KEYNOTE355 Pembrolizumab Chemotherapy for Previously Untreated Advanced TripleNegative

KEYNOTE-355: Pembrolizumab + Chemotherapy for Previously Untreated Advanced Triple-Negative Breast Cancer CCO Independent Conference Highlights* of the 2020 ASCO Virtual Scientific Program, May 29 - 31, 2020 *CCO is an independent medical education company that provides state-of-the-art medical information to healthcare professionals through conference coverage and other educational programs. This activity is provided by Clinical Care Options, LLC Supported by educational grants from Glaxo. Smith. Kline LLC, Merck Sharp & Dohme Corp. , and Novartis Pharmaceuticals Corporation
About These Slides § Please feel free to use, update, and share some or all of these slides in your noncommercial presentations to colleagues or patients § When using our slides, please retain the source attribution: Slide credit: clinicaloptions. com § These slides may not be published, posted online, or used in commercial presentations without permission. Please contact permissions@clinicaloptions. com for details

KEYNOTE-355: Background § Pembrolizumab monotherapy has demonstrated efficacy in patients with m. TNBC with a manageable toxicity profile[1 -4] ‒ Antitumor activity observed in treatment-naive and previously treated patients; responses more durable than with chemotherapy § Pembrolizumab + standard chemotherapy in the neoadjuvant setting improved p. CR rates with no unexpected safety signals[5 -7] ‒ US FDA Breakthrough Therapy Designation for high-risk, early-stage TNBC § KEYNOTE-355 explores the addition of pembrolizumab to chemotherapy in patients with previously untreated locally recurrent inoperable TNBC or m. TNBC[8] 1. Nanda. JCO. 2016; 34: 2460. 2. Adams. Ann Oncol. 2019; 30: 397. 3. Adams. Ann Oncol. 2019; 30: 405. 4. Cortes. ESMO 2019. Abstr XXXX. 5. Schmid. Ann Oncol. 2020; 31: 569. 6. Nanda. JAMA Oncol. 2020; [epub ahead of print]. 7. Schmid. NEJM. 2020; 382: 810. 8. Cortes. ASCO 2020. Abstr 1000. Slide credit: clinicaloptions. com
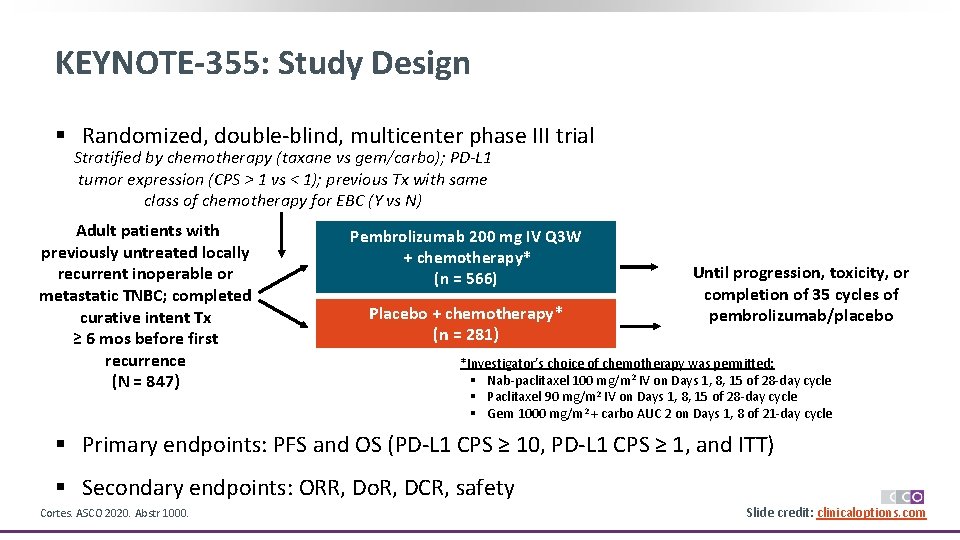
KEYNOTE-355: Study Design § Randomized, double-blind, multicenter phase III trial Stratified by chemotherapy (taxane vs gem/carbo); PD-L 1 tumor expression (CPS > 1 vs < 1); previous Tx with same class of chemotherapy for EBC (Y vs N) Adult patients with previously untreated locally recurrent inoperable or metastatic TNBC; completed curative intent Tx ≥ 6 mos before first recurrence (N = 847) Pembrolizumab 200 mg IV Q 3 W + chemotherapy* (n = 566) Placebo + chemotherapy* (n = 281) Until progression, toxicity, or completion of 35 cycles of pembrolizumab/placebo *Investigator’s choice of chemotherapy was permitted: § Nab-paclitaxel 100 mg/m 2 IV on Days 1, 8, 15 of 28 -day cycle § Paclitaxel 90 mg/m 2 IV on Days 1, 8, 15 of 28 -day cycle § Gem 1000 mg/m 2 + carbo AUC 2 on Days 1, 8 of 21 -day cycle § Primary endpoints: PFS and OS (PD-L 1 CPS ≥ 10, PD-L 1 CPS ≥ 1, and ITT) § Secondary endpoints: ORR, Do. R, DCR, safety Cortes. ASCO 2020. Abstr 1000. Slide credit: clinicaloptions. com
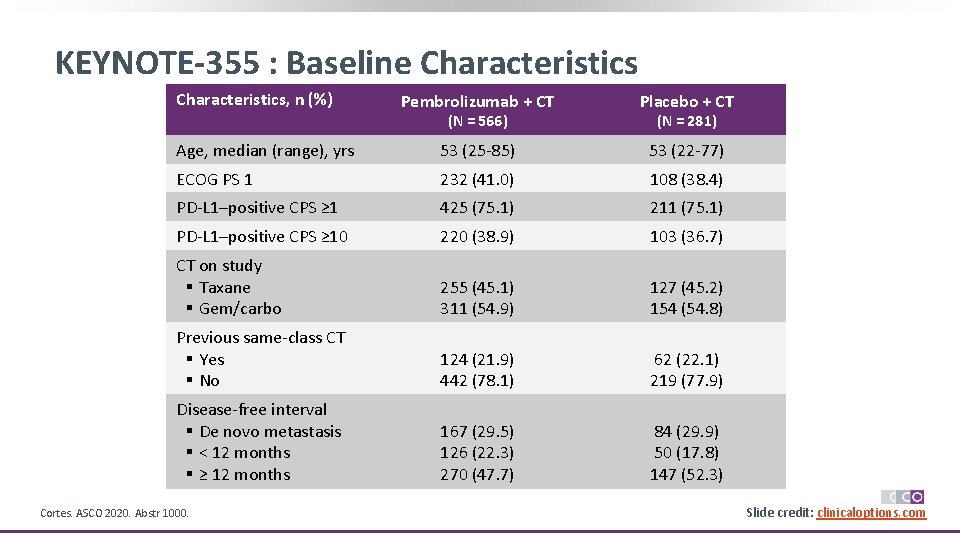
KEYNOTE-355 : Baseline Characteristics, n (%) Pembrolizumab + CT Placebo + CT Age, median (range), yrs 53 (25 -85) 53 (22 -77) ECOG PS 1 232 (41. 0) 108 (38. 4) PD-L 1–positive CPS ≥ 1 425 (75. 1) 211 (75. 1) PD-L 1–positive CPS ≥ 10 220 (38. 9) 103 (36. 7) CT on study § Taxane § Gem/carbo 255 (45. 1) 311 (54. 9) 127 (45. 2) 154 (54. 8) Previous same-class CT § Yes § No 124 (21. 9) 442 (78. 1) 62 (22. 1) 219 (77. 9) Disease-free interval § De novo metastasis § < 12 months § ≥ 12 months 167 (29. 5) 126 (22. 3) 270 (47. 7) 84 (29. 9) 50 (17. 8) 147 (52. 3) (N = 566) Cortes. ASCO 2020. Abstr 1000. (N = 281) Slide credit: clinicaloptions. com
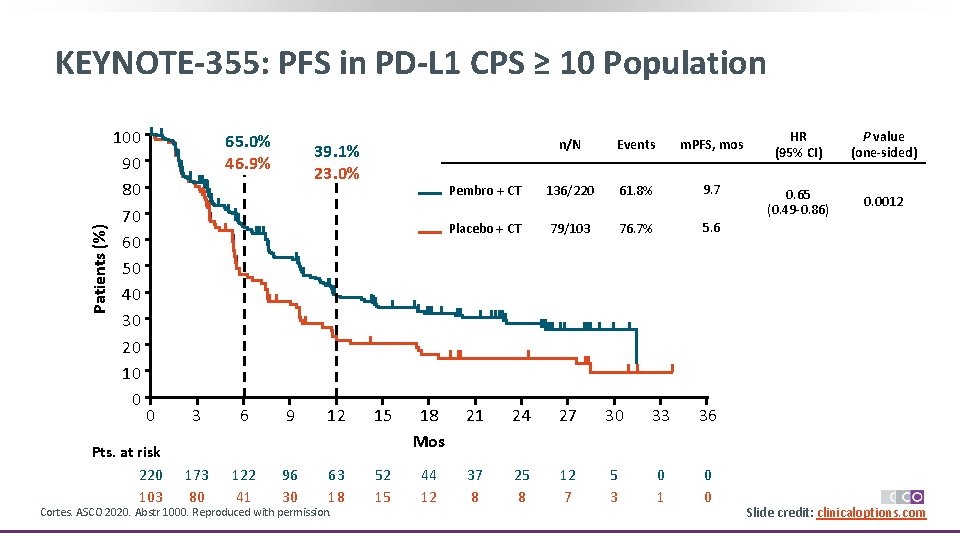
Patients (%) KEYNOTE-355: PFS in PD-L 1 CPS ≥ 10 Population 100 90 80 70 60 50 40 30 20 10 0 65. 0% 46. 9% 0 Pts. at risk 220 103 39. 1% 23. 0% n/N Events m. PFS, mos Pembro + CT 136/220 61. 8% 9. 7 Placebo + CT 79/103 76. 7% 5. 6 3 6 9 12 15 18 Mos 21 24 27 30 33 36 173 80 122 41 96 30 63 18 52 15 44 12 37 8 25 8 12 7 5 3 0 1 0 0 Cortes. ASCO 2020. Abstr 1000. Reproduced with permission. HR (95% CI) P value (one-sided) 0. 65 (0. 49 -0. 86) 0. 0012 Slide credit: clinicaloptions. com
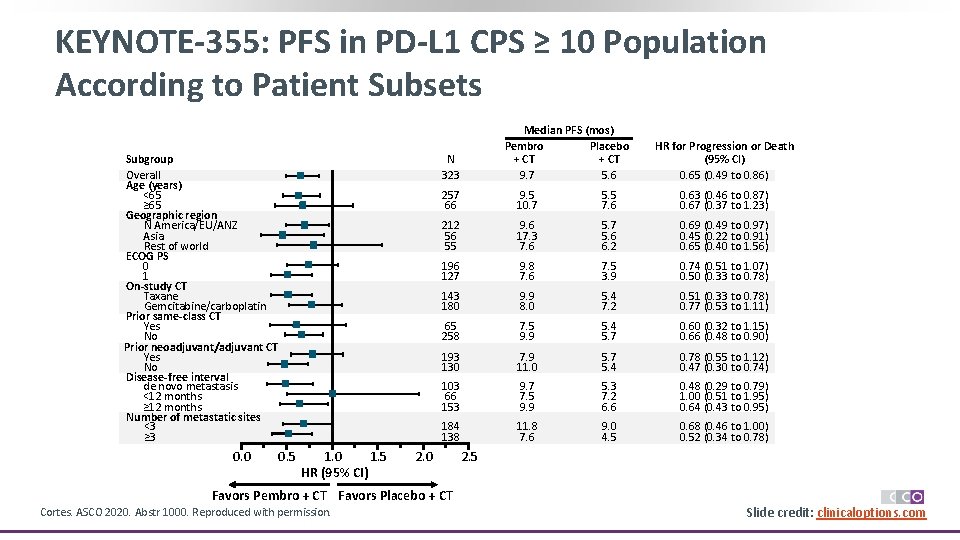
KEYNOTE-355: PFS in PD-L 1 CPS ≥ 10 Population According to Patient Subsets Subgroup Overall Age (years) <65 ≥ 65 Geographic region N America/EU/ANZ Asia Rest of world ECOG PS 0 1 On-study CT Taxane Gemcitabine/carboplatin Prior same-class CT Yes No Prior neoadjuvant/adjuvant CT Yes No Disease-free interval de novo metastasis <12 months ≥ 12 months Number of metastatic sites <3 ≥ 3 0. 0 Median PFS (mos) Pembro Placebo + CT 9. 7 5. 6 N 323 0. 5 1. 0 1. 5 HR (95% CI) HR for Progression or Death (95% CI) 0. 65 (0. 49 to 0. 86) 257 66 9. 5 10. 7 5. 5 7. 6 0. 63 (0. 46 to 0. 87) 0. 67 (0. 37 to 1. 23) 212 56 55 9. 6 17. 3 7. 6 5. 7 5. 6 6. 2 0. 69 (0. 49 to 0. 97) 0. 45 (0. 22 to 0. 91) 0. 65 (0. 40 to 1. 56) 196 127 9. 8 7. 6 7. 5 3. 9 0. 74 (0. 51 to 1. 07) 0. 50 (0. 33 to 0. 78) 143 180 9. 9 8. 0 5. 4 7. 2 0. 51 (0. 33 to 0. 78) 0. 77 (0. 53 to 1. 11) 65 258 7. 5 9. 9 5. 4 5. 7 0. 60 (0. 32 to 1. 15) 0. 66 (0. 48 to 0. 90) 193 130 7. 9 11. 0 5. 7 5. 4 0. 78 (0. 55 to 1. 12) 0. 47 (0. 30 to 0. 74) 103 66 153 9. 7 7. 5 9. 9 5. 3 7. 2 6. 6 0. 48 (0. 29 to 0. 79) 1. 00 (0. 51 to 1. 95) 0. 64 (0. 43 to 0. 95) 184 138 11. 8 7. 6 9. 0 4. 5 0. 68 (0. 46 to 1. 00) 0. 52 (0. 34 to 0. 78) 2. 0 2. 5 Favors Pembro + CT Favors Placebo + CT Cortes. ASCO 2020. Abstr 1000. Reproduced with permission. Slide credit: clinicaloptions. com
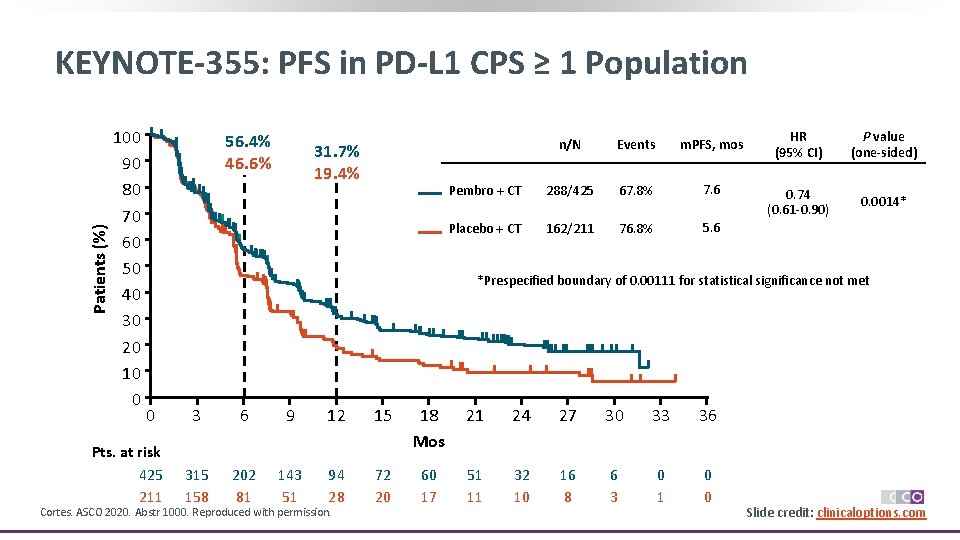
Patients (%) KEYNOTE-355: PFS in PD-L 1 CPS ≥ 1 Population 100 90 80 70 60 50 40 30 20 10 0 56. 4% 46. 6% 31. 7% 19. 4% n/N Events m. PFS, mos Pembro + CT 288/425 67. 8% 7. 6 Placebo + CT 162/211 76. 8% 5. 6 HR (95% CI) P value (one-sided) 0. 74 (0. 61 -0. 90) 0. 0014* *Prespecified boundary of 0. 00111 for statistical significance not met 0 Pts. at risk 425 211 3 6 9 12 15 18 Mos 21 24 27 30 33 36 315 158 202 81 143 51 94 28 72 20 60 17 51 11 32 10 16 8 6 3 0 1 0 0 Cortes. ASCO 2020. Abstr 1000. Reproduced with permission. Slide credit: clinicaloptions. com
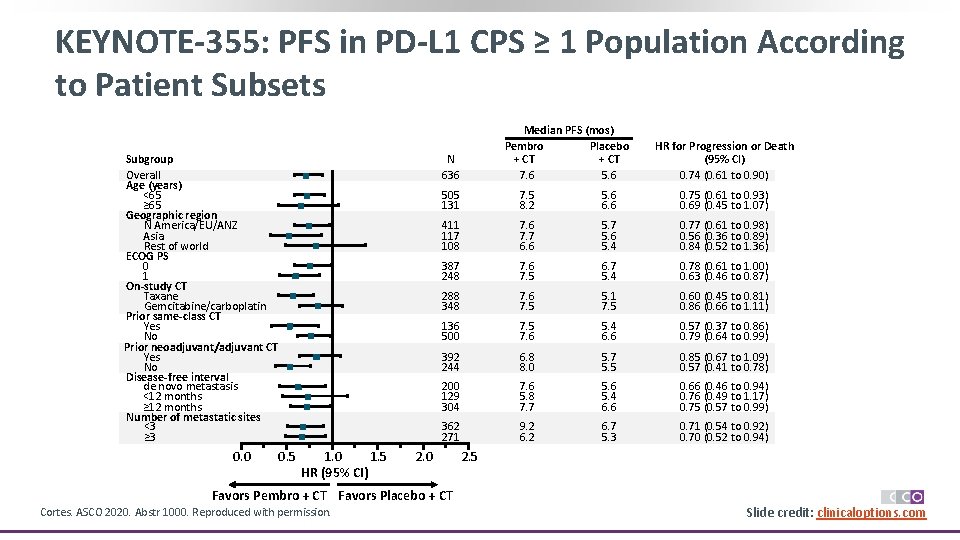
KEYNOTE-355: PFS in PD-L 1 CPS ≥ 1 Population According to Patient Subsets Subgroup Overall Age (years) <65 ≥ 65 Geographic region N America/EU/ANZ Asia Rest of world ECOG PS 0 1 On-study CT Taxane Gemcitabine/carboplatin Prior same-class CT Yes No Prior neoadjuvant/adjuvant CT Yes No Disease-free interval de novo metastasis <12 months ≥ 12 months Number of metastatic sites <3 ≥ 3 0. 0 Median PFS (mos) Pembro Placebo + CT 7. 6 5. 6 N 636 0. 5 1. 0 1. 5 HR (95% CI) HR for Progression or Death (95% CI) 0. 74 (0. 61 to 0. 90) 505 131 7. 5 8. 2 5. 6 6. 6 0. 75 (0. 61 to 0. 93) 0. 69 (0. 45 to 1. 07) 411 117 108 7. 6 7. 7 6. 6 5. 7 5. 6 5. 4 0. 77 (0. 61 to 0. 98) 0. 56 (0. 36 to 0. 89) 0. 84 (0. 52 to 1. 36) 387 248 7. 6 7. 5 6. 7 5. 4 0. 78 (0. 61 to 1. 00) 0. 63 (0. 46 to 0. 87) 288 348 7. 6 7. 5 5. 1 7. 5 0. 60 (0. 45 to 0. 81) 0. 86 (0. 66 to 1. 11) 136 500 7. 5 7. 6 5. 4 6. 6 0. 57 (0. 37 to 0. 86) 0. 79 (0. 64 to 0. 99) 392 244 6. 8 8. 0 5. 7 5. 5 0. 85 (0. 67 to 1. 09) 0. 57 (0. 41 to 0. 78) 200 129 304 7. 6 5. 8 7. 7 5. 6 5. 4 6. 6 0. 66 (0. 46 to 0. 94) 0. 76 (0. 49 to 1. 17) 0. 75 (0. 57 to 0. 99) 362 271 9. 2 6. 7 5. 3 0. 71 (0. 54 to 0. 92) 0. 70 (0. 52 to 0. 94) 2. 0 2. 5 Favors Pembro + CT Favors Placebo + CT Cortes. ASCO 2020. Abstr 1000. Reproduced with permission. Slide credit: clinicaloptions. com
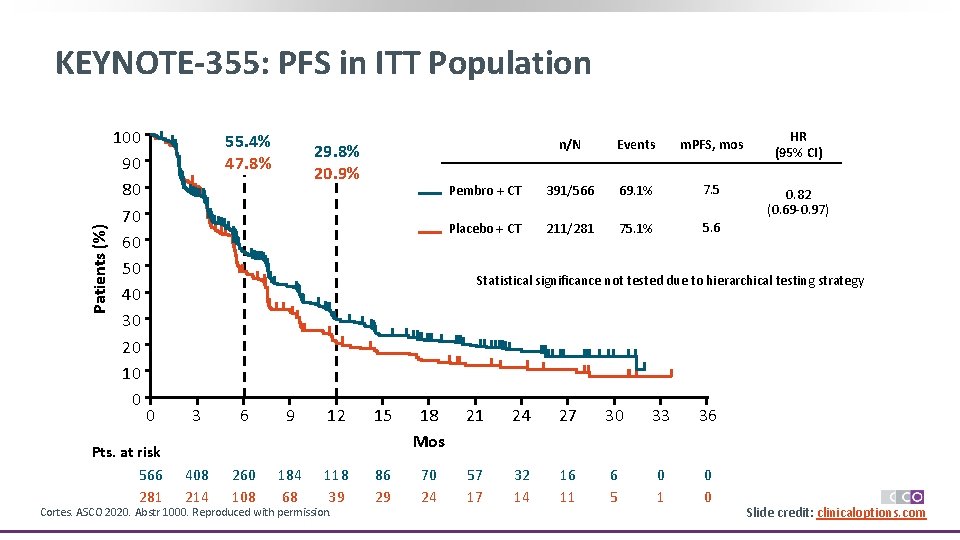
Patients (%) KEYNOTE-355: PFS in ITT Population 100 90 80 70 60 50 40 30 20 10 0 55. 4% 47. 8% 29. 8% 20. 9% n/N Events m. PFS, mos Pembro + CT 391/566 69. 1% 7. 5 Placebo + CT 211/281 75. 1% 5. 6 HR (95% CI) 0. 82 (0. 69 -0. 97) Statistical significance not tested due to hierarchical testing strategy 0 Pts. at risk 566 281 3 6 9 12 15 18 Mos 21 24 27 30 33 36 408 214 260 108 184 68 118 39 86 29 70 24 57 17 32 14 16 11 6 5 0 1 0 0 Cortes. ASCO 2020. Abstr 1000. Reproduced with permission. Slide credit: clinicaloptions. com
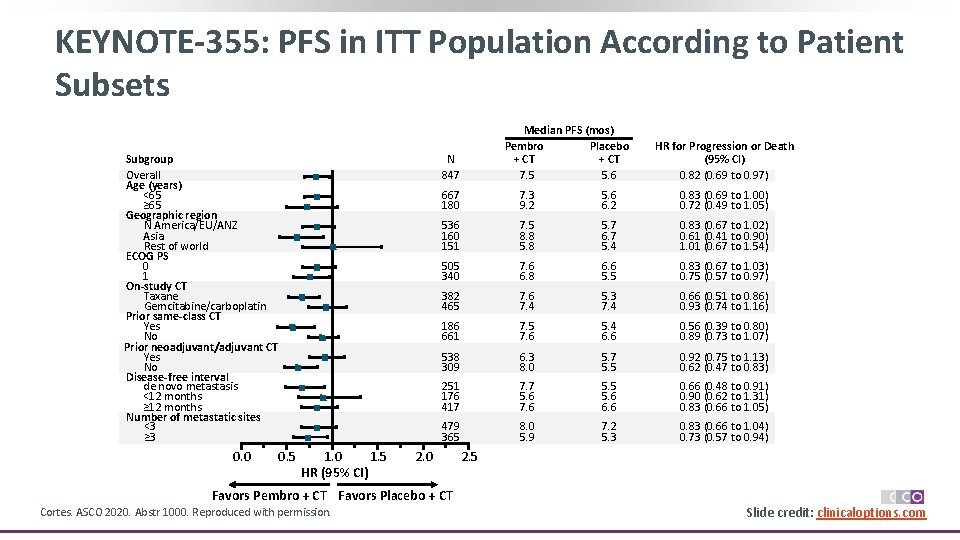
KEYNOTE-355: PFS in ITT Population According to Patient Subsets Subgroup Overall Age (years) <65 ≥ 65 Geographic region N America/EU/ANZ Asia Rest of world ECOG PS 0 1 On-study CT Taxane Gemcitabine/carboplatin Prior same-class CT Yes No Prior neoadjuvant/adjuvant CT Yes No Disease-free interval de novo metastasis <12 months ≥ 12 months Number of metastatic sites <3 ≥ 3 0. 0 Median PFS (mos) Pembro Placebo + CT 7. 5 5. 6 N 847 0. 5 1. 0 1. 5 HR (95% CI) HR for Progression or Death (95% CI) 0. 82 (0. 69 to 0. 97) 667 180 7. 3 9. 2 5. 6 6. 2 0. 83 (0. 69 to 1. 00) 0. 72 (0. 49 to 1. 05) 536 160 151 7. 5 8. 8 5. 7 6. 7 5. 4 0. 83 (0. 67 to 1. 02) 0. 61 (0. 41 to 0. 90) 1. 01 (0. 67 to 1. 54) 505 340 7. 6 6. 8 6. 6 5. 5 0. 83 (0. 67 to 1. 03) 0. 75 (0. 57 to 0. 97) 382 465 7. 6 7. 4 5. 3 7. 4 0. 66 (0. 51 to 0. 86) 0. 93 (0. 74 to 1. 16) 186 661 7. 5 7. 6 5. 4 6. 6 0. 56 (0. 39 to 0. 80) 0. 89 (0. 73 to 1. 07) 538 309 6. 3 8. 0 5. 7 5. 5 0. 92 (0. 75 to 1. 13) 0. 62 (0. 47 to 0. 83) 251 176 417 7. 7 5. 6 7. 6 5. 5 5. 6 6. 6 0. 66 (0. 48 to 0. 91) 0. 90 (0. 62 to 1. 31) 0. 83 (0. 66 to 1. 05) 479 365 8. 0 5. 9 7. 2 5. 3 0. 83 (0. 66 to 1. 04) 0. 73 (0. 57 to 0. 94) 2. 0 2. 5 Favors Pembro + CT Favors Placebo + CT Cortes. ASCO 2020. Abstr 1000. Reproduced with permission. Slide credit: clinicaloptions. com
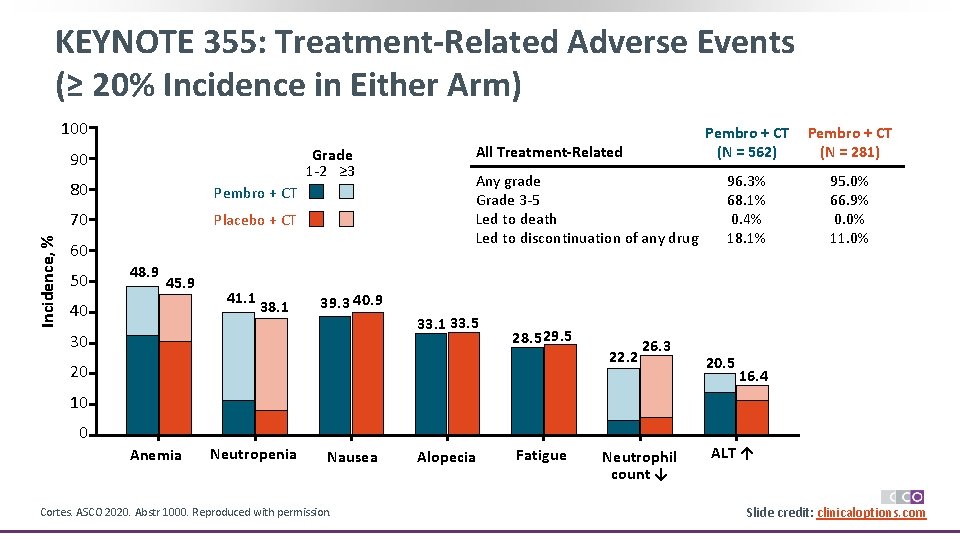
KEYNOTE 355: Treatment-Related Adverse Events (≥ 20% Incidence in Either Arm) 100 Grade 1 -2 ≥ 3 Incidence, % 90 80 Pembro + CT 70 Placebo + CT 60 50 48. 9 45. 9 40 41. 1 38. 1 All Treatment-Related Any grade Grade 3 -5 Led to death Led to discontinuation of any drug Pembro + CT (N = 562) Pembro + CT (N = 281) 96. 3% 68. 1% 0. 4% 18. 1% 95. 0% 66. 9% 0. 0% 11. 0% 39. 3 40. 9 33. 1 33. 5 30 28. 5 29. 5 20 22. 2 26. 3 20. 5 16. 4 10 0 Anemia Neutropenia Nausea Cortes. ASCO 2020. Abstr 1000. Reproduced with permission. Alopecia Fatigue Neutrophil count ↓ ALT ↑ Slide credit: clinicaloptions. com
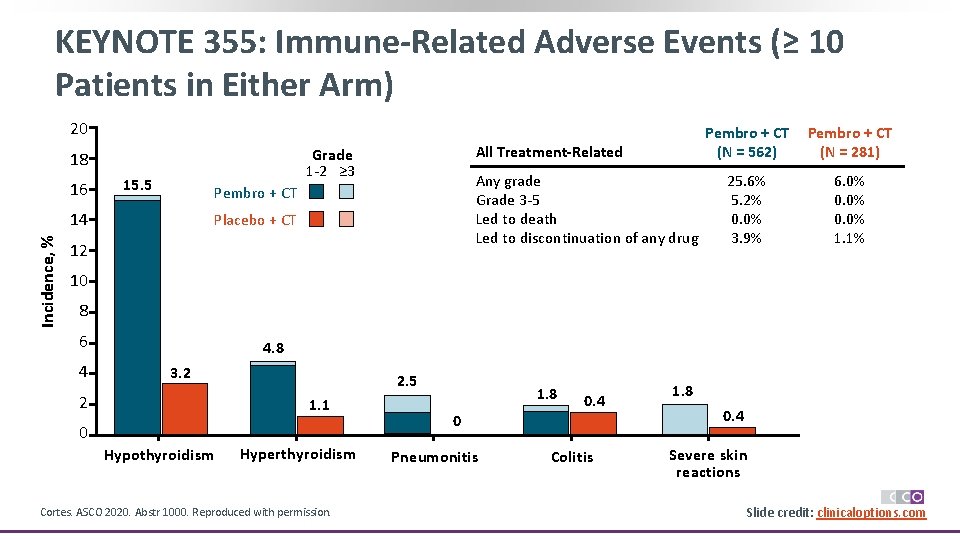
KEYNOTE 355: Immune-Related Adverse Events (≥ 10 Patients in Either Arm) 20 16 15. 5 Any grade Grade 3 -5 Led to death Led to discontinuation of any drug Pembro + CT 14 Incidence, % All Treatment-Related Grade 1 -2 ≥ 3 18 Placebo + CT 12 Pembro + CT (N = 562) Pembro + CT (N = 281) 25. 6% 5. 2% 0. 0% 3. 9% 6. 0% 0. 0% 1. 1% 10 8 6 4 4. 8 3. 2 2 2. 5 1. 1 0 Hypothyroidism Hyperthyroidism Cortes. ASCO 2020. Abstr 1000. Reproduced with permission. 1. 8 0. 4 0 Pneumonitis Colitis 1. 8 0. 4 Severe skin reactions Slide credit: clinicaloptions. com

KEYNOTE-355: Conclusions § Pembrolizumab + chemotherapy significantly improved PFS compared with chemotherapy alone as first-line therapy in patients with m. TNBC with PD-L 1 CPS ≥ 10 ‒ PFS improvement reported across patient subsets § Safety outcomes were consistent with previous data § Investigators suggest that the addition of pembrolizumab to standard chemotherapy may have a role for the first-line treatment of m. TNBC Cortes. ASCO 2020. Abstr 1000. Slide credit: clinicaloptions. com

Go Online for More CCO Coverage of ASCO 2020! Short slideset summaries and additional CME-certified analyses with expert faculty commentary on key studies in: § Breast cancer § Gynecologic cancers § Gastrointestinal cancers § Hematologic malignancies § Genitourinary cancers § Lung cancer clinicaloptions. com/oncology
- Slides: 15