Regulation of RBF • The extrinsic regulation of RBF is mediated by nervous and chemical factors 1. Nervous Factors: sympathetic N. S. 2. Humeral Factors: autacoids and hormones A) Angiotensin II (strong V. C. agent) • At low concentration → V. C. of efferent → protect GFR • At high concentration → generalized V. C. → ↓GFR and RBF b) ADH, serotonin, and endothelin → V. C. of renal blood vessels →↓ RBF c) Prostaglandins (PGE 2 & PGI 2), NO, ANP, dopamine and bradykinin → V. D. d) Hormonal changes during pregnancy → ↑ RBF by 50% e) High protein diet → ↑ RBF by 30%

• Angiotensin converting enzyme inhibitors (ACEIs) are a group of drugs that inhibits AII thus dilate the efferent arteriole and reduce GFR. • These drugs also reduce the hyperfiltration occurring in DM, thereby minimizing the occurrence of nephropathy.
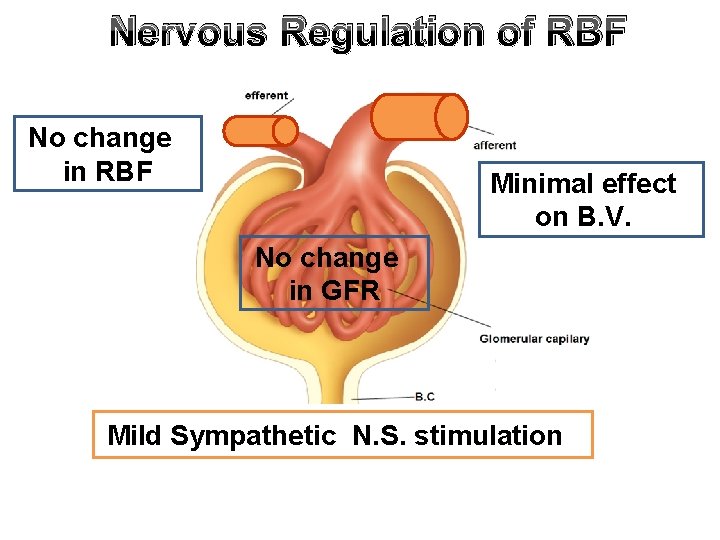
Nervous Regulation of RBF No change in RBF Minimal effect on B. V. No change in GFR Mild Sympathetic N. S. stimulation
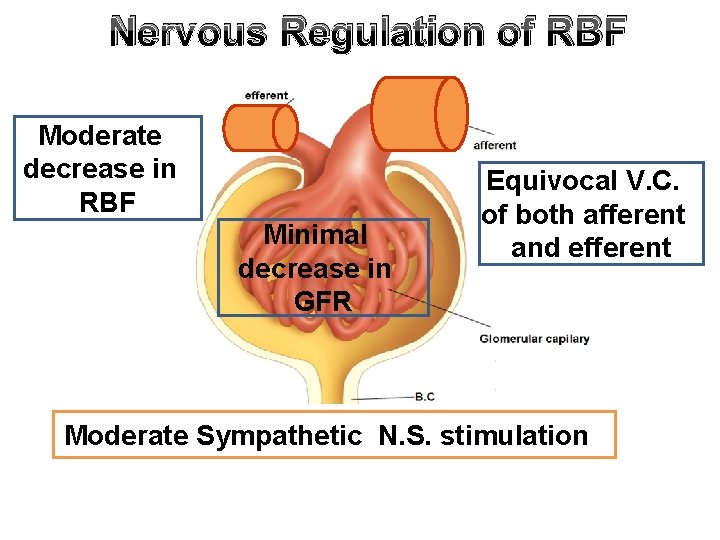
Nervous Regulation of RBF Moderate decrease in RBF Minimal decrease in GFR Equivocal V. C. of both afferent and efferent Moderate Sympathetic N. S. stimulation
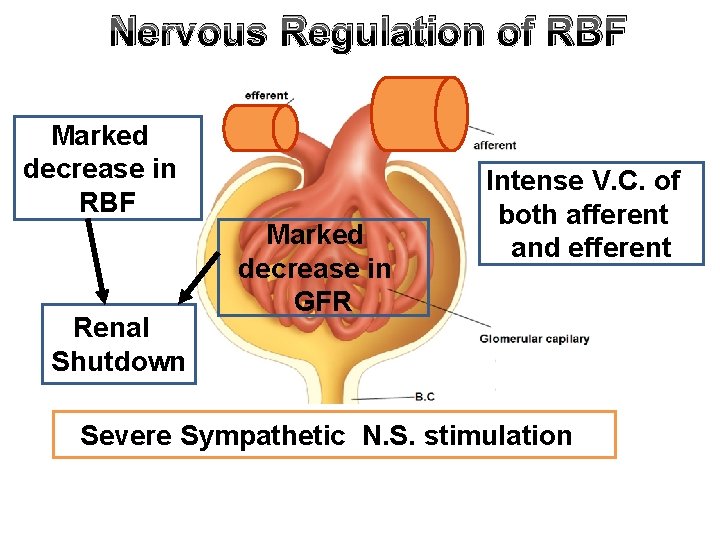
Nervous Regulation of RBF Marked decrease in RBF Renal Shutdown Marked decrease in GFR Intense V. C. of both afferent and efferent Severe Sympathetic N. S. stimulation

Given these structures: • 1. major calyx • 2. minor calyx • 3. renal papilla • 4. renal pelvis • Choose the arrangement that lists the structures in order as urine leaves the collecting duct and travels to the ureter. • • • a. 1, 4, 2, 3 b. 2, 3, 1, 4 c. 3, 2, 1, 4 d. 4, 1, 3, 2 e. 4, 3, 2, 1

Which of these structures contains blood? • a. glomerulus • b. vasa recta • c. distal tubule • d. Bowman’s capsule • e. both a and b

• The juxtaglomerular cells of the …………. and the macula densa cells of the………… form the juxtaglomerular apparatus. • • a. afferent arteriole, proximal tubule b. afferent arteriole, distal tubule c. efferent arteriole, proximal tubule d. efferent arteriole, distal tubule

Given these blood vessels: • 1. afferent arteriole • 2. efferent arteriole • 3. glomerulus • 4. peritubular capillaries Choose the correct order as blood passes from an interlobular artery to an interlobular vein. • a. 1, 2, 3, 4 • b. 1, 3, 2, 4 • c. 2, 1, 4, 3 • d. 3, 2, 4, 1 • e. 4, 3, 1, 2

Urine Formation
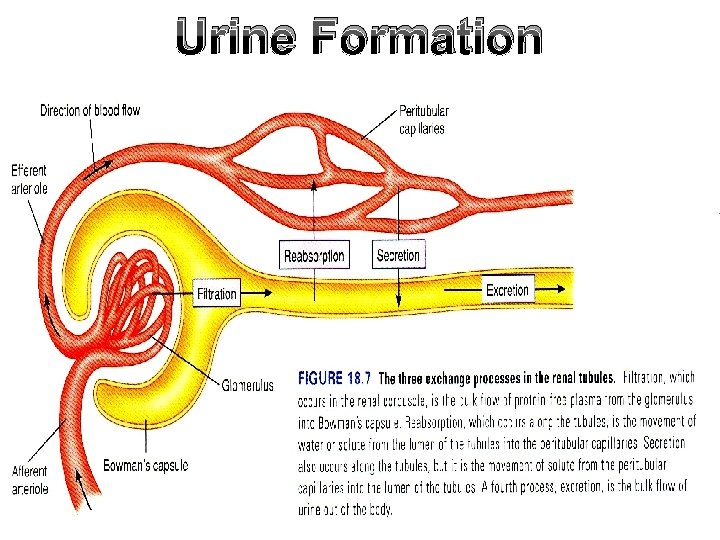
Urine Formation
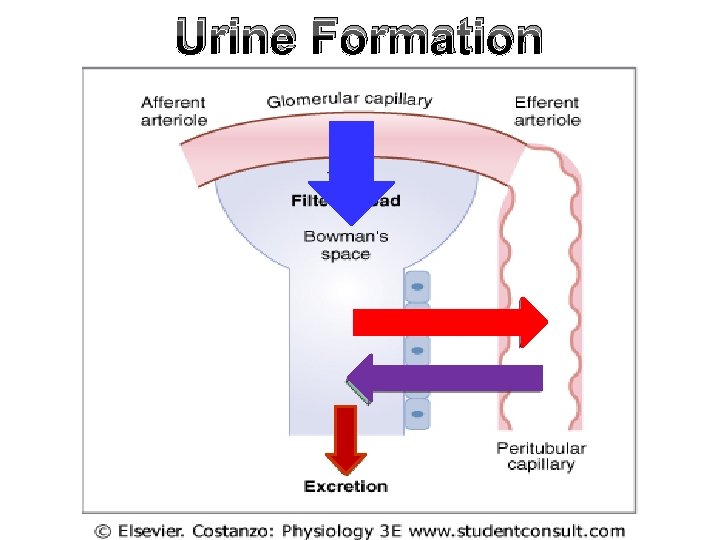
Urine Formation

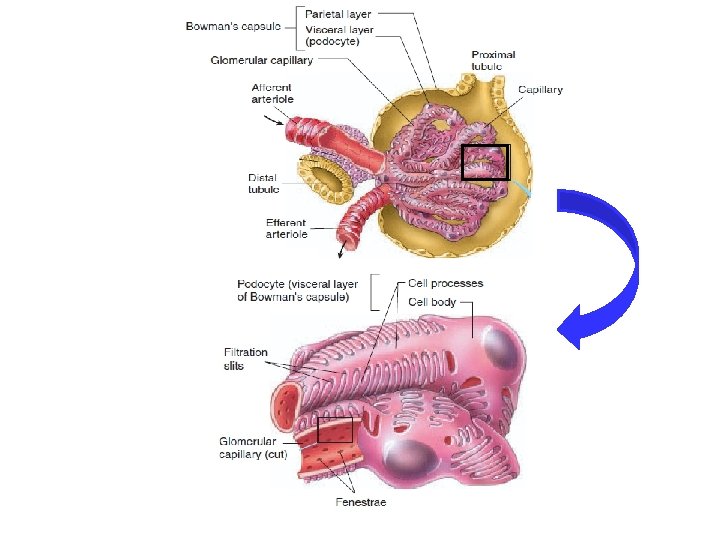
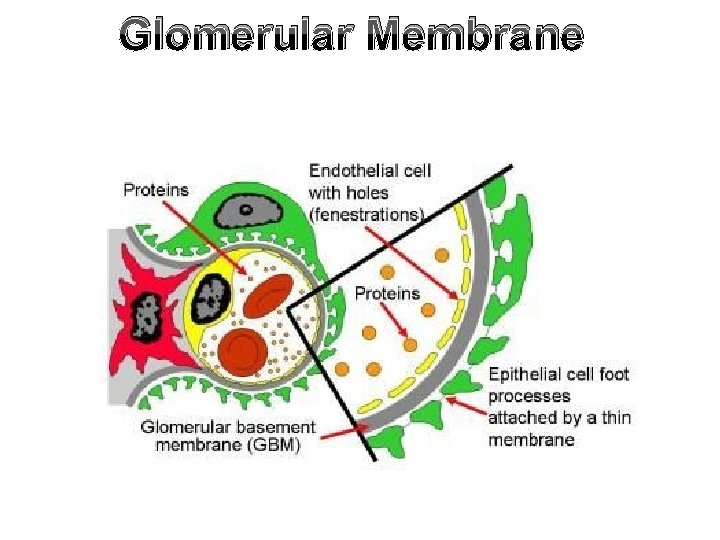
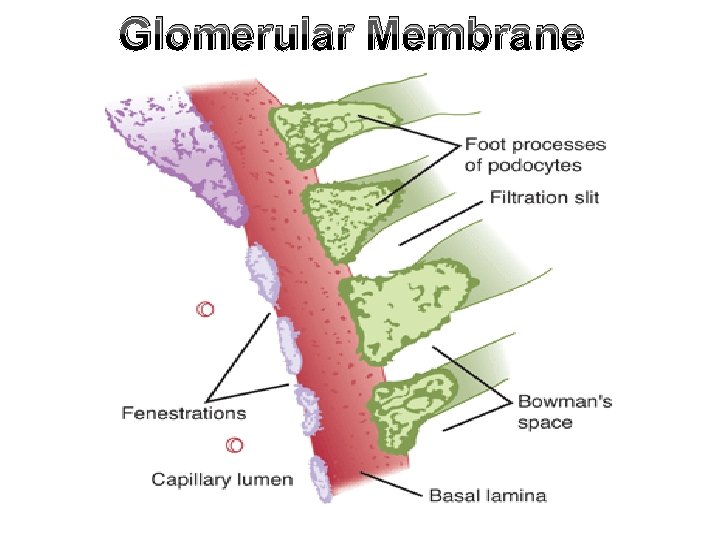
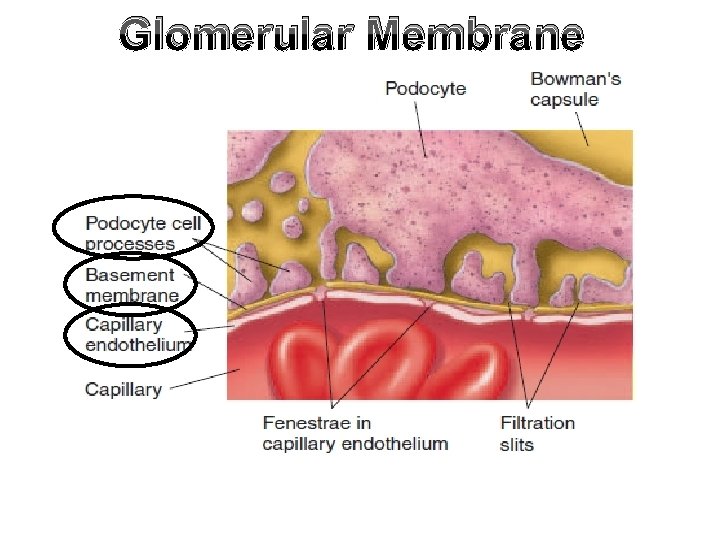
Glomerular Membrane

Glomerular Membrane Def. , • It the membrane through which the plasma is filtered Composition: 3 layers; 1. Capillary endothelium 2. Basement membrane 3. Capsular epithelium
Glomerular Membrane
Glomerular Membrane
Glomerular Membrane
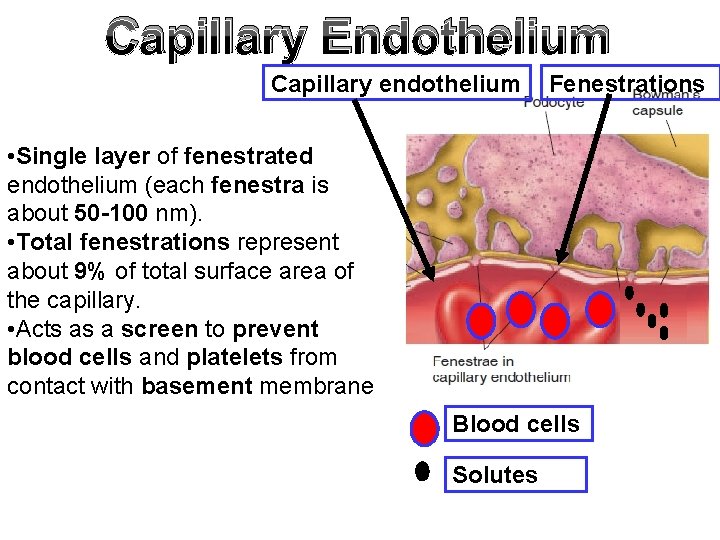
Capillary Endothelium Capillary endothelium Fenestrations • Single layer of fenestrated endothelium (each fenestra is about 50 -100 nm). • Total fenestrations represent about 9% of total surface area of the capillary. • Acts as a screen to prevent blood cells and platelets from contact with basement membrane Blood cells Solutes
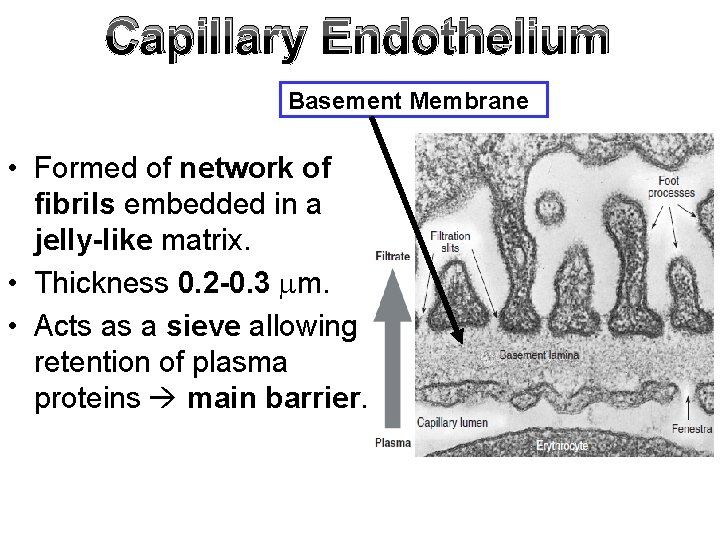
Capillary Endothelium Basement Membrane • Formed of network of fibrils embedded in a jelly-like matrix. • Thickness 0. 2 -0. 3 m. • Acts as a sieve allowing retention of plasma proteins main barrier.
cells PP others Blood endothelium Glom. Memb. Bowman space Basement m. Cap. epith.
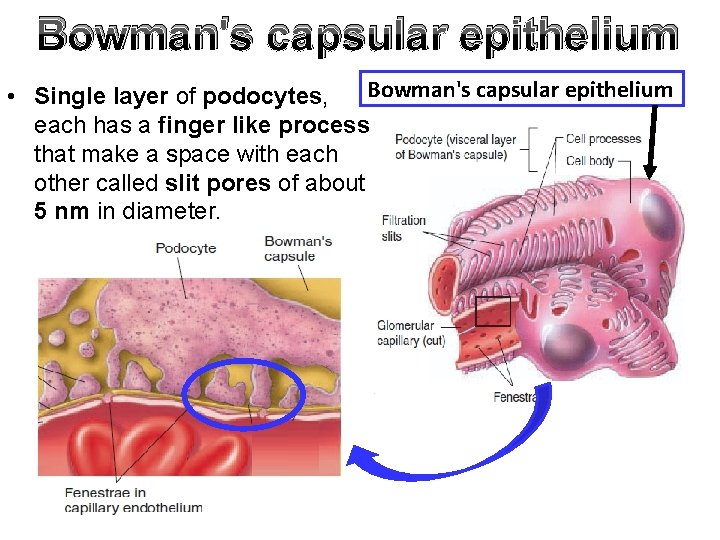
Bowman's capsular epithelium • Single layer of podocytes, each has a finger like process that make a space with each other called slit pores of about 5 nm in diameter.
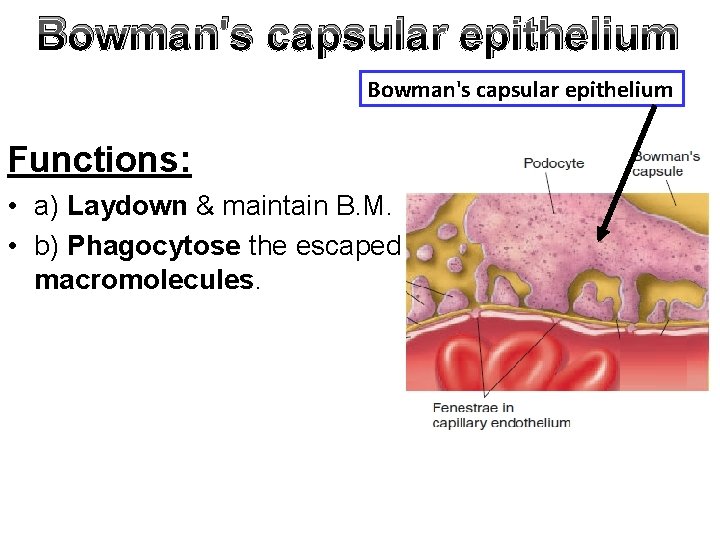
Bowman's capsular epithelium Functions: • a) Laydown & maintain B. M. • b) Phagocytose the escaped macromolecules.

Factors Affecting Permeability of Glomerular Membrane

Factors Affecting Permeability of Glomerular Membrane • 3 factors affecting permeability of G. M. A) Molecular weight and size B) Shape and configuration C) Charge

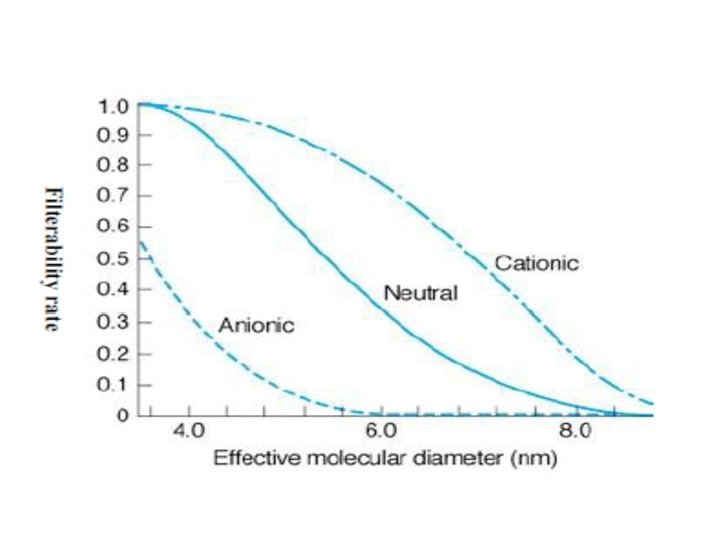
1) Molecular Size and Weight Size: • Radius below 18 A 0 freely filtered. • Radius between 18 - 36 A 0 filtered by rate dependent on the charges; cationic more filtered than anionic of same radius (as the filtering membrane has net negative charge). • Radius more than 36 A 0 not filtered.
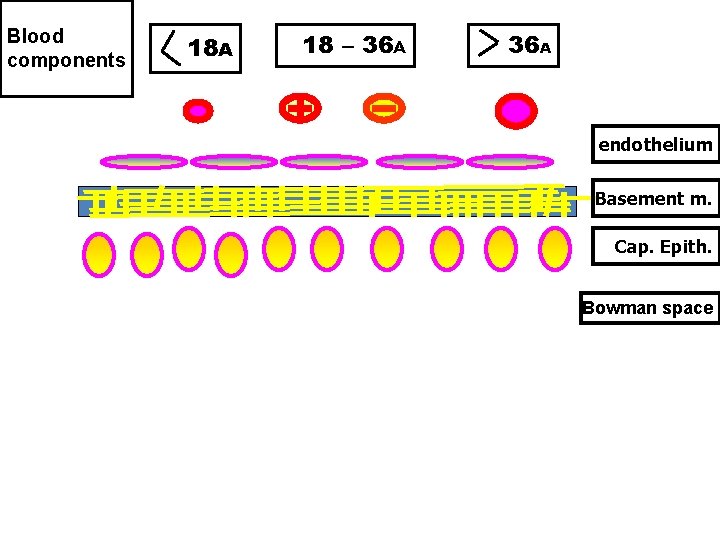
Blood components 18 A 18 – 36 A + 36 A endothelium Basement m. Cap. Epith. Bowman space

1) Molecular Size and Weight: • Molecular weight below 7. 000 freely filtered. • Molecular weight above 70. 000 difficult to pass. • In between 7. 000 – 70. 000 depend on charge & configuration.
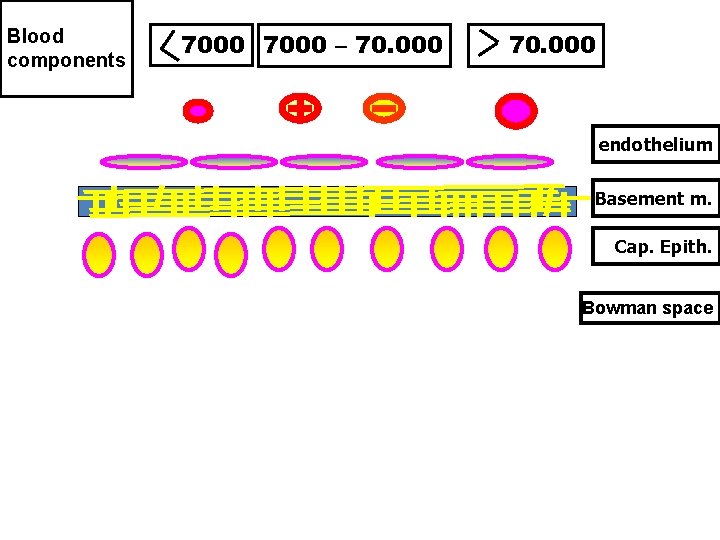
Blood components 7000 – 70. 000 + 70. 000 endothelium Basement m. Cap. Epith. Bowman space

2) Shape and Configuration • Glomerular membrane has a net hole or cylindrical pore of 7. 5 – 10 nm wide • Substances of shape & configuration similar to the holes of the glomerular membrane freely pass & vice versa.
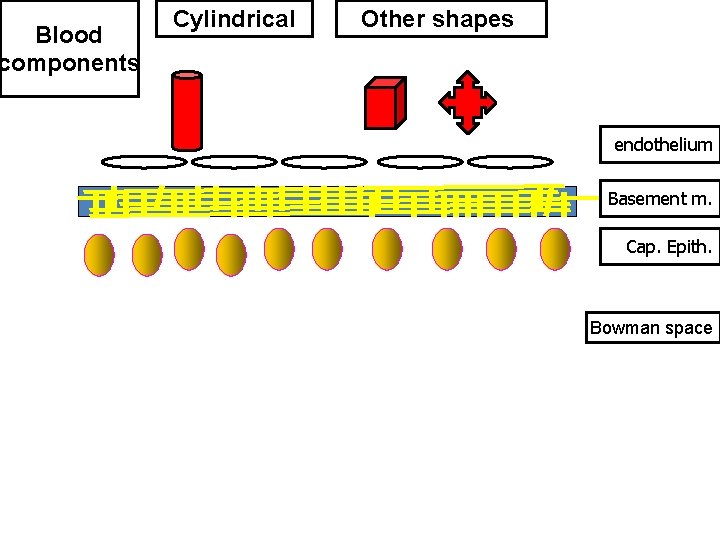
Blood components Cylindrical Other shapes endothelium Basement m. Cap. Epith. Bowman space

3) Charge • Anionic repelled & cationic pass. • Loss of negative charge from glomerular membrane early proteinuria even before any detectable microscopic changes in the membrane.
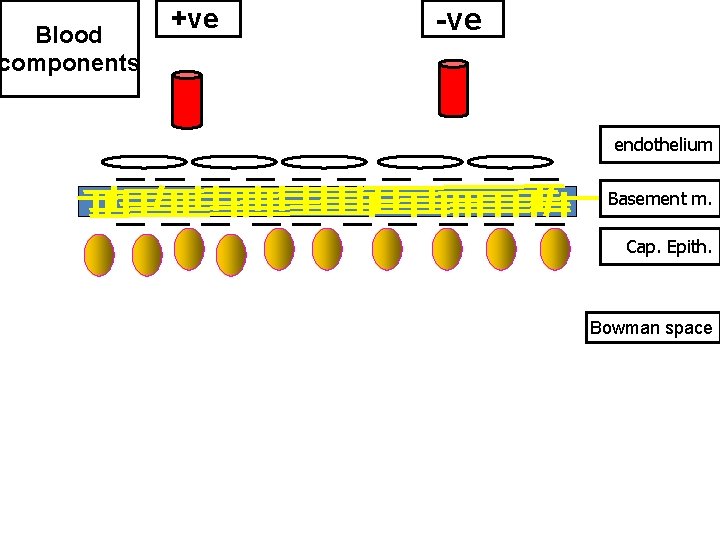
Blood components +ve -ve endothelium Basement m. Cap. Epith. Bowman space
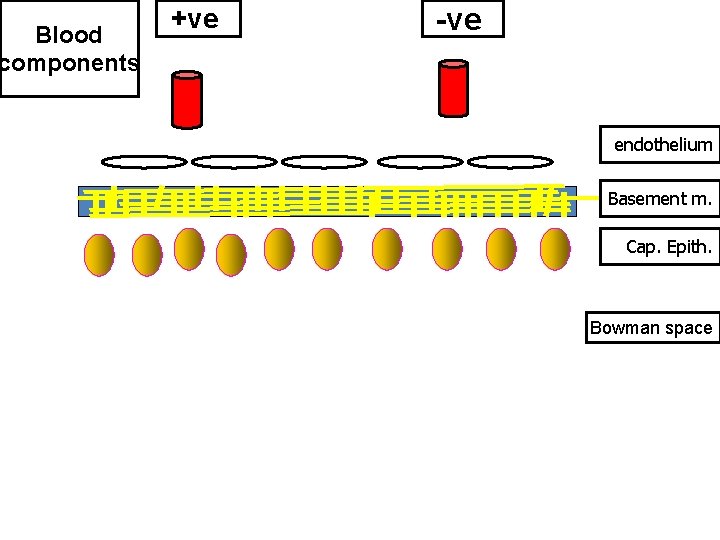
Blood components +ve -ve endothelium Basement m. Cap. Epith. Bowman space

Intraglomerular Mesangial Cells
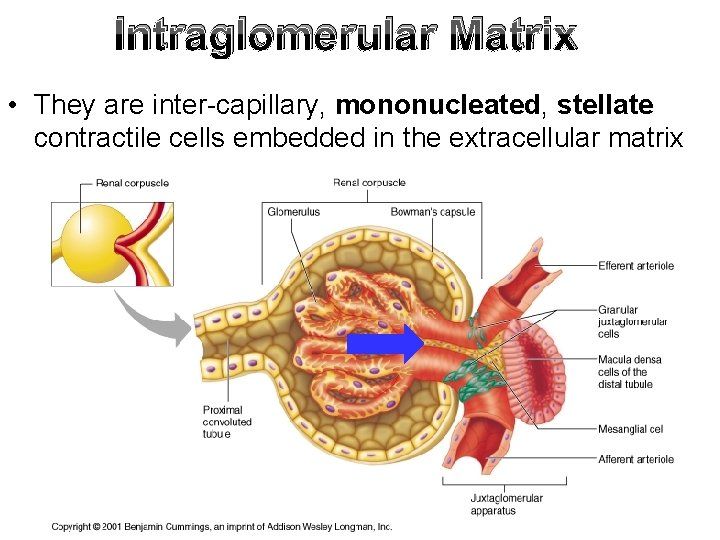
Intraglomerular Matrix • They are inter-capillary, mononucleated, stellate contractile cells embedded in the extracellular matrix

Intraglomerular Matrix 1. Phagocytic for the trapped immunoglobulins 2. Support & hold the delicate glomerular structures in position 3. Control surface area available for filtration by their contraction and relaxation
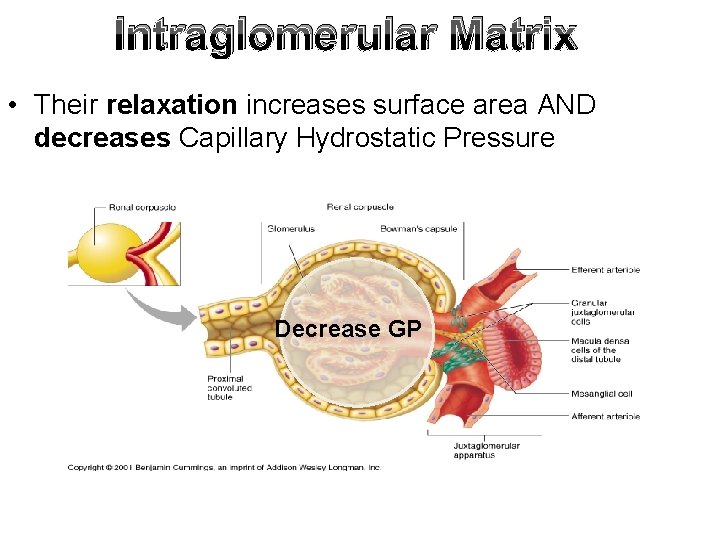
Intraglomerular Matrix • Their relaxation increases surface area AND decreases Capillary Hydrostatic Pressure Decrease GP
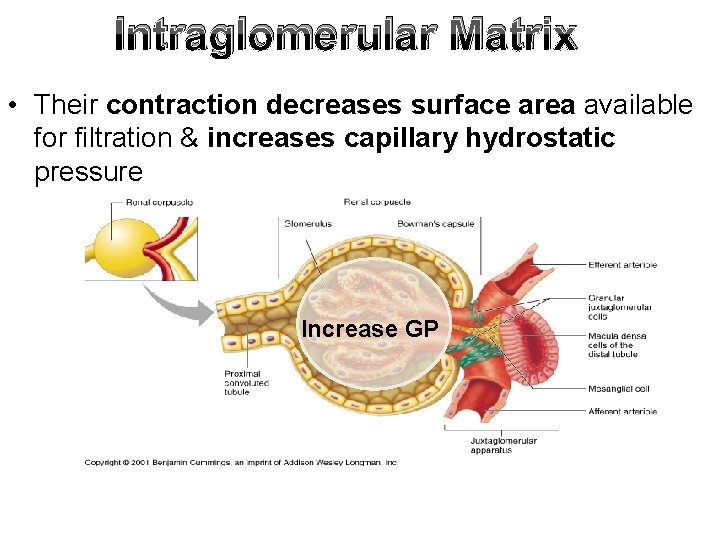
Intraglomerular Matrix • Their contraction decreases surface area available for filtration & increases capillary hydrostatic pressure Increase GP
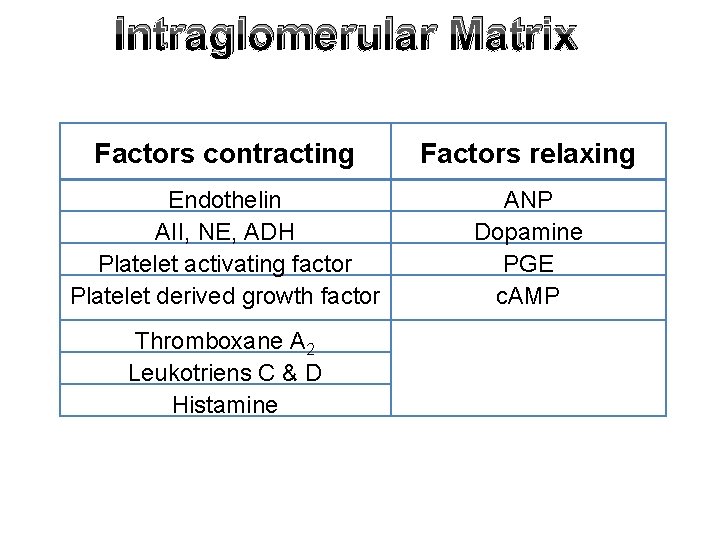
Intraglomerular Matrix Factors contracting Factors relaxing Endothelin AII, NE, ADH Platelet activating factor Platelet derived growth factor ANP Dopamine PGE c. AMP Thromboxane A 2 Leukotriens C & D Histamine

THANKS
- Slides: 62