Kharkiv National Medical University Medical and Bioorganic Chemistry

Kharkiv National Medical University Medical and Bioorganic Chemistry Department Lecture 10 Physico-Chemical Properties of Biopolymers Solutions

High-molecular compounds (HMC) – are compounds with the molecular mass Mr (HMC) 104, whose macromolecules are composed of a large number of repeating monomeric units or molecular aggregates. HMC Natural Synthetic Artificial

• Natural HMC are divided into: • - inorganic, (silicon oxide [Si. O 2]n, aluminium oxide [Al 2 O 3]n); • -organic, polynucleic acids, polysaccharides (glycogen), enzymes, etc. • -mixed : these are proteins containing simultaneously carbohydrate, lipidic and inorganic components. • Synthetic HMC are polymers obtained by synthesis from low-molecular compounds (polyethylene, polypropylene, teflon, kapron etc. ) • Artificial HMC are high-molecular compounds formed as a result of chemical reactions with participation of natural high polymer compounds.

• Macromolecules of polymers are divided according to their spatial structure into: • Linear polymers are a certain sequence of units connected between themselves by covalent bonds (rubber, gelatin, cellulose). Branched polymers consist of units having lateral branches (starch, glycogen). • Cross-linked polymers are three-dimensional polymers whose units form a uniform chemically bonded spatial grid (proteins, enzymes).

Macromolecules of polymers are divided according to their chemical composition into: Homopolymer is a polymer formed from one type of monomers, e. g. starch, cellulose, glycogen (glucose). Copolymer (mixed polymer) is polymer formed from two or more different monomers, e. g. proteins, nucleic acids, heteropolysaccharides. • According to the basis of mode of synthesis into: - Addition polymers are formed by direct addition of repeated monomers without the elimination of any byproduct molecules, e. g. polyethylene, polypropylene, synthetic and natural rubber, Plexiglass, PVC, Teflon. - Condensation polymers are formed by the condensation of two or more than two monomers with the elimination of simple molecules like water, ammonia, HCl, etc. e. g. proteins,
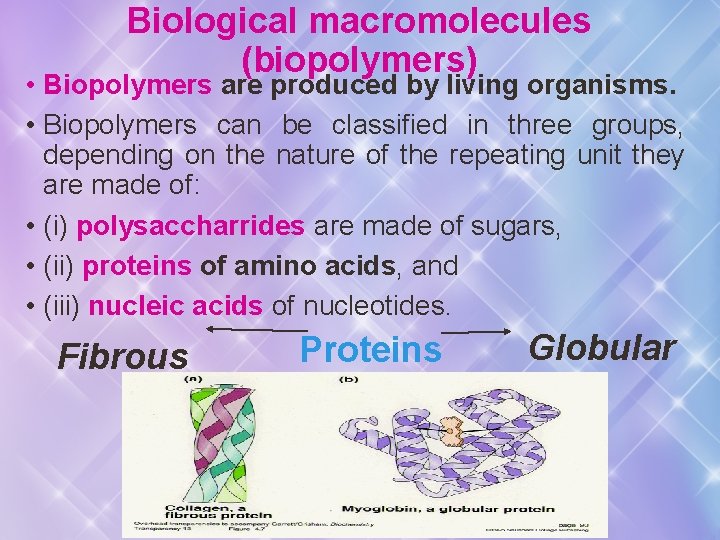
Biological macromolecules (biopolymers) • Biopolymers are produced by living organisms. • Biopolymers can be classified in three groups, depending on the nature of the repeating unit they are made of: • (i) polysaccharrides are made of sugars, • (ii) proteins of amino acids, and • (iii) nucleic acids of nucleotides. Fibrous Proteins Globular
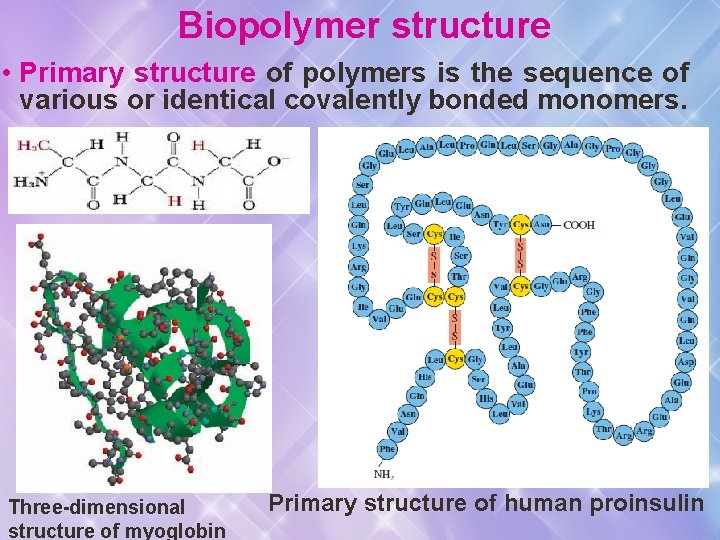
Biopolymer structure • Primary structure of polymers is the sequence of various or identical covalently bonded monomers. Three-dimensional structure of myoglobin Primary structure of human proinsulin
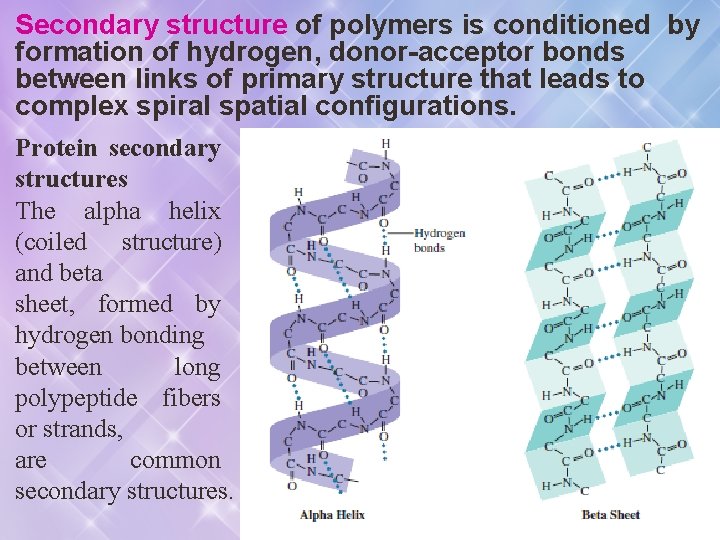
Secondary structure of polymers is conditioned by formation of hydrogen, donor-acceptor bonds between links of primary structure that leads to complex spiral spatial configurations. Protein secondary structures The alpha helix (coiled structure) and beta sheet, formed by hydrogen bonding between long polypeptide fibers or strands, are common secondary structures.
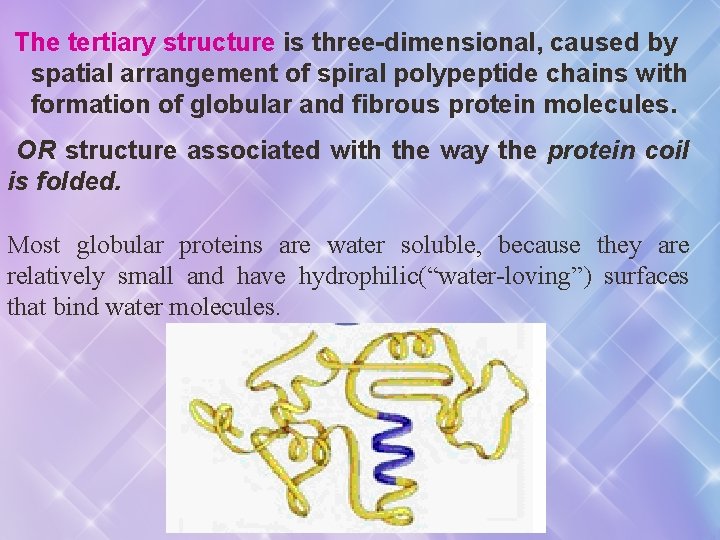
The tertiary structure is three-dimensional, caused by spatial arrangement of spiral polypeptide chains with formation of globular and fibrous protein molecules. OR structure associated with the way the protein coil is folded. Most globular proteins are water soluble, because they are relatively small and have hydrophilic(“water-loving”) surfaces that bind water molecules.
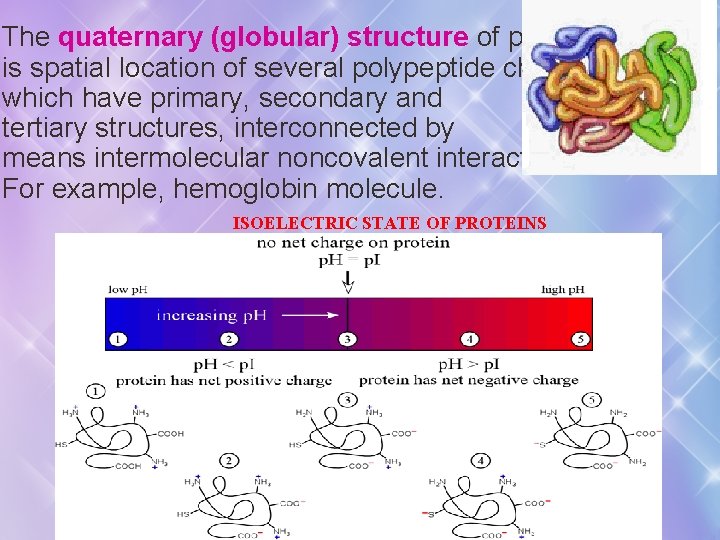
The quaternary (globular) structure of protein is spatial location of several polypeptide chains which have primary, secondary and tertiary structures, interconnected by means intermolecular noncovalent interactions. For example, hemoglobin molecule. ISOELECTRIC STATE OF PROTEINS

Methods of isoelectric point determination 1. Electrophoresis. Electrophoretic mobility is researched. • 2. Salting-out. The coagulation degree is defined. • 3. Jellification. The rate of jelly formation is examined. The rate of jellification is the highest in a solution with p. H approximated to its isoelectric point. • 4. Swelling. The degree of swelling is

HMC solutions and their properties • HMC solutions are homogeneous, thermodynamically stable solutions, although approaching typical colloidal solutions by their physicochemical properties. • HMC solutions and colloidal solutions have a number of common features: • - dissolved molecules of HMC do not pass through membranes; • - precipitate by centrifugation; • - are good light diffusers, especially branched and spatial-volumetric HMC; • - electrophoresis is observed in solutions of HMC electrolytes;

In contrast to colloidal solutions, HMC solutions have: • a considerable constant concentration of a dissolved substance; • higher osmotic pressure in comparison with sols under isomolarity conditions; • abnormally high viscosity, conditioned by the length of HMC molecules and their spatial structure; • high solubility in a disperse medium; • macromolecular nature of a disperse phase. • HMC solutions differ from LMC solutions in: • dissolution mechanism; • lower osmotic pressure (under isomolarity conditions); • abnormally high viscosity; • formation of jellies under certain conditions; • formation of new phase with rise in concentration and decrease in temperature due to ability of macromolecules aggregation into steady associates;

Polymer Swelling is a processdissolution of solvent molecules penetration into a polymeric structure that is accompanied by an increase of the polymer sample volume. The quantitative characteristic of swelling is the swelling degree: m 0 - mass of the polymer sample before swelling; m - mass of. Limited the polymer sample after swelling. Unlimited Swelling at which the polymer First the polymer absorbs a certain amount absorbs liquid ant of the solvent and does then forms with it a not pass into the homogeneoussystem. dissolution stage. At It results in the

Swelling depends on: • Nature of polymer and solvent: HMC, containing polar functional groups (-OH, -NH 2, -COOH) swell in polar solvent (H 2 O), hydrocarbon polymers, such as rubber, swell better in nonpolar solvent, for example, in benzene, toluene. • Temperature : with rise in temperature swelling also increases, because diffusion grows, which intensifies loosening of structure. • p. H : Swelling occurs at the isoelectric point is minimal , which is conditioned by minimal solvation of ionogenic groups. • Presence of electrolytes. The swelling of proteins is maximal in the presence of non-hydrated ions ->Cl. O ->SO 2 CNS->I- >Br 4 4 and minimal in the+ presence of 2+highly hydrated + + 2+ 2+ 2+ Cs >Rb >K >Na >Li >Ba >Sr >Ca >Mg ions. This phenomenon is explained by the Maximal swelling Minimal swelling
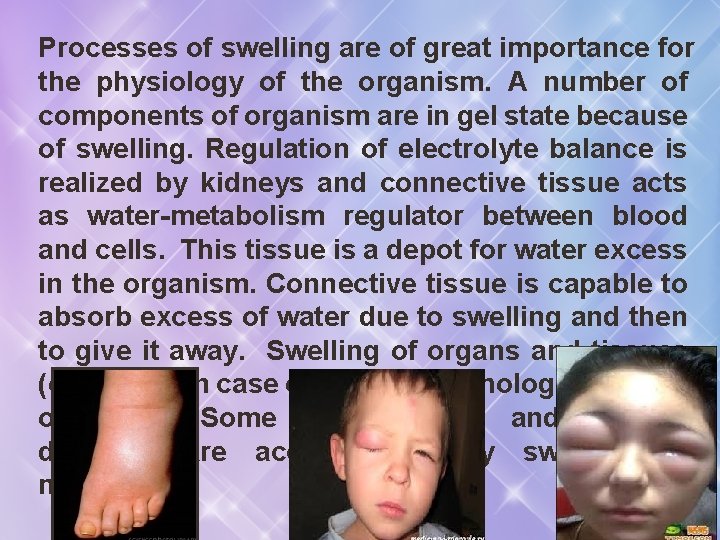
Processes of swelling are of great importance for the physiology of the organism. A number of components of organism are in gel state because of swelling. Regulation of electrolyte balance is realized by kidneys and connective tissue acts as water-metabolism regulator between blood and cells. This tissue is a depot for water excess in the organism. Connective tissue is capable to absorb excess of water due to swelling and then to give it away. Swelling of organs and tissues (oedemas) in case of different pathologies can be observed. Some inflammatory and allergic diseases are accompanied by swelling of mucosas.

Thermodynamic stability of HMC solutions. • High hydrophility of biopolymers is the main factor of thermodynamic stability of their solutions. Carboxylic groups (-COOH), amino groups (-NH 2), peptide bonds and other polar groups are hydrated. • Proteins can be isolated from solution by means of salts. This process is known as salting out. Na 2 SO 4, (NH 4)2 SO 4, magnesium salts and phosphates are usually used for this purpose. Salting out is most effective in the isoelectric point of protein. • • The process of protein salting-out is influenced by the presence of both anions and cations. It is possible to arrange them in the corresponding lyotropic series: • Li+ Na+ K+ Cs+; Mg 2+ Ca 2+ Ba 2+ Sr 2+
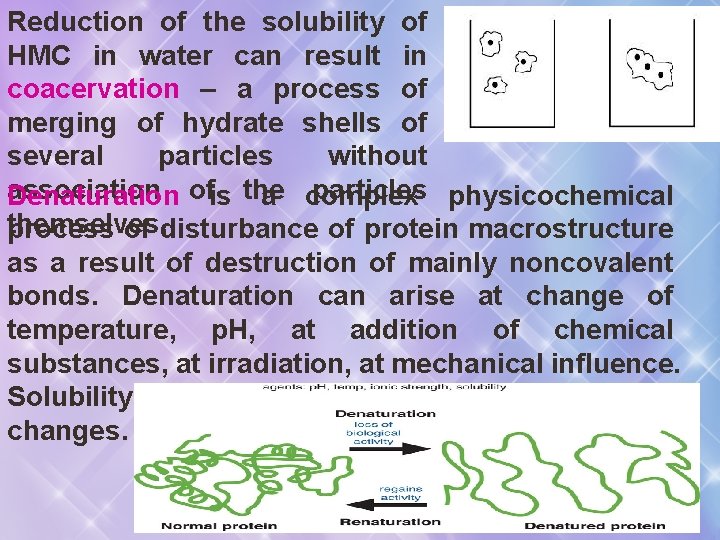
Reduction of the solubility of HMC in water can result in coacervation – a process of merging of hydrate shells of several particles without association particles physicochemical Denaturation ofis the a complex themselves. process of disturbance of protein macrostructure as a result of destruction of mainly noncovalent bonds. Denaturation can arise at change of temperature, p. H, at addition of chemical substances, at irradiation, at mechanical influence. Solubility and biological activity of proteins changes.

Viscosity of HMC solutions Unlike true and colloidal solutions HMC solutions possess a high viscosity. High viscosity of HMC solutions can be explained by hydrophilic nature of HMC, i. e. strong intermolecular forces exist between polymers and water molecules. Viscosity depends on: 1)Molar mass 2)Concentration. of HMC macromolecules. 3) Form of macromolecules. Physiological liquids are not Newtonian liquids, their viscosity depends on: 1) Size and form of particles; 2) Concentration; 3) р. Н (at the isoelectric point solutions of proteins have the least viscosity; that is conditioned by their structure and electroneutrality);

• Jellies are nonfluid structured systems formed as a result of intermolecular interacton of macromolecules of polymers, whose spatial grid cells are filled with disperse medium molecules. • According to the origin jellies are distinguished: • -natural – the cytoplasm of living cells, skin, eye crystalline lens. • -artificial – gelatin, rubber, agar-agar, synthetic rubber, cellulose ethers. • Jellification is distinctive coagulation, at which the disperse phase together with the disperse medium form structured systems. • Jellies can be prepared by two ways: • - gelatinization of HMC solutions;

1) The process of jellies destruction with their subsequent structurization is called thixotropy. This is name is applied to a reversible demolition of a structure with the transition to the fluid state upon mechanical action, for example, under shaking, and spontaneous restoration of the structure. For example, this process is observed in case of brain concussion and restoration of its initial structures. 2) Division of jellies into two phases with liquid escape is called syneresis. Gels loose homogeneity with time. Syneresis

Thank you for your attention!
- Slides: 22