KEYNOTE189 Randomized DoubleBlind Phase 3 Study of Pembrolizumab

KEYNOTE-189: Randomized, Double-Blind, Phase 3 Study of Pembrolizumab or Placebo plus Pemetrexed and Platinum as First-Line Therapy for Metastatic NSCLC Leena Gandhi, Delvys Rodríguez-Abreu, Shirish Gadgeel, Emilio Esteban, Enriqueta Felip, Flávia De Angelis, Manuel Domine, Philip Clingan, Maximilian J. Hochmair, Steven Powell, Susanna Yee-Shan Cheng, Helge G. Bischoff, Nir Peled, Francesco Grossi, Ross R. Jennens, Martin Reck, Rina Hui, Edward B. Garon, Michael Boyer, Belén Rubio-Viqueira, Silvia Novello, Takayasu Kurata, Jhanelle E. Gray, John Vida, Ziwen Wei, Jing Yang, Harry Raftopoulos, M. Catherine Pietanza, Marina C. Garassino

Disclosures • Leena Gandhi: advisory board member for and research funding from Merck & Co. , Inc. , Kenilworth, NJ, USA • Study funding: Merck Sharp & Dohme Corp. , a subsidiary of Merck & Co. , Inc. , Kenilworth, NJ, USA • Medical writing assistance: Melanie Leiby, Merck & Co. , Inc. , Kenilworth, NJ, USA

Chemotherapy Has Complex and Pleiotropic Effects on Antitumor Immune Responses Promotion of Antitumor Immune Response • Antigen shedding and presentation • Release of cancer antigens • Upregulation of MHC I • Enhanced DC activation • Altered immune regulatory receptors, ligands, and cytokines • Increased Teff function, proliferations, and recruitment • Activation of innate immunity • e. g. , STING, RIG-1, TLR 9 • Favorable effect on immune regulatory cells • Suppression of Tregs , MDSCs, etc Enhances positive immune effects of chemotherapy Impairment of Antitumor Immune Response C H E M O T H E R A P Y Anti–PD-1 • Post chemotherapy Induction of immune regulatory receptors, ligands, and cytokines • e. g. , negative feedback from IFNγ • Decreased Teff function • Unfavorable effect on immune regulatory cells • Reduced number of circulating lymphocytes • Increased number of circulating monocytes, MDSCs, etc Reduces negative immune effects of chemotherapy Bracci L et al. Cell Death Differ 2014; 21: 15 -25. Roselli M et al. Oncoimmunology 2013; 2: e 27025. Galluzzi L et al. Cancer Cell 2015; 28: 690 -714. Medler TR et al. Trends Cancer 2015; 1: 66 -75. van Meir H et al. Oncoimmunology 2017; 6: e 1267095. Peng J et al. Cancer Res 2015; 75: 5034 -5045. Zhang P et al. Cancer Sci 2016; 107: 1563 -1571. Novosiadly RD et al. 18 th IASLC World Conference on Lung Cancer; Oct 15 -18, 2017; abstract P 3. 07 -006.
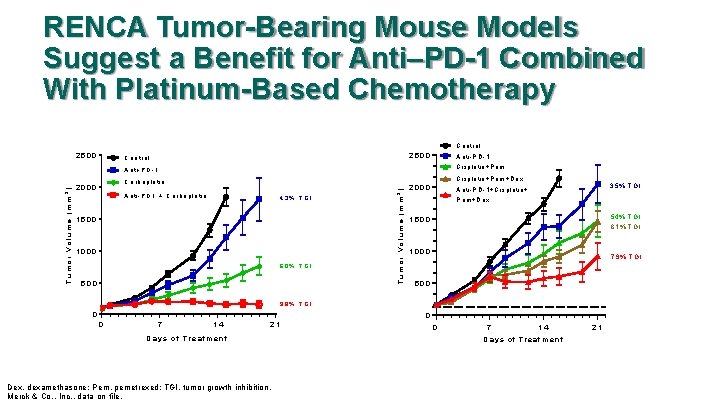
RENCA Tumor-Bearing Mouse Models Suggest a Benefit for Anti–PD-1 Combined With Platinum-Based Chemotherapy C on tro l 2500 C o n tr o l A n t i-P D -1 C is p la tin + P e m + D e x C a r b o p la tin 2000 A n ti- P D 1 + C a r b o p la t in 43% TG I 1500 1000 60% TG I 500 T u m o r V o lu m e (m m 3 ) A n t i- P D - 1 2000 35% TGI A n t i-P D -1 + C is p la tin + P em + D ex 54% TGI 61% TGI 1500 1000 79% TGI 500 98% TG I 0 0 0 7 14 21 D a y s o f T re a tm e n t Dex, dexamethasone; Pem, pemetrexed; TGI, tumor growth inhibition. Merck & Co. , Inc. , data on file. 0 7 14 D a y s o f T re a tm e n t 21

Pembrolizumab in NSCLC • Humanized Ig. G 4 monoclonal antibody against PD-1 approved as monotherapy for: – First-line treatment of advanced NSCLC with PD-L 1 TPS ≥ 50% – Previously treated advanced NSCLC with PD-L 1 TPS ≥ 1% • Pembrolizumab in combination with pemetrexed and carboplatin approved in US and several other countries based on data from KEYNOTE-021 cohort G 1, 2 – Randomized, open-label, phase 2 study of pemetrexed and carboplatin with or without pembrolizumab – Improved ORR and PFS TPS, tumor proportion score. 1. Langer CJ et al. Lancet Oncol 2016; 17: 1497 -1508; 2. Borghaei H et al. ESMO Congress 2017; Sep 8 -11, 2017; Madrid, Spain; abstract LBA 49.
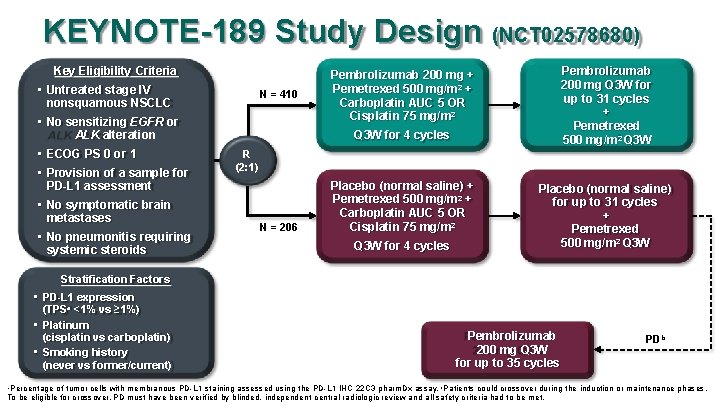
KEYNOTE-189 Study Design Key Eligibility Criteria • Untreated stage IV nonsquamous NSCLC • No sensitizing EGFR or ALK alteration • ECOG PS 0 or 1 • Provision of a sample for PD-L 1 assessment • No symptomatic brain metastases • No pneumonitis requiring systemic steroids N = 410 (NCT 02578680) Pembrolizumab 200 mg Q 3 W for up to 31 cycles + Pemetrexed 500 mg/m 2 Q 3 W Pembrolizumab 200 mg + Pemetrexed 500 mg/m 2 + Carboplatin AUC 5 OR Cisplatin 75 mg/m 2 Q 3 W for 4 cycles R (2: 1) N = 206 Placebo (normal saline) + Pemetrexed 500 mg/m 2 + Carboplatin AUC 5 OR Cisplatin 75 mg/m 2 Q 3 W for 4 cycles Placebo (normal saline) for up to 31 cycles + Pemetrexed 500 mg/m 2 Q 3 W Stratification Factors • PD-L 1 expression (TPSa <1% vs ≥ 1%) • Platinum (cisplatin vs carboplatin) • Smoking history (never vs former/current) Pembrolizumab 200 mg Q 3 W for up to 35 cycles PD b Percentage of tumor cells with membranous PD-L 1 staining assessed using the PD-L 1 IHC 22 C 3 pharm. Dx assay. b Patients could crossover during the induction or maintenance phases. To be eligible for crossover, PD must have been verified by blinded, independent central radiologic review and all safety criteria had to be met. a

Analysis Populations and End Points Analysis Populations • Efficacy – Intention-to-treat (ITT) • Safety – All patients who received ≥ 1 dose of study medication • End Points for Discussion • Primary – Overall survival – Progression-free survival • Secondary – Response rate – Duration of response – Safety • Exploratory – Effect of PD-L 1 expression on efficacy – Patient-reported outcomes

Statistical Considerations • Planned enrollment: 570 patients – Actual enrollment: 616 patients • Overall alpha for study: strictly controlled at one-sided α = 2. 5% – Study had 90% power to show HR for PFS of 0. 70 at one-sided α = 0. 0095 and HR for OS of 0. 70 at a one-sided α = 0. 0155 – Protocol specified 2 interim analyses before the final analysis • First interim analysis (reviewed by external, independent data monitoring committee) – – – a b Planned to occur after enrollment complete and ~370 PFS events observeda Data cutoff date: Nov 8, 2017 Median follow-up: 10. 5 months (range, 0. 2 -20. 4) Observed number of events: 410 for PFS, 235 for OS One-sided alpha levelsb: 0. 00559 for PFS, 0. 00128 for OS It was anticipated that there would be ~242 OS events at that time. Multiplicity adjusted based on the observed number of events using the O’Brien-Fleming spending function.
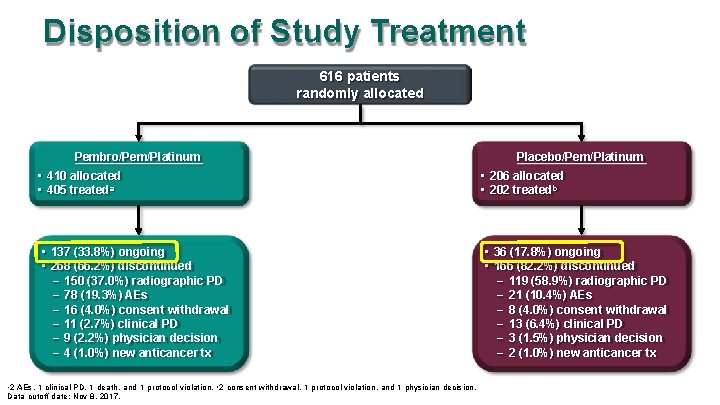
Disposition of Study Treatment 616 patients randomly allocated Pembro/Pem/Platinum Placebo/Pem/Platinum • 410 allocated • 405 treateda • 206 allocated • 202 treatedb • 137 (33. 8%) ongoing • 268 (66. 2%) discontinued – 150 (37. 0%) radiographic PD – 78 (19. 3%) AEs – 16 (4. 0%) consent withdrawal – 11 (2. 7%) clinical PD – 9 (2. 2%) physician decision – 4 (1. 0%) new anticancer tx • 36 (17. 8%) ongoing • 166 (82. 2%) discontinued – 119 (58. 9%) radiographic PD – 21 (10. 4%) AEs – 8 (4. 0%) consent withdrawal – 13 (6. 4%) clinical PD – 3 (1. 5%) physician decision – 2 (1. 0%) new anticancer tx a 2 AEs, 1 clinical PD, 1 death, and 1 protocol violation. b 2 consent withdrawal, 1 protocol violation, and 1 physician decision. Data cutoff date: Nov 8, 2017.
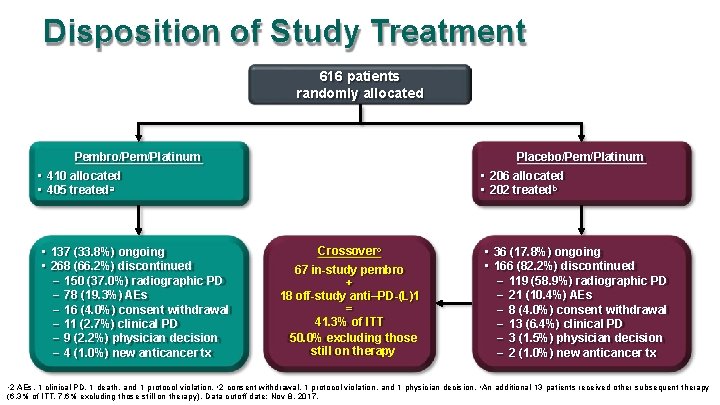
Disposition of Study Treatment 616 patients randomly allocated Pembro/Pem/Platinum Placebo/Pem/Platinum • 410 allocated • 405 treateda • 137 (33. 8%) ongoing • 268 (66. 2%) discontinued – 150 (37. 0%) radiographic PD – 78 (19. 3%) AEs – 16 (4. 0%) consent withdrawal – 11 (2. 7%) clinical PD – 9 (2. 2%) physician decision – 4 (1. 0%) new anticancer tx • 206 allocated • 202 treatedb Crossoverc 67 in-study pembro + 18 off-study anti–PD-(L)1 = 41. 3% of ITT 50. 0% excluding those still on therapy • 36 (17. 8%) ongoing • 166 (82. 2%) discontinued – 119 (58. 9%) radiographic PD – 21 (10. 4%) AEs – 8 (4. 0%) consent withdrawal – 13 (6. 4%) clinical PD – 3 (1. 5%) physician decision – 2 (1. 0%) new anticancer tx a 2 AEs, 1 clinical PD, 1 death, and 1 protocol violation. b 2 consent withdrawal, 1 protocol violation, and 1 physician decision. c An additional 13 patients received other subsequent therapy (6. 3% of ITT, 7. 6% excluding those still on therapy). Data cutoff date: Nov 8, 2017.
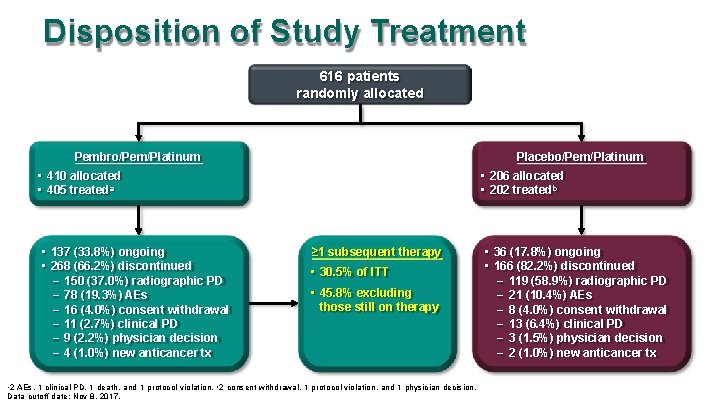
Disposition of Study Treatment 616 patients randomly allocated Pembro/Pem/Platinum Placebo/Pem/Platinum • 410 allocated • 405 treateda • 137 (33. 8%) ongoing • 268 (66. 2%) discontinued – 150 (37. 0%) radiographic PD – 78 (19. 3%) AEs – 16 (4. 0%) consent withdrawal – 11 (2. 7%) clinical PD – 9 (2. 2%) physician decision – 4 (1. 0%) new anticancer tx • 206 allocated • 202 treatedb ≥ 1 subsequent therapy • 30. 5% of ITT • 45. 8% excluding those still on therapy a 2 AEs, 1 clinical PD, 1 death, and 1 protocol violation. b 2 consent withdrawal, 1 protocol violation, and 1 physician decision. Data cutoff date: Nov 8, 2017. • 36 (17. 8%) ongoing • 166 (82. 2%) discontinued – 119 (58. 9%) radiographic PD – 21 (10. 4%) AEs – 8 (4. 0%) consent withdrawal – 13 (6. 4%) clinical PD – 3 (1. 5%) physician decision – 2 (1. 0%) new anticancer tx
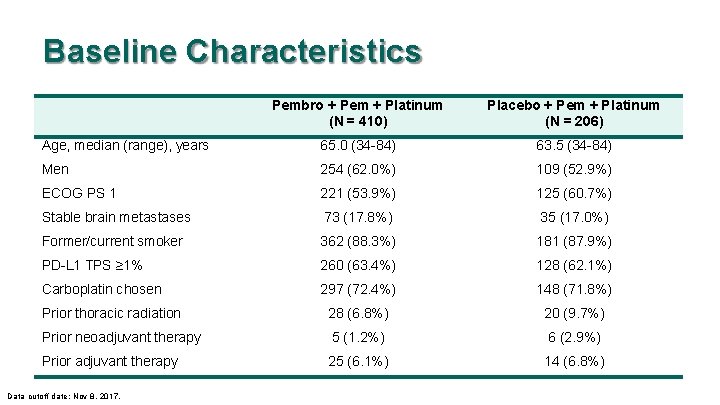
Baseline Characteristics Pembro + Pem + Platinum (N = 410) Placebo + Pem + Platinum (N = 206) Age, median (range), years 65. 0 (34 -84) 63. 5 (34 -84) Men 254 (62. 0%) 109 (52. 9%) ECOG PS 1 221 (53. 9%) 125 (60. 7%) Stable brain metastases 73 (17. 8%) 35 (17. 0%) Former/current smoker 362 (88. 3%) 181 (87. 9%) PD-L 1 TPS ≥ 1% 260 (63. 4%) 128 (62. 1%) Carboplatin chosen 297 (72. 4%) 148 (71. 8%) Prior thoracic radiation 28 (6. 8%) 20 (9. 7%) Prior neoadjuvant therapy 5 (1. 2%) 6 (2. 9%) Prior adjuvant therapy 25 (6. 1%) 14 (6. 8%) Data cutoff date: Nov 8, 2017.
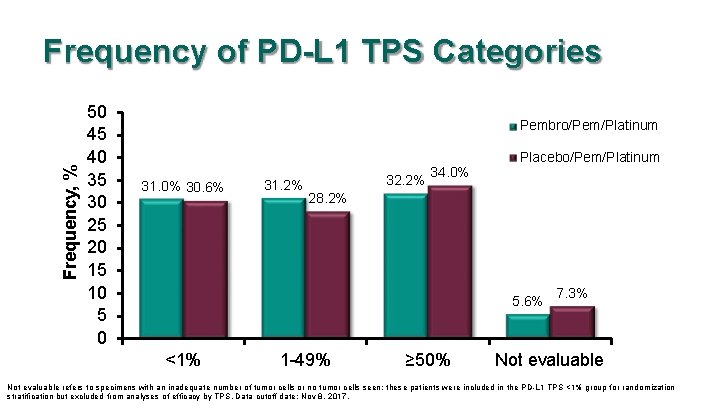
Frequency, % Frequency of PD-L 1 TPS Categories 50 45 40 35 30 25 20 15 10 5 0 Pembro/Pem/Platinum 31. 0% 30. 6% 31. 2% 32. 2% 34. 0% Placebo/Pem/Platinum 28. 2% 5. 6% <1% 1 -49% ≥ 50% 7. 3% Not evaluable refers to specimens with an inadequate number of tumor cells or no tumor cells seen; these patients were included in the PD-L 1 TPS <1% group for randomization stratification but excluded from analyses of efficacy by TPS. Data cutoff date: Nov 8, 2017.
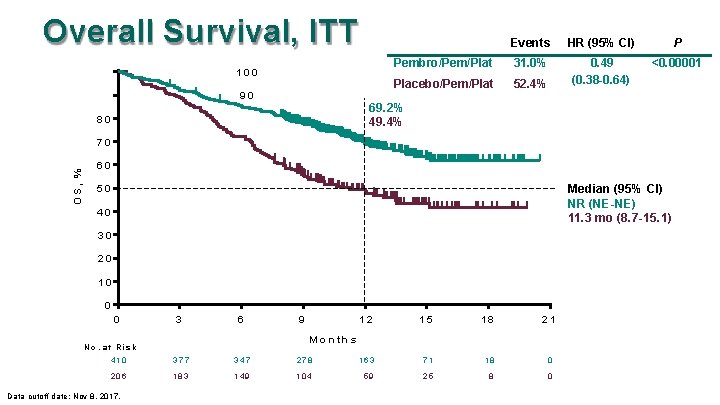
Overall Survival, ITT 100 90 Events HR (95% CI) P Pembro/Pem/Plat 31. 0% <0. 00001 Placebo/Pem/Plat 52. 4% 0. 49 (0. 38 -0. 64) 69. 2% 49. 4% 80 OS, % 70 60 Median (95% CI) NR (NE-NE) 11. 3 mo (8. 7 -15. 1) 50 40 30 20 10 0 0 3 6 9 12 15 18 21 M o n th s N o. a t R is k 410 377 347 278 163 71 18 0 206 183 149 104 59 25 8 0 Data cutoff date: Nov 8, 2017.
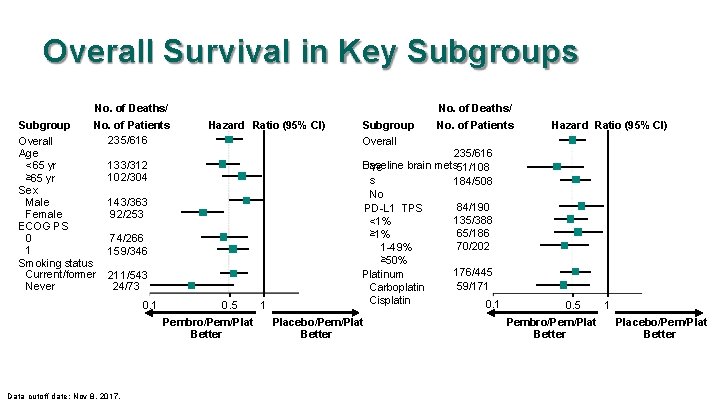
Overall Survival in Key Subgroups No. of Deaths/ Subgroup No. of Patients 235/616 Overall Age 133/312 <65 yr ≥ 65 yr 102/304 Sex 143/363 Male 92/253 Female ECOG PS 74/266 0 1 159/346 Smoking status Current/former 211/543 Never 24/73 0. 1 No. of Deaths/ Hazard Ratio (95% CI) 0. 5 Pembro/Pem/Plat Better Data cutoff date: Nov 8, 2017. 1 Subgroup Overall No. of Patients 235/616 Baseline brain mets 51/108 Ye s 184/508 No 84/190 PD-L 1 TPS 135/388 <1% 65/186 ≥ 1% 70/202 1 -49% ≥ 50% 176/445 Platinum 59/171 Carboplatin Cisplatin 0. 1 Placebo/Pem/Plat Better Hazard Ratio (95% CI) 0. 5 Pembro/Pem/Plat Better 1 Placebo/Pem/Plat Better
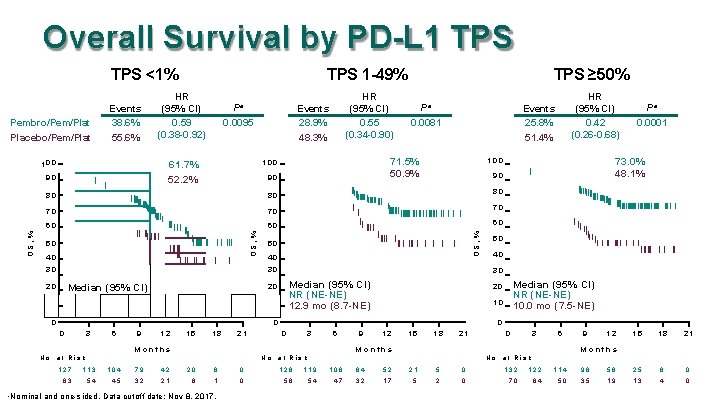
Overall Survival by PD-L 1 TPS <1% 100 Pa HR (95% CI) 0. 55 (0. 34 -0. 90) Events 28. 9% 48. 3% 0. 0095 90 80 70 70 60 60 OS, % 80 50 40 30 0. 0081 100 0 3 6 9 12 15 18 21 70 60 50 40 30 Median (95% CI) NR (NE-NE) 12. 9 mo (8. 7 -NE) Median (95% CI) NR (NE-NE) 10. 0 mo (7. 5 -NE) 20 10 0 0 3 6 9 12 15 18 21 0 3 6 M o n th s N o. a t R is k 0. 0001 73. 0% 48. 1% 90 0 0 Pa 80 20 Median (95% CI) HR (95% CI) 0. 42 (0. 26 -0. 68) Events 25. 8% 51. 4% 30 20 a Pa 71. 5% 50. 9% 100 61. 7% 52. 2% 90 OS, % HR (95% CI) 0. 59 (0. 38 -0. 92) TPS ≥ 50% OS, % Pembro/Pem/Plat Placebo/Pem/Plat Events 38. 6% 55. 6% TPS 1 -49% 9 12 15 18 21 M o n th s N o. a t R is k 127 113 104 79 42 20 6 0 128 119 108 84 52 21 5 0 132 122 114 96 56 25 6 0 63 54 45 32 21 6 1 0 58 54 47 32 17 5 2 0 70 64 50 35 19 13 4 0 Nominal and one-sided. Data cutoff date: Nov 8, 2017.
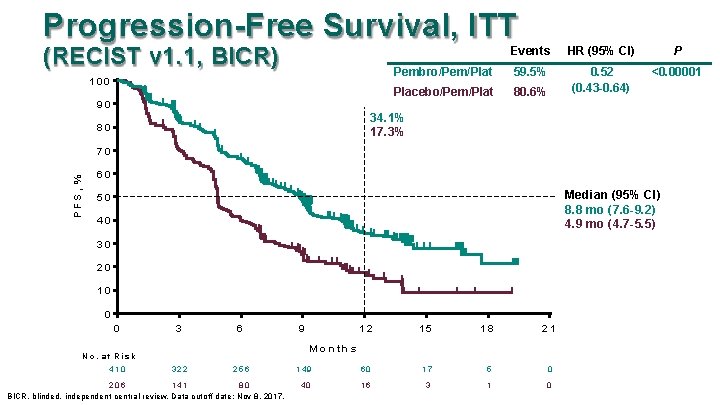
Progression-Free Survival, ITT (RECIST v 1. 1, BICR) 100 Events HR (95% CI) P Pembro/Pem/Plat 59. 5% <0. 00001 Placebo/Pem/Plat 80. 6% 0. 52 (0. 43 -0. 64) 90 34. 1% 17. 3% 80 PFS, % 70 60 Median (95% CI) 8. 8 mo (7. 6 -9. 2) 4. 9 mo (4. 7 -5. 5) 50 40 30 20 10 0 0 3 6 12 15 18 21 149 60 17 5 0 40 16 3 1 0 M o n th s N o. a t R is k 410 9 322 256 206 141 80 BICR, blinded, independent central review. Data cutoff date: Nov 8, 2017.
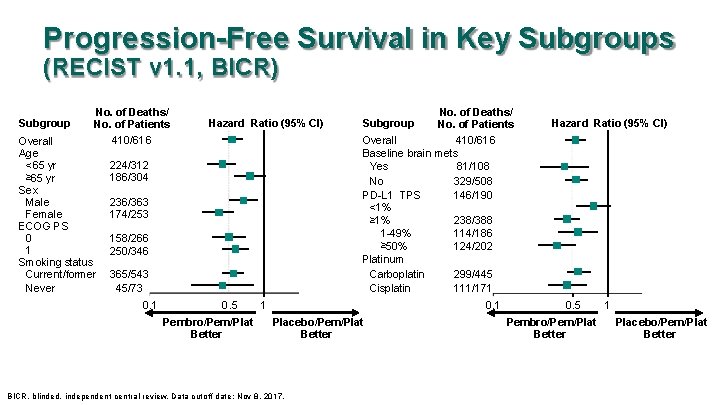
Progression-Free Survival in Key Subgroups (RECIST v 1. 1, BICR) Subgroup No. of Deaths/ No. of Patients Overall Age <65 yr ≥ 65 yr Sex Male Female ECOG PS 0 1 Smoking status Current/former Never Hazard Ratio (95% CI) 410/616 No. of Deaths/ No. of Patients Hazard Ratio (95% CI) 410/616 Overall Baseline brain mets Yes 81/108 No 329/508 PD-L 1 TPS 146/190 <1% ≥ 1% 238/388 1 -49% 114/186 ≥ 50% 124/202 Platinum Carboplatin 299/445 Cisplatin 111/171 224/312 186/304 236/363 174/253 158/266 250/346 365/543 45/73 0. 1 Subgroup 0. 5 Pembro/Pem/Plat Better 1 0. 1 Placebo/Pem/Plat Better BICR, blinded, independent central review. Data cutoff date: Nov 8, 2017. 0. 5 Pembro/Pem/Plat Better 1 Placebo/Pem/Plat Better
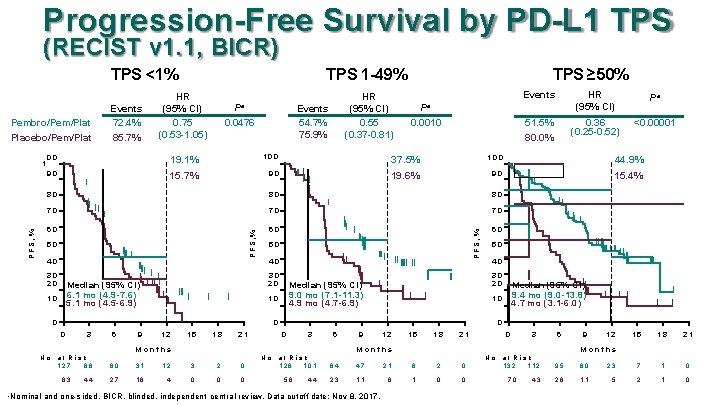
Progression-Free Survival by PD-L 1 TPS (RECIST v 1. 1, BICR) TPS <1% Pembro/Pem/Plat Placebo/Pem/Plat HR (95% CI) 0. 75 (0. 53 -1. 05) Pa HR (95% CI) 0. 55 (0. 37 -0. 81) Events 54. 7% 75. 9% 0. 0476 Events HR (95% CI) Pa 51. 5% 80. 0% 0. 36 (0. 25 -0. 52) <0. 00001 Pa 0. 0010 19. 1% 100 37. 5% 100 44. 9% 90 15. 7% 90 19. 6% 90 15. 4% 80 80 70 70 70 60 60 60 50 40 PFS, % 80 30 20 50 40 30 20 Median (95% CI) 1 0 6. 1 mo (4. 9 -7. 6) 5. 1 mo (4. 5 -6. 9) 3 6 9 12 15 18 21 40 Median (95% CI) 9. 4 mo (9. 0 -13. 8) 4. 7 mo (3. 1 -6. 0) 10 0 0 50 30 20 Median (95% CI) 9. 0 mo (7. 1 -11. 3) 4. 9 mo (4. 7 -6. 9) 10 0 3 6 M o n th s a TPS ≥ 50% 00 P F S , % PFS, % 1 Events 72. 4% 85. 7% TPS 1 -49% 9 12 15 18 21 0 3 6 M o n th s 9 12 15 18 21 M o n th s N o. a t R is k 127 88 60 31 12 3 2 0 N o. a t R is k 128 101 84 47 21 6 2 0 N o. a t R is k 132 112 95 60 23 7 1 0 63 27 16 4 0 0 0 58 23 11 6 1 0 0 70 26 11 5 2 1 0 44 44 Nominal and one-sided. BICR, blinded, independent central review. Data cutoff date: Nov 8, 2017. 43
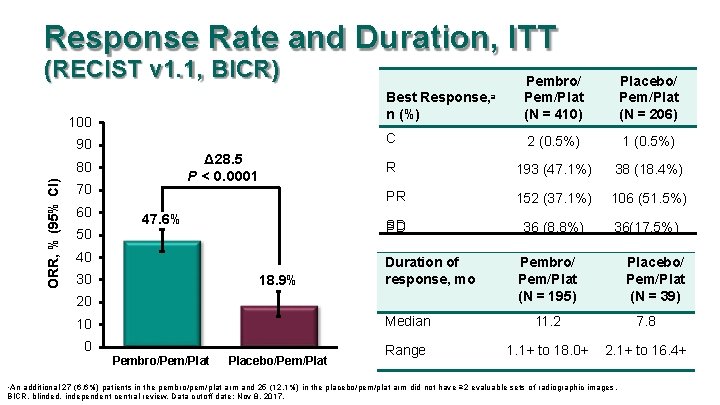
Response Rate and Duration, ITT (RECIST v 1. 1, BICR) 90 Δ 28. 5 P < 0. 0001 ORR, % (95% CI) 80 70 50 Placebo/ Pem/Plat (N = 206) C 2 (0. 5%) 1 (0. 5%) R 193 (47. 1%) 38 (18. 4%) PR 152 (37. 1%) 106 (51. 5%) SD PD 36 (8. 8%) 36(17. 5%) Best n (%) 100 60 Pembro/ Pem/Plat (N = 410) 47. 6% 40 30 18. 9% Response, a Duration of response, mo 20 10 0 Pembro/Pem/Plat Placebo/Pem/Plat Pembro/ Pem/Plat (N = 195) Placebo/ Pem/Plat (N = 39) Median 11. 2 7. 8 Range 1. 1+ to 18. 0+ 2. 1+ to 16. 4+ An additional 27 (6. 6%) patients in the pembro/pem/plat arm and 25 (12. 1%) in the placebo/pem/plat arm did not have ≥ 2 evaluable sets of radiographic images. BICR, blinded, independent central review. Data cutoff date: Nov 8, 2017. a
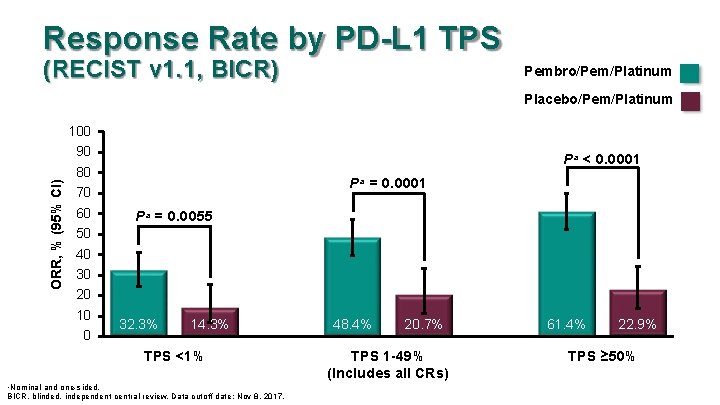
Response Rate by PD-L 1 TPS (RECIST v 1. 1, BICR) Pembro/Pem/Platinum ORR, % (95% CI) Placebo/Pem/Platinum 100 90 80 70 60 50 40 30 20 10 0 Pa < 0. 0001 Pa = 0. 0055 32. 3% 14. 3% TPS <1% Nominal and one-sided. BICR, blinded, independent central review. Data cutoff date: Nov 8, 2017. a 48. 4% 20. 7% TPS 1 -49% (Includes all CRs) 61. 4% 22. 9% TPS ≥ 50%
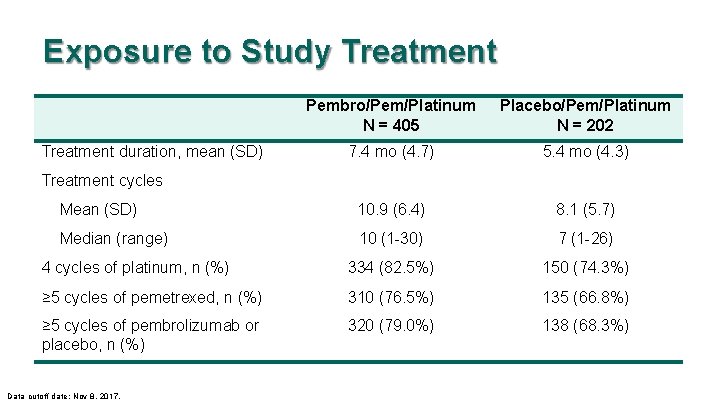
Exposure to Study Treatment Pembro/Pem/Platinum N = 405 Placebo/Pem/Platinum N = 202 7. 4 mo (4. 7) 5. 4 mo (4. 3) Mean (SD) 10. 9 (6. 4) 8. 1 (5. 7) Median (range) 10 (1 -30) 7 (1 -26) 4 cycles of platinum, n (%) 334 (82. 5%) 150 (74. 3%) ≥ 5 cycles of pemetrexed, n (%) 310 (76. 5%) 135 (66. 8%) ≥ 5 cycles of pembrolizumab or placebo, n (%) 320 (79. 0%) 138 (68. 3%) Treatment duration, mean (SD) Treatment cycles Data cutoff date: Nov 8, 2017.
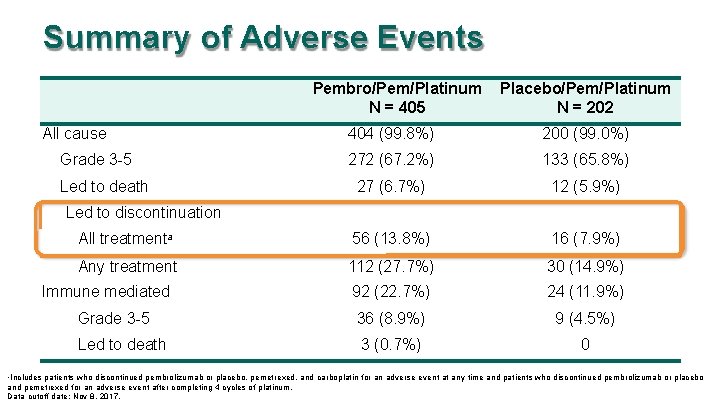
Summary of Adverse Events Pembro/Pem/Platinum N = 405 All cause Placebo/Pem/Platinum N = 202 404 (99. 8%) 200 (99. 0%) 272 (67. 2%) 133 (65. 8%) 27 (6. 7%) 12 (5. 9%) All treatmenta 56 (13. 8%) 16 (7. 9%) Any treatment 112 (27. 7%) 30 (14. 9%) 92 (22. 7%) 24 (11. 9%) Grade 3 -5 36 (8. 9%) 9 (4. 5%) Led to death 3 (0. 7%) 0 Grade 3 -5 Led to death Led to discontinuation Immune mediated Includes patients who discontinued pembrolizumab or placebo, pemetrexed, and carboplatin for an adverse event at any time and patients who discontinued pembrolizumab or placebo and pemetrexed for an adverse event after completing 4 cycles of platinum. Data cutoff date: Nov 8, 2017. a
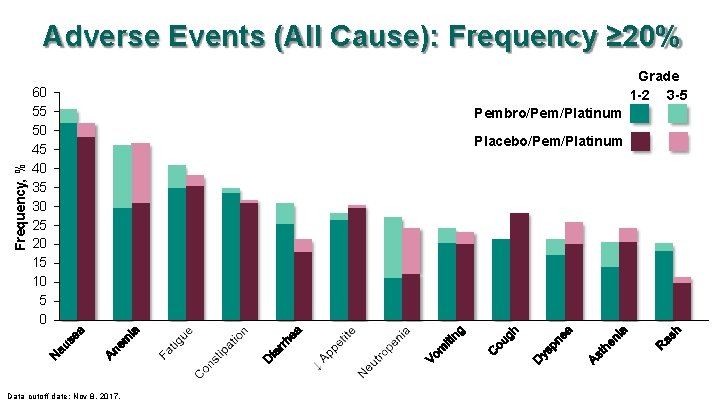
Frequency, % Adverse Events (All Cause): Frequency ≥ 20% 60 55 50 45 40 35 30 25 20 15 10 5 0 Data cutoff date: Nov 8, 2017. Grade 1 -2 3 -5 Pembro/Pem/Platinum Placebo/Pem/Platinum
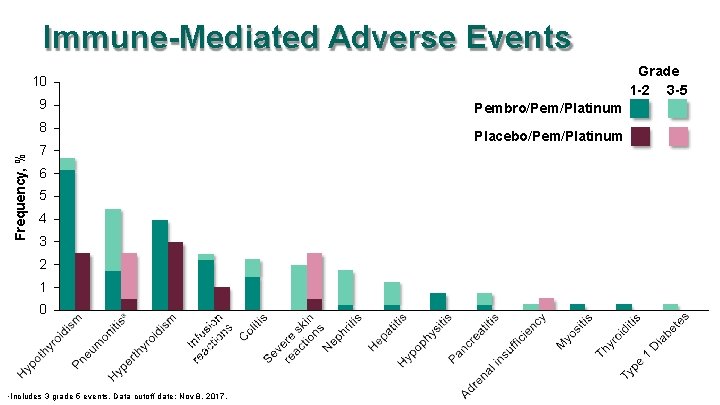
Immune-Mediated Adverse Events Grade 1 -2 3 -5 10 9 Frequency, % 8 7 6 5 4 3 2 1 0 a Includes 3 grade 5 events. Data cutoff date: Nov 8, 2017. Pembro/Pem/Platinum Placebo/Pem/Platinum

Renal Events • Acute kidney injury – Frequency: 5. 2% in pembrolizumab/pemetrexed/platinum arm vs 0. 5% in placebo/pemetrexed/platinum arm – Grade 3 -5 frequency: 2. 0% vs 0% – Grade 5 frequency: 0. 5% (n = 2) – Most events occurred in the setting of acute illness and were likely multifactorial • Nephritisa – Frequency: 1. 7% in pembrolizumab/pemetrexed/platinum vs 0% in placebo/pemetrexed/platinum arm – Grade 3 -5 frequency: 1. 5% vs 0% – Grade 5 frequency: 0% Includes preferred terms of autoimmune nephritis, and tubulointerstitial nephritis. Data cutoff date: Nov 8, 2017. a

Summary • Pembrolizumab plus pemetrexed and platinum provided superior OS compared with placebo plus pemetrexed and platinum (HR 0. 49) – Benefit observed irrespective of PD-L 1 TPS: HR 0. 59 for TPS <1%, 0. 55 for TPS 1 -49%, and 0. 42 for TPS ≥ 50% • PFS was superior (HR 0. 52) • ORR was higher (47. 6% vs 18. 9%) with durable response • AE frequency and severity similar between arms – Pembrolizumab did not appear to exacerbate pemetrexed and platinum-associated AEs – Pemetrexed and platinum did not appear to exacerbate pembrolizumab-associated AEs – Exception may be renal toxicity

Conclusions • Adding pembrolizumab to pemetrexed and platinum induction therapy and pemetrexed maintenance therapy significantly improves OS, PFS, and ORR in patients with untreated metastatic nonsquamous NSCLC without sensitizing EGFR or ALK alterations • Pembrolizumab plus pemetrexed and platinum has a manageable safety profile • Pembrolizumab plus pemetrexed and platinum may be a new standard of care for first-line treatment of metastatic nonsquamous NSCLC, irrespective of PD-L 1 expression

The NEW ENGLAND JOURNAL of M E D I C I N E o_R_r G _IN __A_L_A_R_T_ rc_L_E ll Pembrolizurnab plus Chernotherapy in Metastatic N on-Srnall-Cell Lung Cancer L. Gandhi, D. Rodrrguez-Abreu, S. Gadgeel, E. Esteban, E. Felip, F. De Angelis, M. Domine, P. Clingan, M. j. Hochmair, S. F. Powell, S. Y. -S. Cheng, H. G. Bischoff, N. Peled, F. Grossi, R. R. jennens, M. Reck, R. Hui, E. B. Garon, M. Boyer, B. Rubio-Viqueira, S. Novello, T. Kurata, J. E. Gray, J. Vida, Z. Wei, J. Yang, H. Raftopoulos, M. C. Pietanza, and M. C. Garassino, for the KEYNOTE-189 l nvestigators'

Acknowledgements • PATIENTS AND THEIR FAMILIES • Investigators and site personnel from 126 sites in 16 countries • Eli Lilly and Company (Indianapolis, IN) for providing pemetrexed • Merck & Co. , Inc. (Kenilworth, NJ, USA): Roger Dansey and • Gregory M. Lubiniecki for study support and critical review of presentation;

1 • IMpower 150: Efficacy of Atezolizumab Plus Bevacizumab and Chemotherapy in 1 L Metastatic Nonsquamous NSCLC Across Key Subgroups Marcin Kowanetz, 1 Mark A. Socinski, 2 Wei Zou, 1 Mark Mc. Cleland, 1 Nancy Yang, 1 Ariel Lopez Chavez, 1 Alexander Spira, 3 Julien Mazières, 4 Fadi Braiteh, 5 David Shames, 1 Alan Sandler, 1 Martin Reck 6 1 Genentech, Inc. , South San Francisco, CA; 2 Florida Hospital Cancer Institute, Orlando, FL; 3 Virginia Cancer Specialists Research Institute, Fairfax, VA; 4 Toulouse University Hospital, Toulouse, France; 5 Comprehensive Cancer Centers of Nevada, Las Vegas, NV; 6 Lung Clinic Grosshansdorf, Airway Research Center North, German Center for Lung Research, Grosshansdorf, Germany

2 Disclosure Information AACR Annual Meeting 2018 Mark A. Socinski I have the following financial relationships to disclose: – Received honoraria and research funding from Genentech, Inc. – Participated in speakers’ bureau for Genentech, Inc. I will discuss the following off-label use and/or investigational use: – Atezolizumab in combination with bevacizumab and platinum-doublet chemotherapy in chemotherapy-naïve metastatic nonsquamous NSCLC Kowanetz M, Socinski M, et al. AACR 2018 IMpower 150: Efficacy Across Subgroups

3 Background: Atezolizumab and Treatment of Advanced NSCLC Standards of care for patients with advanced 1 L NSCLC include 1, 2: – Targeted therapies (patients with EGFR mutation or ALK rearrangement) – Pembrolizumab (anti–PD-1) in patients with PD-L 1–expressing tumors with TPS ≥ 50% (≈ 25%-30% prevalence) – Platinum-based chemotherapy +/- bevacizumab 3 , or +/- pembrolizumab 4 Atezolizumab (anti‒PD-L 1) blocks the binding of PD-L 1 to its receptors PD-1 and B 7. 1, thus restoring tumor-specific immunity 5, 6 Atezolizumab has demonstrated overall survival benefit 7 and is approved in the US 8 and EU 9 for the treatment of 2 L+ NSCLC regardless of PD-L 1 expression Phase Ib data of atezolizumab + platinum-doublet chemotherapy in patients with 1 L NSCLC demonstrated promising efficacy and tolerable safety 10 NSCLC; non-small cell lung cancer; PD-1, programmed death-1; PD-L 1, programmed death-ligand 1; TPS, tumour proportion score. 1. Novello S, et al. Ann Oncol, 2016. 2. NCCN Clinical Practice Guidelines in Oncology. NSCLC. V 7. 2017. 3. Sandler A, et al. N Engl J Med, 2006. 4. Langer, C. et al. , Lancet , 2016. 5. Chen DS, Mellman I. Immunity, 2013. 6. Herbst RS, et al. Nature, 2014. 7. Rittmeyer A, et al. Lancet, 2017. 8. TECENTRIQ [USPI]. Genentech Inc, 2017. 9. TECENTRIQ [Sm. PC]. Roche Registration Ltd, 2017. 10. Liu SV, et al. ASCO 2017. Kowanetz M, Socinski M, et al. AACR 2018 IMpower 150: Efficacy Across Subgroups
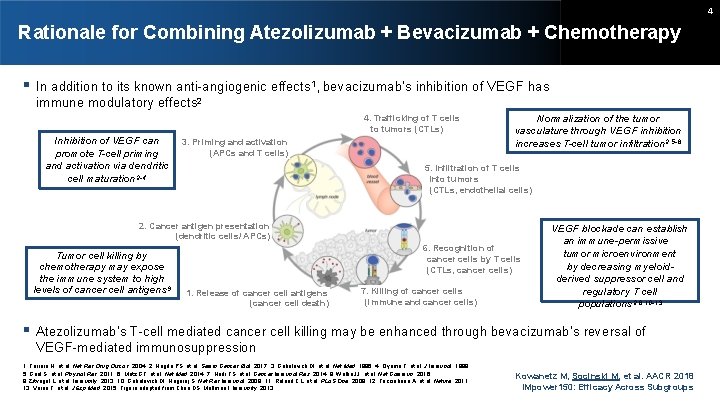
4 Rationale for Combining Atezolizumab + Bevacizumab + Chemotherapy In addition to its known anti-angiogenic effects 1, bevacizumab’s inhibition of VEGF has immune modulatory effects 2 4. Trafficking of T cells to tumors (CTLs) Inhibition of VEGF can promote T-cell priming and activation via dendritic cell maturation 2 -4 3. Priming and activation (APCs and T cells) Normalization of the tumor vasculature through VEGF inhibition increases T-cell tumor infiltration 2, 5 -8 5. Infiltration of T cells into tumors (CTLs, endothelial cells) 2. Cancer antigen presentation (dendritic cells/ APCs) Tumor cell killing by chemotherapy may expose the immune system to high levels of cancer cell antigens 9 6. Recognition of cancer cells by T cells (CTLs, cancer cells) 1. Release of cancer cell antigens (cancer cell death) 7. Killing of cancer cells (immune and cancer cells) VEGF blockade can establish an immune-permissive tumor microenvironment by decreasing myeloidderived suppressor cell and regulatory T cell populations 2, 8, 10 -13 Atezolizumab’s T-cell mediated cancer cell killing may be enhanced through bevacizumab’s reversal of VEGF-mediated immunosuppression 1. Ferrara N, et al. Nat Rev Drug Discov, 2004. 2. Hegde PS, et al. Semin Cancer Biol. 2017. 3. Gabrilovich DI, et al. Nat Med, 1996. 4. Oyama T, et al. J Immunol, 1998. 5. Goel S, et al. Physiol Rev, 2011. 6. Motz GT, et al. Nat Med, 2014. 7. Hodi FS, et al. Cancer Immunol Res, 2014. 8. Wallin JJ, et al. Nat Commun, 2016. 9. Zitvogel L, et al. Immunity, 2013. 10. Gabrilovich DI, Nagaraj S. Nat Rev Immunol, 2009. 11. Roland CL, et al. PLo. S One, 2009. 12. Facciabene A, et al. Nature, 2011. 13. Voron T, et al. J Exp Med, 2015. Figure adapted from Chen DS, Mellman I. Immunity, 2013. Kowanetz M, Socinski M, et al. AACR 2018 IMpower 150: Efficacy Across Subgroups
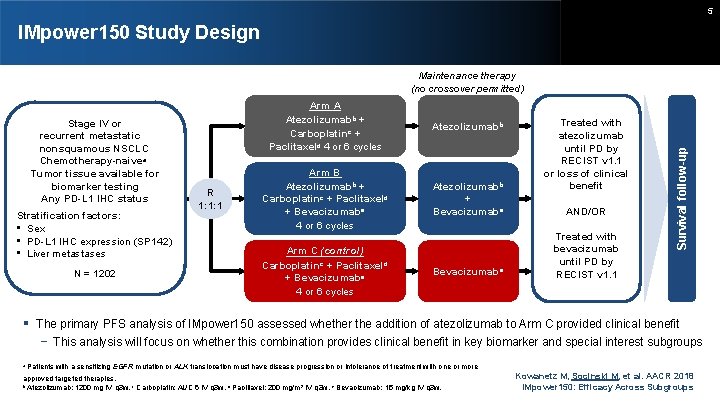
5 IMpower 150 Study Design Stage IV or recurrent metastatic nonsquamous NSCLC Chemotherapy-naivea Tumor tissue available for biomarker testing Any PD-L 1 IHC status Stratification factors: • Sex • PD-L 1 IHC expression (SP 142) • Liver metastases N = 1202 R 1: 1: 1 Arm A Atezolizumabb + Carboplatinc + Paclitaxeld 4 or 6 cycles Atezolizumabb Arm B Atezolizumabb + Carboplatinc + Paclitaxeld + Bevacizumabe 4 or 6 cycles Atezolizumabb + Bevacizumabe Arm C (control) Carboplatinc + Paclitaxeld + Bevacizumabe 4 or 6 cycles Bevacizumabe Treated with atezolizumab until PD by RECIST v 1. 1 or loss of clinical benefit AND/OR Treated with bevacizumab until PD by RECIST v 1. 1 Survival follow-up Maintenance therapy (no crossover permitted) The primary PFS analysis of IMpower 150 assessed whether the addition of atezolizumab to Arm C provided clinical benefit − This analysis will focus on whether this combination provides clinical benefit in key biomarker and special interest subgroups a Patients with a sensitizing EGFR mutation or ALK translocation must have disease progression or intolerance of treatment with one or more approved targeted therapies. b Atezolizumab: 1200 mg IV q 3 w. c Carboplatin: AUC 6 IV q 3 w. d Paclitaxel: 200 mg/m 2 IV q 3 w. e Bevacizumab: 15 mg/kg IV q 3 w. Kowanetz M, Socinski M, et al. AACR 2018 IMpower 150: Efficacy Across Subgroups

6 Biomarkers in IMpower 150 provided the opportunity to evaluate multiple strategies to enrich for PFS, including the T-effector (Teff) gene signature and PD-L 1 IHC (by both the SP 1421 and SP 263 assays) The Teff gene signature is defined by m. RNA expression of 3 genes (PD-L 1, CXCL 9 and IFNγ) and is a surrogate for both PD-L 1 expression and pre-existing immunity − In the Phase III OAK study, Teff gene signature expression appeared to be a more sensitive biomarker of PFS benefit for atezolizumab vs docetaxel than PD-L 1 IHC expression 2 Patients were also screened (locally or centrally) for EGFR mutations or ALK rearrangements IHC, immunohistochemistry. 1. Rittmeyer A, et al. Lancet, 2017. 2. Kowanetz M, et al. WCLC 2017 [abstract MA 05. 09]. Kowanetz M, Socinski M, et al. AACR 2018 IMpower 150: Efficacy Across Subgroups
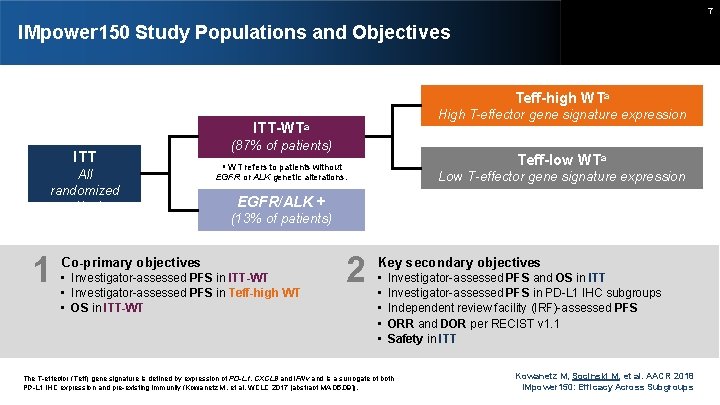
7 IMpower 150 Study Populations and Objectives Teff-high WTa High T-effector gene signature expression ITT-WTa ITT All randomized patients 1 (87% of patients) Teff-low WTa WT refers to patients without EGFR or ALK genetic alterations. a Low T-effector gene signature expression EGFR/ALK + (13% of patients) Co-primary objectives • Investigator-assessed PFS in ITT-WT • Investigator-assessed PFS in Teff-high WT • OS in ITT-WT 2 Key secondary objectives • • • Investigator-assessed PFS and OS in ITT Investigator-assessed PFS in PD-L 1 IHC subgroups Independent review facility (IRF)-assessed PFS ORR and DOR per RECIST v 1. 1 Safety in ITT The T-effector (Teff) gene signature is defined by expression of PD-L 1, CXCL 9 and IFNγ and is a surrogate of both PD-L 1 IHC expression and pre-existing immunity (Kowanetz M, et al. WCLC 2017 [abstract MA 05. 09]). Kowanetz M, Socinski M, et al. AACR 2018 IMpower 150: Efficacy Across Subgroups
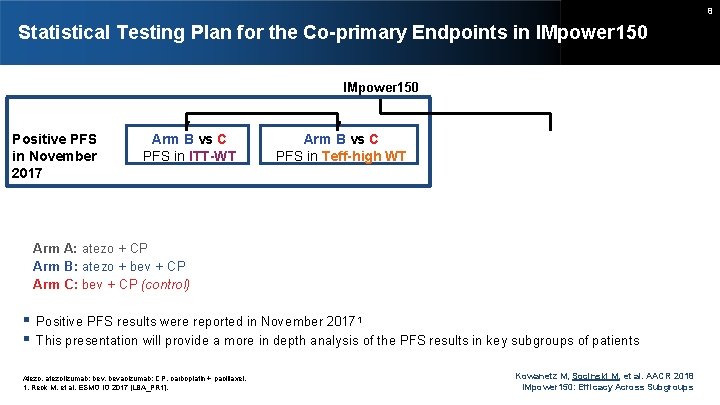
8 Statistical Testing Plan for the Co-primary Endpoints in IMpower 150 Positive PFS in November 2017 Arm B vs C PFS in ITT-WT Arm B vs C PFS in Teff-high WT Arm B vs C OS in ITT-WT Positive OS in March 2018 If OS is significant Arm A: atezo + CP Arm B: atezo + bev + CP Arm C: bev + CP (control) Arm A vs C PFS in ITT-WT and Teff-high WT Arm A vs C OS in ITT-WT Positive PFS results were reported in November 20171 This presentation will provide a more in depth analysis of the PFS results in key subgroups of patients Atezo, atezolizumab; bev, bevacizumab; CP, carboplatin + paclitaxel. 1. Reck M, et al. ESMO IO 2017 [LBA_PR 1]. Kowanetz M, Socinski M, et al. AACR 2018 IMpower 150: Efficacy Across Subgroups
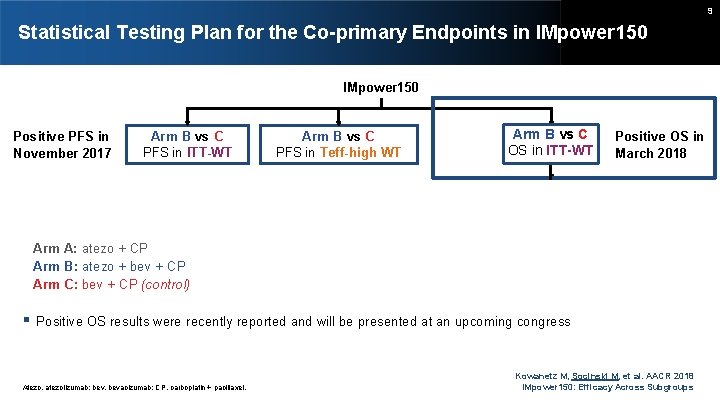
9 Statistical Testing Plan for the Co-primary Endpoints in IMpower 150 Positive PFS in November 2017 Arm B vs C PFS in ITT-WT Arm B vs C PFS in Teff-high WT Arm B vs C OS in ITT-WT Positive OS in March 2018 If OS is significant Arm A: atezo + CP Arm B: atezo + bev + CP Arm C: bev + CP (control) Arm A vs C PFS in ITT-WT and Teff-high WT Arm A vs C OS in ITT-WT Positive OS results were recently reported and will be presented at an upcoming congress Atezo, atezolizumab; bev, bevacizumab; CP, carboplatin + paclitaxel. Kowanetz M, Socinski M, et al. AACR 2018 IMpower 150: Efficacy Across Subgroups
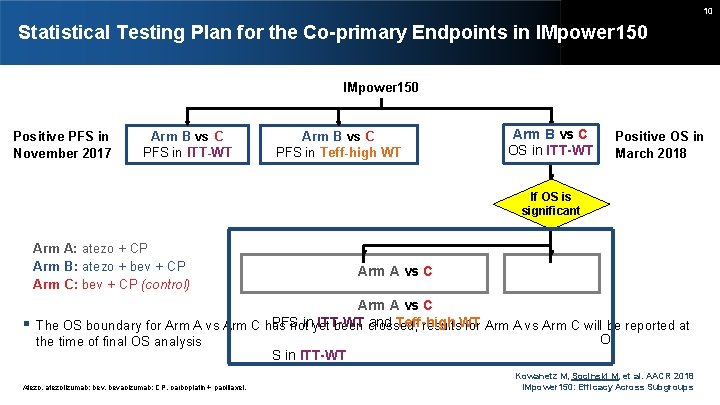
10 Statistical Testing Plan for the Co-primary Endpoints in IMpower 150 Positive PFS in November 2017 Arm B vs C PFS in ITT-WT Arm B vs C PFS in Teff-high WT Arm B vs C OS in ITT-WT Positive OS in March 2018 If OS is significant Arm A: atezo + CP Arm B: atezo + bev + CP Arm C: bev + CP (control) Arm A vs C PFS ITT-WT and Teff-high The OS boundary for Arm A vs Arm C has notinyet been crossed; results. WT for Arm A vs Arm C will be reported at O the time of final OS analysis S in ITT-WT Atezo, atezolizumab; bev, bevacizumab; CP, carboplatin + paclitaxel. Kowanetz M, Socinski M, et al. AACR 2018 IMpower 150: Efficacy Across Subgroups
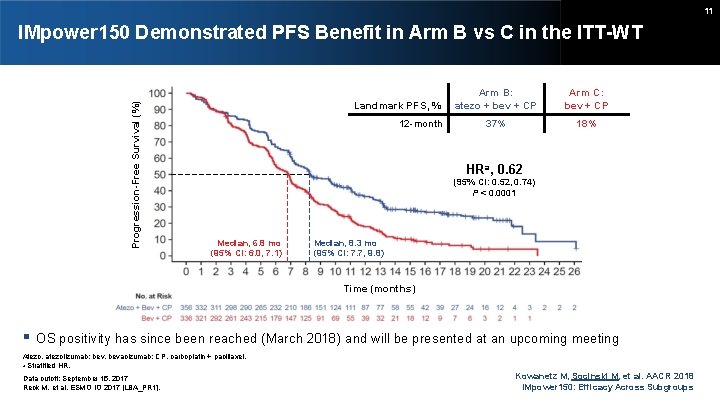
11 Progression-Free Survival (%) IMpower 150 Demonstrated PFS Benefit in Arm B vs C in the ITT-WT Landmark PFS, % 12 -month Arm B: atezo + bev + CP Arm C: bev + CP 37% 18% HRa, 0. 62 (95% CI: 0. 52, 0. 74) P < 0. 0001 Median, 6. 8 mo (95% CI: 6. 0, 7. 1) Median, 8. 3 mo (95% CI: 7. 7, 9. 8) Time (months) OS positivity has since been reached (March 2018) and will be presented at an upcoming meeting Atezo, atezolizumab; bev, bevacizumab; CP, carboplatin + paclitaxel. a Stratified HR. Data cutoff: September 15, 2017 Reck M, et al. ESMO IO 2017 [LBA_PR 1]. Kowanetz M, Socinski M, et al. AACR 2018 IMpower 150: Efficacy Across Subgroups

12 Efficacy in Key Subgroups in the IMpower 150 Study This analysis aims to further understand the PFS efficacy of atezolizumab + bevacizumab + chemotherapy in key patient populations in the IMpower 150 study – PD-L 1 IHC expression subgroups defined by the SP 142 and SP 263 assays (N= 503) – Patients with EGFR/ALK genetic alterations – Patients with liver metastases at baseline Kowanetz M, Socinski M, et al. AACR 2018 IMpower 150: Efficacy Across Subgroups
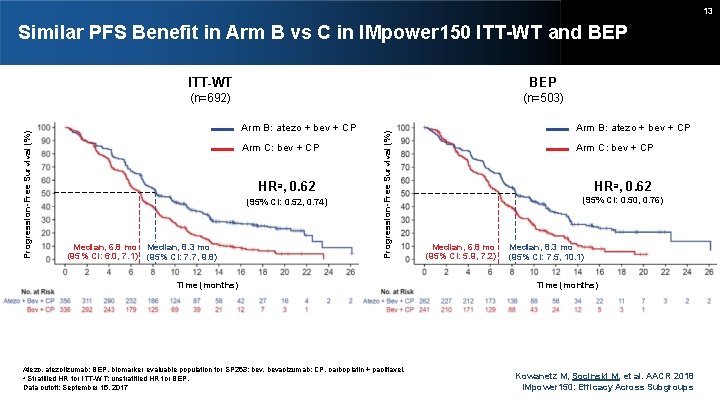
13 ITT-WT BEP (n=692) (n=503) Arm B: atezo + bev + CP Arm C: bev + CP HRa, 0. 62 (95% CI: 0. 52, 0. 74) Median, 6. 8 mo (95% CI: 6. 0, 7. 1) Median, 8. 3 mo (95% CI: 7. 7, 9. 8) Progression-Free Survival (%) Similar PFS Benefit in Arm B vs C in IMpower 150 ITT-WT and BEP Time (months) Atezo, atezolizumab; BEP, biomarker evaluable population for SP 263; bev, bevacizumab; CP, carboplatin + paclitaxel. a Stratified HR for ITT-WT; unstratified HR for BEP. Data cutoff: September 15, 2017 Arm B: atezo + bev + CP Arm C: bev + CP HRa, 0. 62 (95% CI: 0. 50, 0. 76) Median, 6. 8 mo (95% CI: 5. 9, 7. 2) Median, 8. 3 mo (95% CI: 7. 5, 10. 1) Time (months) Kowanetz M, Socinski M, et al. AACR 2018 IMpower 150: Efficacy Across Subgroups
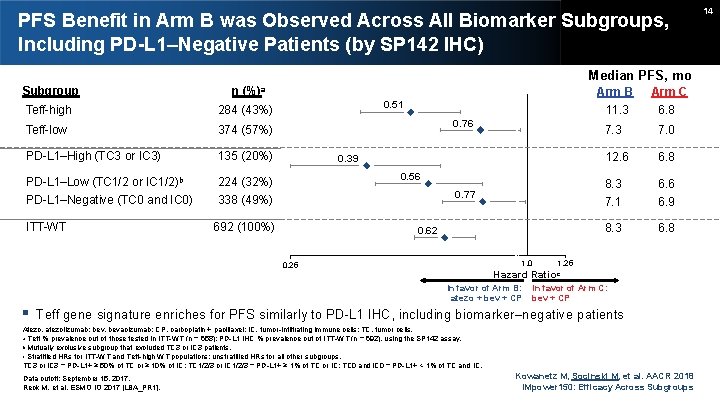
PFS Benefit in Arm B was Observed Across All Biomarker Subgroups, Including PD-L 1–Negative Patients (by SP 142 IHC) Median PFS, mo Subgroup Teff-high n (%)a Teff-low 374 (57%) PD-L 1–High (TC 3 or IC 3) 135 (20%) PD-L 1–Low (TC 1/2 or IC 1/2)b 224 (32%) PD-L 1–Negative (TC 0 and IC 0) ITT-WT 0. 51 284 (43%) 0. 76 0. 39 0. 56 0. 77 338 (49%) 692 (100%) 0. 62 1. 0 0. 25 Arm B Arm C 11. 3 6. 8 7. 3 7. 0 12. 6 6. 8 8. 3 7. 1 6. 6 6. 9 8. 3 6. 8 1. 25 Hazard Ratioc In favor of Arm B: atezo + bev + CP In favor of Arm C: bev + CP Teff gene signature enriches for PFS similarly to PD-L 1 IHC, including biomarker–negative patients Atezo, atezolizumab; bev, bevacizumab; CP, carboplatin + paclitaxel; IC, tumor-infiltrating immune cells; TC, tumor cells. a Teff % prevalence out of those tested in ITT-WT (n = 658); PD-L 1 IHC % prevalence out of ITT-WT (n = 692), using the SP 142 assay. b Mutually exclusive subgroup that excluded TC 3 or IC 3 patients. c Stratified HRs for ITT-WT and Teff-high WT populations; unstratified HRs for all other subgroups. TC 3 or IC 3 = PD-L 1+ ≥ 50% of TC or ≥ 10% of IC; TC 1/2/3 or IC 1/2/3 = PD-L 1+ ≥ 1% of TC or IC; TC 0 and IC 0 = PD-L 1+ < 1% of TC and IC. Data cutoff: September 15, 2017. Reck M, et al. ESMO IO 2017 [LBA_PR 1]. Kowanetz M, Socinski M, et al. AACR 2018 IMpower 150: Efficacy Across Subgroups 14
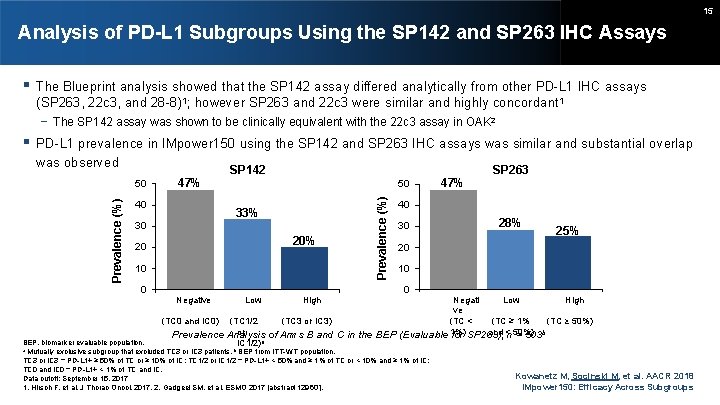
15 Analysis of PD-L 1 Subgroups Using the SP 142 and SP 263 IHC Assays The Blueprint analysis showed that the SP 142 assay differed analytically from other PD-L 1 IHC assays (SP 263, 22 c 3, and 28 -8)1; however SP 263 and 22 c 3 were similar and highly concordant 1 − The SP 142 assay was shown to be clinically equivalent with the 22 c 3 assay in OAK 2 PD-L 1 prevalence in IMpower 150 using the SP 142 and SP 263 IHC assays was similar and substantial overlap Prevalence (%) 50 47% 40 SP 142 50 33% 30 20% 20 10 0 Negative Low High Prevalence (%) was observed SP 263 40 30 28% 25% 20 10 0 Negati Low High ve (TC ≥ 50%) (TC 0 and IC 0) (TC 1/2 (TC 3 or IC 3) (TC < (TC ≥ 1% or 1%) and < 50%) b Prevalence Analysis of Arms B and C in the BEP (Evaluable for SP 263), n = 503 IC 1/2)a BEP, biomarker evaluable population. Mutually exclusive subgroup that excluded TC 3 or IC 3 patients. b BEP from ITT-WT population. TC 3 or IC 3 = PD-L 1+ ≥ 50% of TC or ≥ 10% of IC; TC 1/2 or IC 1/2 = PD-L 1+ < 50% and ≥ 1% of TC or < 10% and ≥ 1% of IC; TC 0 and IC 0 = PD-L 1+ < 1% of TC and IC. Data cutoff: September 15, 2017 1. Hirsch F, et al. J Thorac Oncol. 2017. 2. Gadgeel SM, et al. ESMO 2017 [abstract 1296 O]. a 47% Kowanetz M, Socinski M, et al. AACR 2018 IMpower 150: Efficacy Across Subgroups
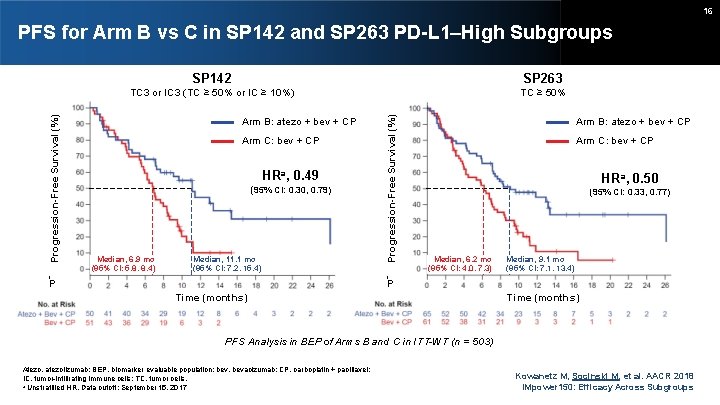
16 PFS for Arm B vs C in SP 142 and SP 263 PD-L 1–High Subgroups SP 142 SP 263 TC 3 or IC 3 (TC ≥ 50% or IC ≥ 10%) TC ≥ 50% on i s s e og r r P Arm B: atezo + bev + CP Arm C: bev + CP HRa, 0. 49 (95% CI: 0. 30, 0. 79) Median, 6. 9 mo (95% CI: 5. 8, 8. 4) Median, 11. 1 mo (95% CI: 7. 2, 15. 4) ) % (l a v i v ur S e Fr e - Progression-Free Survival (%) ) % (l a v i v ur S e Fr e - on i s s e og r r P Arm B: atezo + bev + CP Arm C: bev + CP HRa, 0. 50 (95% CI: 0. 33, 0. 77) Median, 6. 2 mo (95% CI: 4. 0, 7. 3) Time (months) Median, 9. 1 mo (95% CI: 7. 1, 13. 4) Time (months) PFS Analysis in BEP of Arms B and C in ITT-WT (n = 503) Atezo, atezolizumab; BEP, biomarker evaluable population; bev, bevacizumab; CP, carboplatin + paclitaxel; IC, tumor-infiltrating immune cells; TC, tumor cells. a Unstratified HR. Data cutoff: September 15, 2017 Kowanetz M, Socinski M, et al. AACR 2018 IMpower 150: Efficacy Across Subgroups
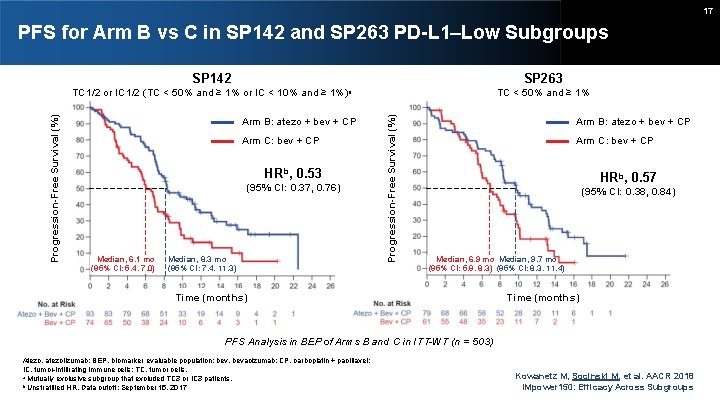
17 PFS for Arm B vs C in SP 142 and SP 263 PD-L 1–Low Subgroups SP 142 SP 263 Arm B: atezo + bev + CP Arm C: bev + CP HRb, 0. 53 (95% CI: 0. 37, 0. 76) Median, 6. 1 mo (95% CI: 5. 4, 7. 0) Median, 8. 3 mo (95% CI: 7. 4, 11. 3) TC < 50% and ≥ 1% Progression-Free Survival (%) TC 1/2 or IC 1/2 (TC < 50% and ≥ 1% or IC < 10% and ≥ 1%)a Arm B: atezo + bev + CP Arm C: bev + CP HRb, 0. 57 (95% CI: 0. 38, 0. 84) Median, 6. 9 mo Median, 9. 7 mo (95% CI: 5. 9, 8. 3) (95% CI: 8. 3, 11. 4) Time (months) PFS Analysis in BEP of Arms B and C in ITT-WT (n = 503) Atezo, atezolizumab; BEP, biomarker evaluable population; bev, bevacizumab; CP, carboplatin + paclitaxel; IC, tumor-infiltrating immune cells; TC, tumor cells. a Mutually exclusive subgroup that excluded TC 3 or IC 3 patients. b Unstratified HR. Data cutoff: September 15, 2017 Kowanetz M, Socinski M, et al. AACR 2018 IMpower 150: Efficacy Across Subgroups
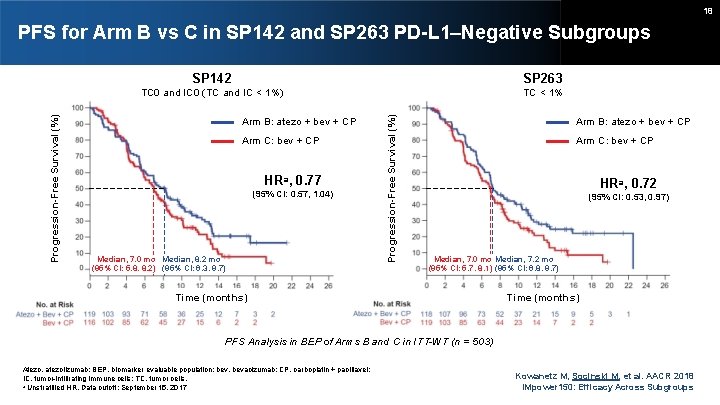
18 SP 142 SP 263 TC 0 and IC 0 (TC and IC < 1%) TC < 1% Arm B: atezo + bev + CP Arm C: bev + CP HRa, 0. 77 (95% CI: 0. 57, 1. 04) Median, 7. 0 mo Median, 8. 2 mo (95% CI: 5. 8, 8. 2) (95% CI: 6. 3, 9. 7) Progression-Free Survival (%) PFS for Arm B vs C in SP 142 and SP 263 PD-L 1–Negative Subgroups Arm B: atezo + bev + CP Arm C: bev + CP HRa, 0. 72 (95% CI: 0. 53, 0. 97) Median, 7. 0 mo Median, 7. 2 mo (95% CI: 5. 7, 8. 1) (95% CI: 6. 8, 9. 7) Time (months) PFS Analysis in BEP of Arms B and C in ITT-WT (n = 503) Atezo, atezolizumab; BEP, biomarker evaluable population; bev, bevacizumab; CP, carboplatin + paclitaxel; IC, tumor-infiltrating immune cells; TC, tumor cells. a Unstratified HR. Data cutoff: September 15, 2017 Kowanetz M, Socinski M, et al. AACR 2018 IMpower 150: Efficacy Across Subgroups
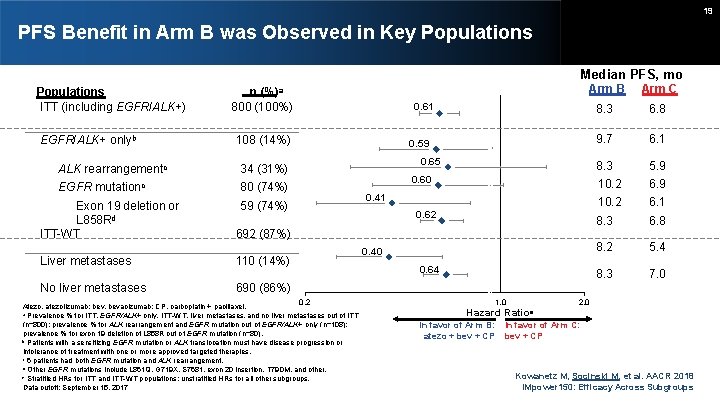
19 PFS Benefit in Arm B was Observed in Key Populations Median PFS, mo Populations ITT (including EGFR/ALK+) EGFR/ALK+ onlyb n (%)a 800 (100%) 34 (31%) EGFR mutationc 80 (74%) Exon 19 deletion or L 858 Rd ITT-WT 59 (74%) 8. 3 6. 8 9. 7 6. 1 8. 3 5. 9 10. 2 6. 1 8. 3 6. 8 8. 2 5. 4 8. 3 7. 0 0. 59 0. 65 0. 60 0. 41 0. 62 692 (87%) Liver metastases 110 (14%) No liver metastases 690 (86%) 0. 2 Atezo, atezolizumab; bev, bevacizumab; CP, carboplatin + paclitaxel. Prevalence % for ITT, EGFR/ALK+ only, ITT-WT, liver metastases, and no liver metastases out of ITT (n=800); prevalence % for ALK rearrangement and EGFR mutation out of EGFR/ALK+ only (n=108); prevalence % for exon 19 deletion of L 858 R out of EGFR mutation (n=80). b Patients with a sensitizing EGFR mutation or ALK translocation must have disease progression or intolerance of treatment with one or more approved targeted therapies. c 6 patients had both EGFR mutation and ALK rearrangement. d Other EGFR mutations include L 861 Q, G 719 X, S 7681, exon 20 insertion, T 790 M, and other. e Stratified HRs for ITT and ITT-WT populations; unstratified HRs for all other subgroups. Data cutoff: September 15, 2017 a Arm C 0. 61 108 (14%) ALK rearrangementc Arm B 0. 40 0. 64 2. 0 1. 0 Hazard Ratioe In favor of Arm B: atezo + bev + CP In favor of Arm C: bev + CP Kowanetz M, Socinski M, et al. AACR 2018 IMpower 150: Efficacy Across Subgroups
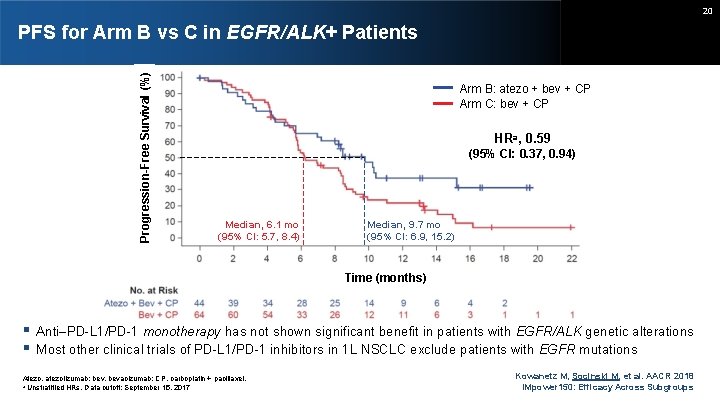
20 Progression-Free Survival (%) PFS for Arm B vs C in EGFR/ALK+ Patients Arm B: atezo + bev + CP Arm C: bev + CP HRa, 0. 59 (95% CI: 0. 37, 0. 94) Median, 6. 1 mo (95% CI: 5. 7, 8. 4) Median, 9. 7 mo (95% CI: 6. 9, 15. 2) Time (months) Anti–PD-L 1/PD-1 monotherapy has not shown significant benefit in patients with EGFR/ALK genetic alterations Most other clinical trials of PD-L 1/PD-1 inhibitors in 1 L NSCLC exclude patients with EGFR mutations Atezo, atezolizumab; bev, bevacizumab; CP, carboplatin + paclitaxel. a Unstratified HRs. Data cutoff: September 15, 2017 Kowanetz M, Socinski M, et al. AACR 2018 IMpower 150: Efficacy Across Subgroups
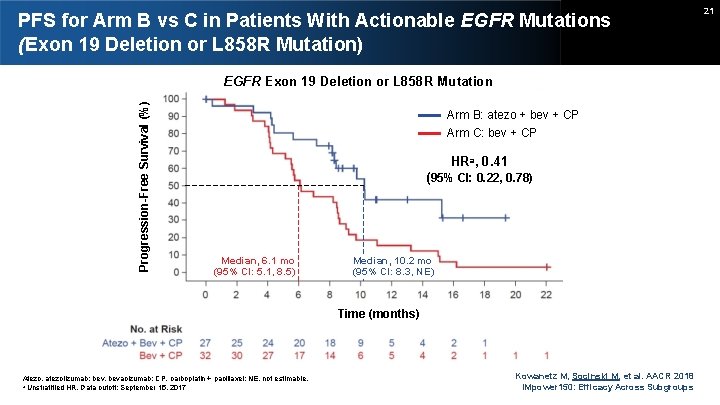
PFS for Arm B vs C in Patients With Actionable EGFR Mutations (Exon 19 Deletion or L 858 R Mutation) Progression-Free Survival (%) EGFR Exon 19 Deletion or L 858 R Mutation Arm B: atezo + bev + CP Arm C: bev + CP HRa, 0. 41 (95% CI: 0. 22, 0. 78) Median, 6. 1 mo (95% CI: 5. 1, 8. 5) Median, 10. 2 mo (95% CI: 8. 3, NE) Time (months) Atezo, atezolizumab; bev, bevacizumab; CP, carboplatin + paclitaxel; NE, not estimable. a Unstratified HR. Data cutoff: September 15, 2017 Kowanetz M, Socinski M, et al. AACR 2018 IMpower 150: Efficacy Across Subgroups 21
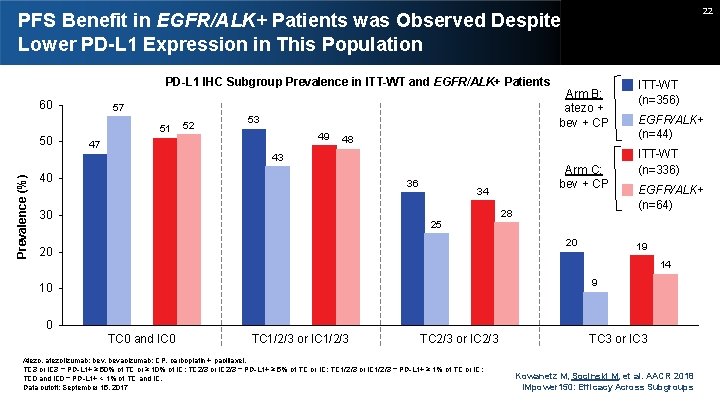
22 PFS Benefit in EGFR/ALK+ Patients was Observed Despite Lower PD-L 1 Expression in This Population PD-L 1 IHC Subgroup Prevalence in ITT-WT and EGFR/ALK+ Patients 60 57 51 50 52 53 49 47 48 43 Prevalence (%) Arm B: atezo + bev + CP 40 36 30 Arm C: bev + CP 34 25 28 20 20 ITT-WT (n=356) EGFR/ALK+ (n=44) ITT-WT (n=336) EGFR/ALK+ (n=64) 19 14 9 10 0 TC 0 and IC 0 TC 1/2/3 or IC 1/2/3 TC 2/3 or IC 2/3 Atezo, atezolizumab; bev, bevacizumab; CP, carboplatin + paclitaxel. TC 3 or IC 3 = PD-L 1+ ≥ 50% of TC or ≥ 10% of IC; TC 2/3 or IC 2/3 = PD-L 1+ ≥ 5% of TC or IC; TC 1/2/3 or IC 1/2/3 = PD-L 1+ ≥ 1% of TC or IC; TC 0 and IC 0 = PD-L 1+ < 1% of TC and IC. Data cutoff: September 15, 2017 TC 3 or IC 3 Kowanetz M, Socinski M, et al. AACR 2018 IMpower 150: Efficacy Across Subgroups
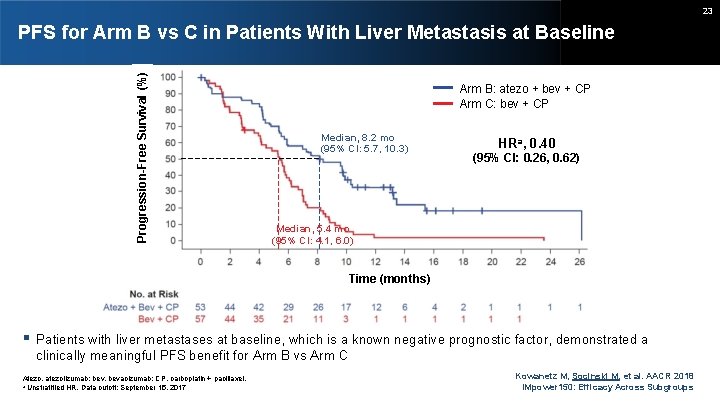
23 Progression-Free Survival (%) PFS for Arm B vs C in Patients With Liver Metastasis at Baseline Arm B: atezo + bev + CP Arm C: bev + CP Median, 8. 2 mo (95% CI: 5. 7, 10. 3) HRa, 0. 40 (95% CI: 0. 26, 0. 62) Median, 5. 4 mo (95% CI: 4. 1, 6. 0) Time (months) Patients with liver metastases at baseline, which is a known negative prognostic factor, demonstrated a clinically meaningful PFS benefit for Arm B vs Arm C Atezo, atezolizumab; bev, bevacizumab; CP, carboplatin + paclitaxel. a Unstratified HR. Data cutoff: September 15, 2017 Kowanetz M, Socinski M, et al. AACR 2018 IMpower 150: Efficacy Across Subgroups
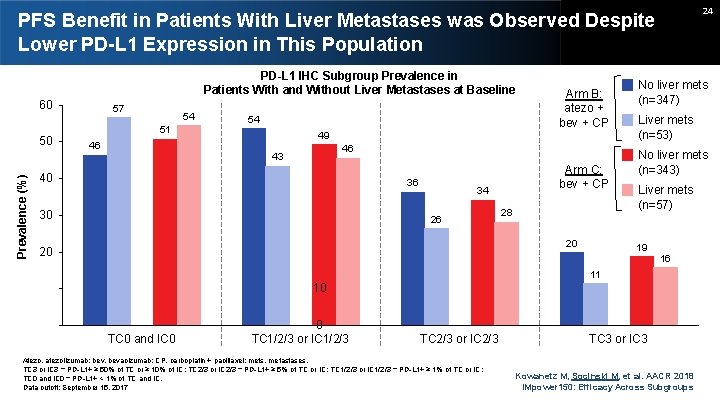
24 PFS Benefit in Patients With Liver Metastases was Observed Despite Lower PD-L 1 Expression in This Population PD-L 1 IHC Subgroup Prevalence in Patients With and Without Liver Metastases at Baseline 60 Prevalence (%) 50 57 54 51 46 54 49 Arm B: atezo + bev + CP 46 43 40 36 30 Arm C: bev + CP 34 26 28 20 20 No liver mets (n=347) Liver mets (n=53) No liver mets (n=343) Liver mets (n=57) 19 16 11 10 TC 0 and IC 0 0 TC 1/2/3 or IC 1/2/3 TC 2/3 or IC 2/3 Atezo, atezolizumab; bev, bevacizumab; CP, carboplatin + paclitaxel; mets, metastases. TC 3 or IC 3 = PD-L 1+ ≥ 50% of TC or ≥ 10% of IC; TC 2/3 or IC 2/3 = PD-L 1+ ≥ 5% of TC or IC; TC 1/2/3 or IC 1/2/3 = PD-L 1+ ≥ 1% of TC or IC; TC 0 and IC 0 = PD-L 1+ < 1% of TC and IC. Data cutoff: September 15, 2017 TC 3 or IC 3 Kowanetz M, Socinski M, et al. AACR 2018 IMpower 150: Efficacy Across Subgroups

25 Summary Atezolizumab + bevacizumab + chemotherapy demonstrated significant PFS benefit in chemotherapy-naïve nonsquamous NSCLC across all PD-L 1 subgroups, regardless of the IHC assay used Clinically meaningful PFS benefit was observed in all patients, including those with EGFR mutations, ALK genomic rearrangements, and liver metastases – Anti–PD-L 1/PD-1 monotherapy has not shown significant benefit in 2 L+ patients with EGFR/ALK genetic alterations – Tumors in patients with liver metastases are characterized by immune suppressive tumor environments, and they usually demonstrate poorer outcomes – The observed efficacy in these key subgroups may be due to the addition of bevacizumab to atezolizumab These data suggest that atezolizumab + bevacizumab + chemotherapy provides a new treatment option for these key patient populations IMpower 150 has recently demonstrated significant OS benefit and these data will be presented at an upcoming meeting Kowanetz M, Socinski M, et al. AACR 2018 IMpower 150: Efficacy Across Subgroups

26 Acknowledgments The patients and their families Participating study investigators and clinical sites This study is sponsored by F. Hoffmann-La Roche, Ltd Anthony Lee, Geetha Shankar, and Wei Yu of Genentech Inc. , for their contributions to the study analyses Medical writing assistance for this presentation was provided by Emily Casey, Ph. D, of Health Interactions and funded by F. Hoffmann-La Roche, Ltd Kowanetz M, Socinski M, et al. AACR 2018 IMpower 150: Efficacy Across Subgroups
- Slides: 56