Key ideas in measurement Repeatability Reproducibility Validity Accuracy

Key ideas in measurement Repeatability, Reproducibility, Validity, Accuracy and Precision

Why measure? We measure to determine the value of a quantity (eg. density, temperature, speed) We will judge our measurement based on whether it is ◦ repeatable (do we get the same value if we repeat it), ◦ reproducible (does someone else get the same value if they repeat it), ◦ valid (does it measure the quantity that was intended to be measured) ◦ precise (how close a repeated measurement would be expected to be to the one we made) ◦ accurate (how close a repeated measurement is to an expected or reference value).

Accuracy Accurate results are close to an accepted reference value, sometimes called a “true value”. For example measuring the density of water, the expected value would be 1. 0 g/cm 3 An experiment is expected to be accurate if it is welldesigned to minimise systematic errors and if all equipment is well calibrated. Try to avoid calling the reference value the “correct” value, because it promotes the idea of a correct answer as being the outcome of an experiment.

Precision The more precise a set of values, the closer they are together. The precision of your experiment will be limited by the resolution of your measuring instruments as well as your experimental skill and choice of procedure.

Significant figures and Measurement Uncertainty When a measurement is made, there is a limit to the number of figures that it is meaningful to write down. No matter how precise the instrument, there will always be a limit to the number of meaningful or significant figures that should be recorded. When you record a measurement value, you should include the range of values that it includes. The range of values that is consistent with the measurement is called the uncertainty.

Uncertainty? When a value of a quantity is obtained experimentally, the value should always be quoted with a range or uncertainty. For example, the specific heat capacity of aluminium was found to be (880 ± 10)J/Kg/K This is the range of values which is consistent with your measurement. The uncertainty allows you to quantitatively compare measurements. If the range of two measurements overlap, then they are the same, within the precision of your experiment.

Systematic and Random effects Systematic effects in your experiment will reduce the accuracy of your measurements. Systematic effects will consistently shift the value of your measurements to either a smaller or higher value. Random effects in your experiment will reduce the precision of your results. By repeating your measurements, the random effects will average out and you can increase your confidence that the average of your set of measurements is the average of the quantity you intended to measure.

Analysing your data 1. Uncertainties 2. Trendlines and quantitative relationships

You should be able to explain/describe: Why measurements are always accompanied by an uncertainty Sources of uncertainty in measurement: ◦ Instrumental ◦ Method Absolute uncertainty and relative uncertainty Strategies for reducing uncertainty in practical work

Instrumental Uncertainty Length of green line = 9. 60 cm ± 0. 05 cm Is instrumental uncertainty the only uncertainty present in my measurement?

A series of measurements: 9. 60 cm ± 0. 05 cm 10. 10 cm ± 0. 05 cm 9. 55 cm ± 0. 05 cm 10. 50 cm ± 0. 05 cm The mean value is 9. 93 cm Does this mean that I expect my next measurement to be within 0. 05 cm of 9. 93 cm?

What we expect For each data point, you will carry out a number of trials and find the expected value for that quantity. Using the ruler example we now calculate the expected value for the measurement. The uncertainty will be the average difference between the mean value and the maximum and minimum values obtained. In previous example, the expected value becomes 9. 9 cm ± 0. 5 cm
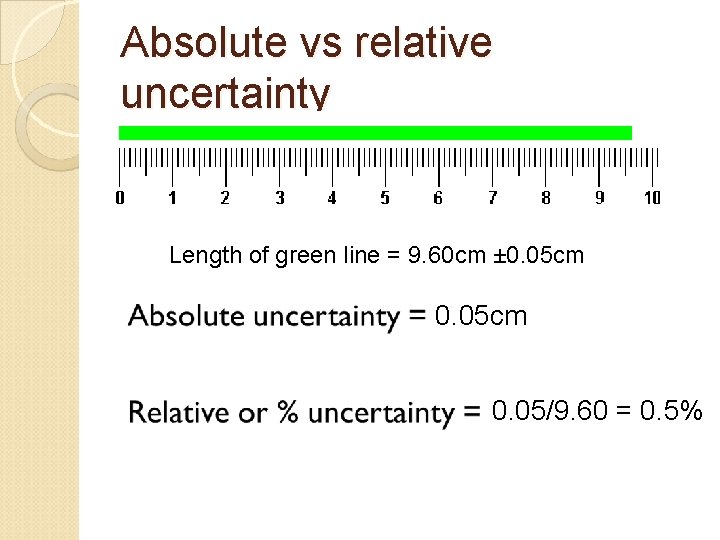
Absolute vs relative uncertainty Length of green line = 9. 60 cm ± 0. 05 cm 0. 05/9. 60 = 0. 5%

Getting really fancy – finding the uncertainty of quantities calculated from data Example: area of a piece of paper length = 29. 0 ± 0. 5 cm, width = 20. 0 ± 0. 3 cm Step 1: Take the largest possible value of the measurement and calculate the derived quantity Areamax = 29. 5× 20. 3 = 598. 85 cm 2 Step II: Take the smallest possible value of the measurement and calculate the derived quantity. Areamin = 28. 5× 19. 7 = 561. 45 cm 2 Step III: The uncertainty in the derived quantity is the difference between the two values, divided by 2. Area = 580± 20 cm 2 Uncertainties should have no more than 1 sig

Guidelines for fitting data 1. Postulate an expected relationship Is your dependent variable proportional to your independent variable? Is your dependent variable inversely proportional to your independent variable? Is your dependent variable proportional to the square of the independent

Fit your expected trendline. II. Fit a trendline with the same form as your expected relationship to your data. Do not simply get Xcel to fit a trendline to your data. Record the equation of the trendline and the R 2 value. The R 2 value is a measure of how well your data fits the trendline. 1. 0 is perfect, 0. 0 is not at all! III. Compare your expected trendline with other possible trendlines.

Trendlines and information The following features of trendlines usually reveal important properties about your data: 1. 2. the gradient the intercept For example, if the force on a spring is measured for different amounts of stretch of the spring, then the gradient is the spring constant. By calculating the gradient, you can find the value of the spring constant for your spring. Like all values, it will also have an uncertainty.
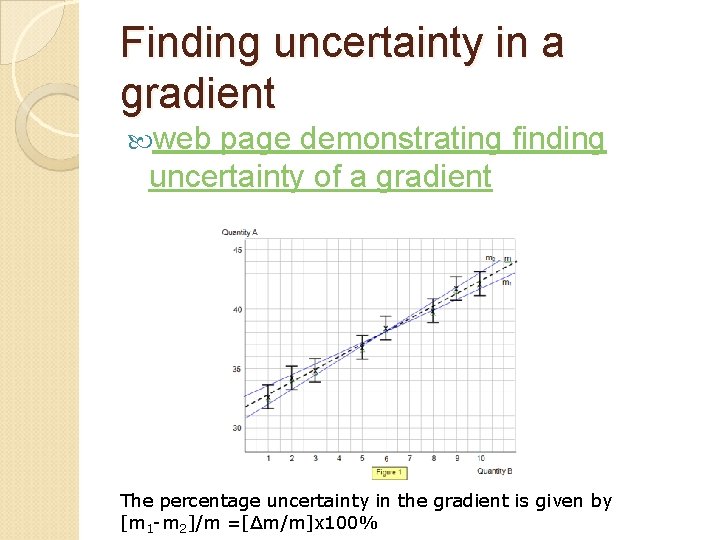
Finding uncertainty in a gradient web page demonstrating finding uncertainty of a gradient The percentage uncertainty in the gradient is given by [m 1 -m 2]/m =[Δm/m]x 100%
- Slides: 18