KEYNOTE059 Cohort 1 Pembrolizumab Monotherapy in Previously Treated

KEYNOTE-059 (Cohort 1): Pembrolizumab Monotherapy in Previously Treated Advanced Gastric or GEJ Adenocarcinoma CCO Independent Conference Highlights* of the 2017 ASCO Annual Meeting; June 2 -6, 2017; Chicago, Illinois *Clinical Care Options (CCO) is an independent medical education organization that provides conference coverage and other unique educational programs for healthcare professionals This activity is supported by educational grants from Abb. Vie, Amgen, Astra. Zeneca, Celgene Corporation, Genentech, Halozyme, Incyte, and Merck & Co. , Inc.

Pembrolizumab for Pretreated Advanced Gastric or GEJ Adenocarcinoma: Background § PD-L 1 overexpression a feature of gastric cancers[1 -3] § Pembrolizumab: humanized anti–PD-1 Ig. G 4 -κ m. Ab; blocks PD-1 interaction with its ligands PD-L 1/2[4] – Phase Ib KEYNOTE-012: manageable toxicity, 22% ORR in pts with advanced PD-L 1+ gastric cancer[5] § Current analysis reports data from phase II KEYNOTE-059 of pembrolizumab with focus on cohort 1 (pts with advanced gastric/GEJ cancer and ≥ 2 prior lines of therapy who received pembrolizumab monotherapy)[6] 1. Kim JW, et al. Gastric Cancer. 2016; 19: 42 -52. 2. Qing Y, et al. Drug Des Devel Ther. 2015; 9: 901 -909. 3. Dong M, et al. Hum Pathol. 2016; 53: 25 -34. 4. Pembrolizumab [package insert]. 5. Muro K, et al. Lancet Oncol. 2016; 17: 717 -726. 6. Fuchs CS, et al. ASCO 2017. Abstract 4003. Slide credit: clinicaloptions. com
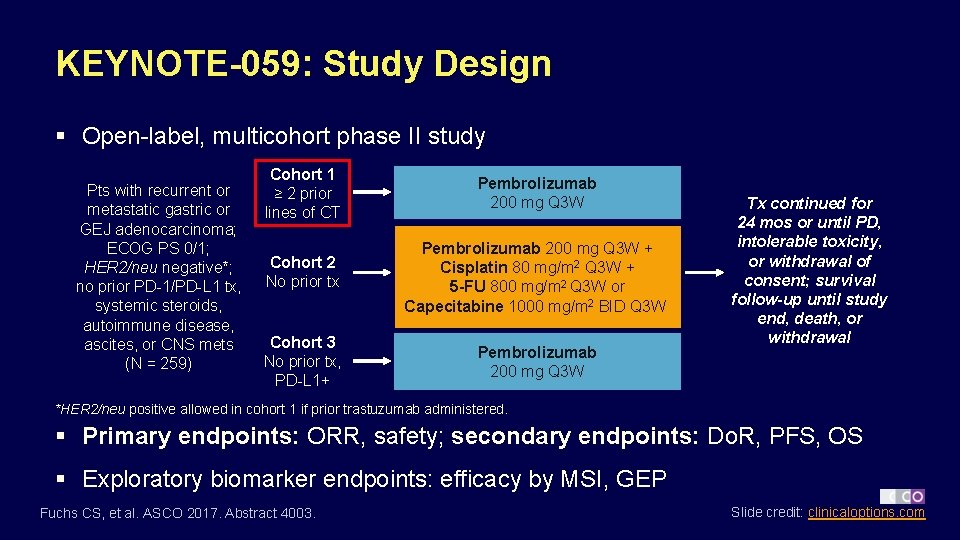
KEYNOTE-059: Study Design § Open-label, multicohort phase II study Pts with recurrent or metastatic gastric or GEJ adenocarcinoma; ECOG PS 0/1; HER 2/neu negative*; no prior PD-1/PD-L 1 tx, systemic steroids, autoimmune disease, ascites, or CNS mets (N = 259) Cohort 1 ≥ 2 prior lines of CT Pembrolizumab 200 mg Q 3 W Cohort 2 No prior tx Pembrolizumab 200 mg Q 3 W + Cisplatin 80 mg/m 2 Q 3 W + 5 -FU 800 mg/m 2 Q 3 W or Capecitabine 1000 mg/m 2 BID Q 3 W Cohort 3 No prior tx, PD-L 1+ Pembrolizumab 200 mg Q 3 W Tx continued for 24 mos or until PD, intolerable toxicity, or withdrawal of consent; survival follow-up until study end, death, or withdrawal *HER 2/neu positive allowed in cohort 1 if prior trastuzumab administered. § Primary endpoints: ORR, safety; secondary endpoints: Do. R, PFS, OS § Exploratory biomarker endpoints: efficacy by MSI, GEP Fuchs CS, et al. ASCO 2017. Abstract 4003. Slide credit: clinicaloptions. com

KEYNOTE-059 (Cohort 1): Baseline Characteristics Characteristic All Pts (N = 259) Characteristic, n (%) Median age, yrs (range) 62 (24 -89) Male, n (%) 198 (76. 4) Geographic region, n (%) §United States §East Asia §Other 124 (47. 9) 34 (13. 1) 101 (39. 0) Prior therapies § 2 § 3 §≥ 4 All Pts (N = 259) 134 (51. 7) 75 (29. 0) 50 (19. 3) Prior surgery for gastric cancer 66 (25. 5) HER 2 positive 63 (24. 3) 148 (57. 1) 109 (42. 1) ECOG PS, n (%) § 0 § 1 107 (41. 3) 151 (58. 3) Primary tumor location, n (%) §Gastric §GEJ PD-L 1 expression §Positive* §Negative 125 (48. 3) 133 (51. 4) *CPS ≥ 1% where CPS is (PD-L 1 staining cells/total tumor cells) x 100. Fuchs CS, et al. ASCO 2017. Abstract 4003. Slide credit: clinicaloptions. com

KEYNOTE-059 (Cohort 1): Response Confirmed Response, % (95% CI) All Pts (N = 259) ORR § Median follow-up: 5. 8 mos (range: 0. 5 -21. 6 mos) 11. 6 (8. 0 -16. 1) CR 2. 3 (0. 9 -5. 0) PR 9. 3 (6. 0 -13. 5) SD 16. 2 (11. 9 -21. 3) PD 56. 0 (49. 7 -62. 1) DCR* 27. 0 (21. 7 -32. 9) *CR + PR + SD ≥ 2 mos. Fuchs CS, et al. ASCO 2017. Abstract 4003. Slide credit: clinicaloptions. com
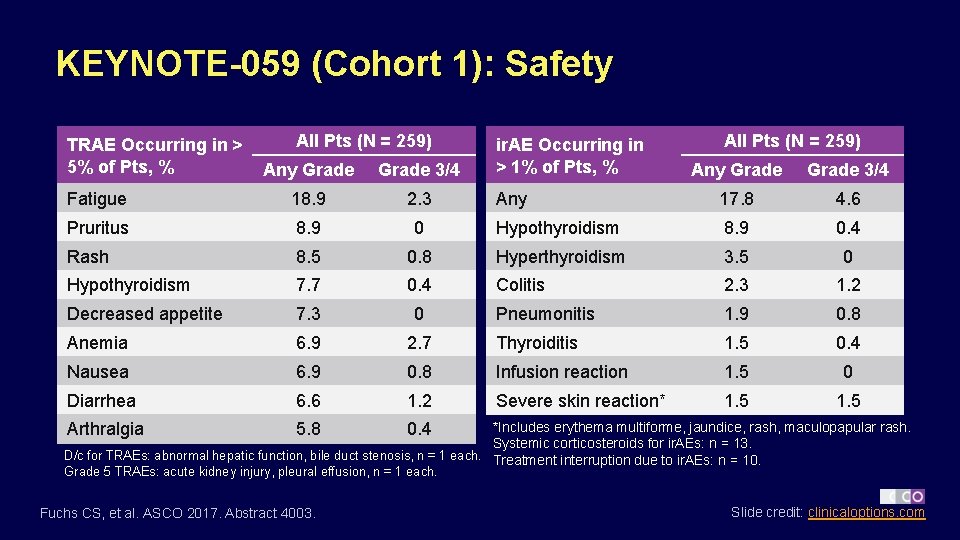
KEYNOTE-059 (Cohort 1): Safety TRAE Occurring in > 5% of Pts, % All Pts (N = 259) Any Grade 3/4 Fatigue 18. 9 2. 3 Pruritus 8. 9 Rash ir. AE Occurring in > 1% of Pts, % All Pts (N = 259) Any Grade 3/4 Any 17. 8 4. 6 0 Hypothyroidism 8. 9 0. 4 8. 5 0. 8 Hyperthyroidism 3. 5 0 Hypothyroidism 7. 7 0. 4 Colitis 2. 3 1. 2 Decreased appetite 7. 3 0 Pneumonitis 1. 9 0. 8 Anemia 6. 9 2. 7 Thyroiditis 1. 5 0. 4 Nausea 6. 9 0. 8 Infusion reaction 1. 5 0 Diarrhea 6. 6 1. 2 Severe skin reaction* 1. 5 *Includes erythema multiforme, jaundice, rash, maculopapular rash. Systemic corticosteroids for ir. AEs: n = 13. D/c for TRAEs: abnormal hepatic function, bile duct stenosis, n = 1 each. Treatment interruption due to ir. AEs: n = 10. Arthralgia 5. 8 0. 4 Grade 5 TRAEs: acute kidney injury, pleural effusion, n = 1 each. Fuchs CS, et al. ASCO 2017. Abstract 4003. Slide credit: clinicaloptions. com
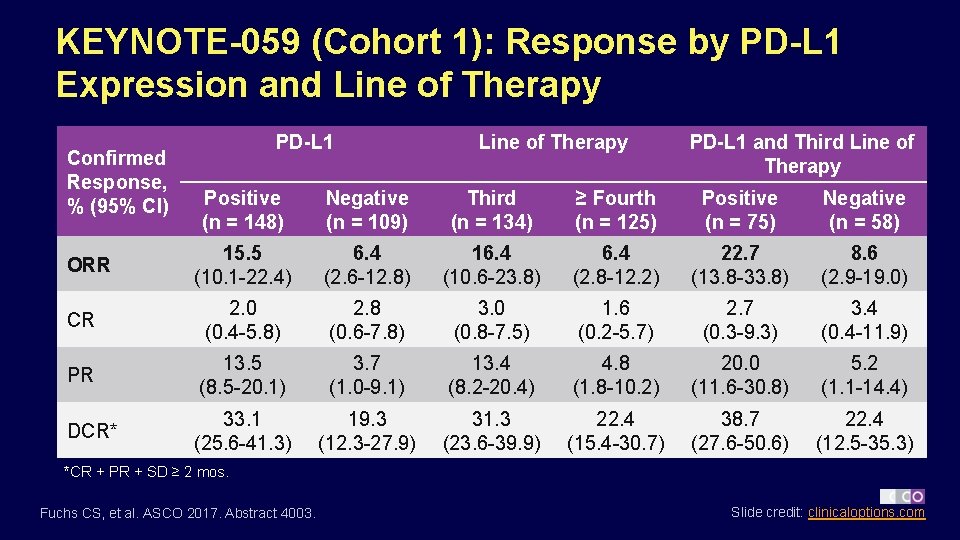
KEYNOTE-059 (Cohort 1): Response by PD-L 1 Expression and Line of Therapy Confirmed Response, % (95% CI) PD-L 1 Line of Therapy PD-L 1 and Third Line of Therapy Positive (n = 148) Negative (n = 109) Third (n = 134) ≥ Fourth (n = 125) Positive (n = 75) Negative (n = 58) 15. 5 (10. 1 -22. 4) 6. 4 (2. 6 -12. 8) 16. 4 (10. 6 -23. 8) 6. 4 (2. 8 -12. 2) 22. 7 (13. 8 -33. 8) 8. 6 (2. 9 -19. 0) CR 2. 0 (0. 4 -5. 8) 2. 8 (0. 6 -7. 8) 3. 0 (0. 8 -7. 5) 1. 6 (0. 2 -5. 7) 2. 7 (0. 3 -9. 3) 3. 4 (0. 4 -11. 9) PR 13. 5 (8. 5 -20. 1) 3. 7 (1. 0 -9. 1) 13. 4 (8. 2 -20. 4) 4. 8 (1. 8 -10. 2) 20. 0 (11. 6 -30. 8) 5. 2 (1. 1 -14. 4) DCR* 33. 1 (25. 6 -41. 3) 19. 3 (12. 3 -27. 9) 31. 3 (23. 6 -39. 9) 22. 4 (15. 4 -30. 7) 38. 7 (27. 6 -50. 6) 22. 4 (12. 5 -35. 3) ORR *CR + PR + SD ≥ 2 mos. Fuchs CS, et al. ASCO 2017. Abstract 4003. Slide credit: clinicaloptions. com

KEYNOTE-059 (Cohort 1): Maximum Change From Baseline in Target Lesion Size 120 Pts With Reduction, % All pts* 42. 4 PD-L 1 positive 47. 3 60 PD-L 1 negative 36. 3 40 *Included pts with measurable disease at BL and ≥ 1 post-BL assessment (n = 223). 100 80 Change From BL (%) PD-L 1 positive PD-L 1 negative PD-L 1 expression unknown 20 0 -20 -40 -60 -80 -100 Fuchs CS, et al. ASCO 2017. Abstract 4003. Reproduced with permission. Slide credit: clinicaloptions. com
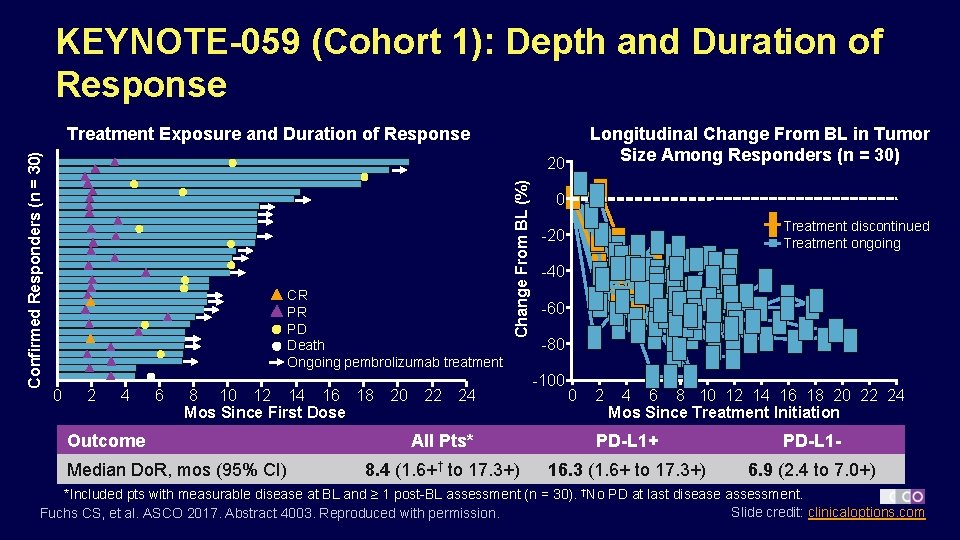
KEYNOTE-059 (Cohort 1): Depth and Duration of Response Longitudinal Change From BL in Tumor Size Among Responders (n = 30) 20 CR PR PD Death Ongoing pembrolizumab treatment 0 2 4 6 8 10 12 14 16 18 Mos Since First Dose Outcome Median Do. R, mos (95% CI) 20 22 Change From BL (%) Confirmed Responders (n = 30) Treatment Exposure and Duration of Response 24 0 Treatment discontinued Treatment ongoing -20 -40 -60 -80 -100 0 2 4 6 8 10 12 14 16 18 20 22 24 Mos Since Treatment Initiation All Pts* PD-L 1+ PD-L 1 - 8. 4 (1. 6+† to 17. 3+) 16. 3 (1. 6+ to 17. 3+) 6. 9 (2. 4 to 7. 0+) *Included pts with measurable disease at BL and ≥ 1 post-BL assessment (n = 30). †No PD at last disease assessment. Slide credit: clinicaloptions. com Fuchs CS, et al. ASCO 2017. Abstract 4003. Reproduced with permission.
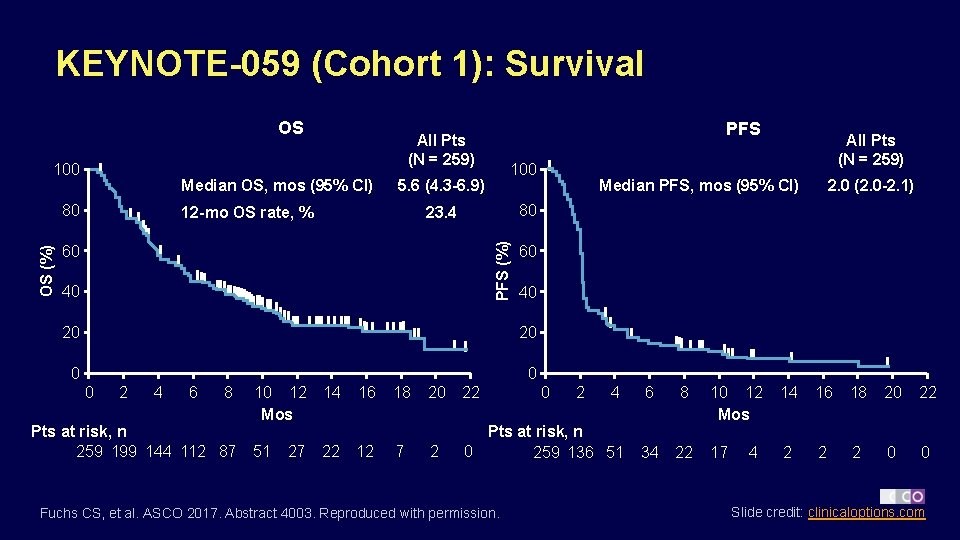
KEYNOTE-059 (Cohort 1): Survival OS 100 Median OS, mos (95% CI) 100 5. 6 (4. 3 -6. 9) 60 40 40 20 0 0 2 4 6 8 Pts at risk, n 259 199 144 112 87 10 12 Mos 14 51 22 27 16 12 18 7 2. 0 (2. 0 -2. 1) Median PFS, mos (95% CI) 60 20 0 All Pts (N = 259) 80 23. 4 12 -mo OS rate, % PFS (%) OS (%) 80 PFS All Pts (N = 259) 20 22 4 6 8 2 Pts at risk, n 0 259 136 51 34 22 Fuchs CS, et al. ASCO 2017. Abstract 4003. Reproduced with permission. 0 2 10 12 Mos 14 16 18 20 22 17 2 2 2 0 0 4 Slide credit: clinicaloptions. com
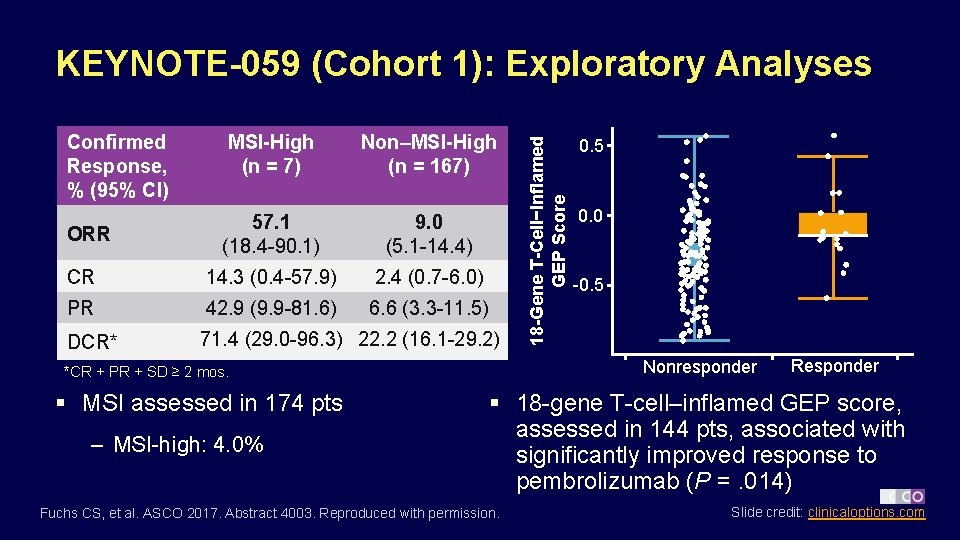
Confirmed Response, % (95% CI) MSI-High (n = 7) Non–MSI-High (n = 167) 57. 1 (18. 4 -90. 1) 9. 0 (5. 1 -14. 4) CR 14. 3 (0. 4 -57. 9) 2. 4 (0. 7 -6. 0) PR 42. 9 (9. 9 -81. 6) 6. 6 (3. 3 -11. 5) DCR* 71. 4 (29. 0 -96. 3) 22. 2 (16. 1 -29. 2) ORR – MSI-high: 4. 0% 0. 5 0. 0 -0. 5 Nonresponder *CR + PR + SD ≥ 2 mos. § MSI assessed in 174 pts 18 -Gene T-Cell−Inflamed GEP Score KEYNOTE-059 (Cohort 1): Exploratory Analyses Responder § 18 -gene T-cell–inflamed GEP score, assessed in 144 pts, associated with significantly improved response to pembrolizumab (P =. 014) Fuchs CS, et al. ASCO 2017. Abstract 4003. Reproduced with permission. Slide credit: clinicaloptions. com

KEYNOTE-059 (Cohort 1): Conclusions § In pts with advanced gastric/GEJ cancer and ≥ 2 prior therapies, pembrolizumab well tolerated with promising antitumor activity, durable responses – ORR: 11. 6%; higher in PD-L 1+ vs PD-L 1 - tumors (15. 5% vs 6. 4%) and MSI-high vs non–MSI-high tumors (57. 1% vs 9. 0%) § Study investigators suggest pembrolizumab as potential therapeutic option for this pt population § Pembrolizumab in earlier-line therapy and in chemotherapy combinations under investigation for advanced gastric/GEJ cancer in ongoing randomized trials Fuchs CS, et al. ASCO 2017. Abstract 4003. Slide credit: clinicaloptions. com

Go Online for More CCO Coverage of ASCO 2017! Short slideset summaries and additional CME-certified analyses with expert faculty commentary on key studies in: § Breast cancer § Hematologic malignancies § Gastrointestinal cancer § Lung cancer § Genitourinary cancer § Skin cancer § Gynecologic cancers clinicaloptions. com/oncology
- Slides: 13