Key Concepts An organisms metabolism transforms matter and

Key Concepts • An organisms metabolism transforms matter and energy, subject to the laws of thermodynamics • The free-energy change of a reaction tells us whether or not the reaction occurs spontaneously • ATP powers cellular work by coupling exergonic reactions to endergonic reactions • Enzymes speed up metabolic reactions by lowering energy barriers • Regulation of enzyme activity helps control metabolism

AP BIG IDEAS • All living organisms require a constant input of free energy, which is used for organization, growth, and reproduction (BIG IDEA 2). Common to life are core processes that depend on enzymes to regulate the chemical reactions that drive the metabolic activities of life (BIG IDEA 1 & 4). • The concepts of metabolism that you learn in this chapter will help you understand how matter and energy flow during life’s processes and how that flow is regulated.

• The totality of an organism’s chemical reactions is called metabolism. Metabolism is an emergent property of life that arises from orderly interactions between molecules. • Some metabolic pathways release energy by breaking down complex molecules to simpler compounds; These degradative processes are called catabolic pathways. A major pathway of catabolism is cellular respiration, in which the sugar glucose and other organic fuels are broken down in the presence of oxygen to carbon dioxide and water. • Hydrolysis, digestion • Anabolic pathways consume energy to build complicated molecules from simpler ones; they are sometimes called biosynthetic pathways. Examples of anabolism are the synthesis of a protein from amino acids. • Dehydration synthesis
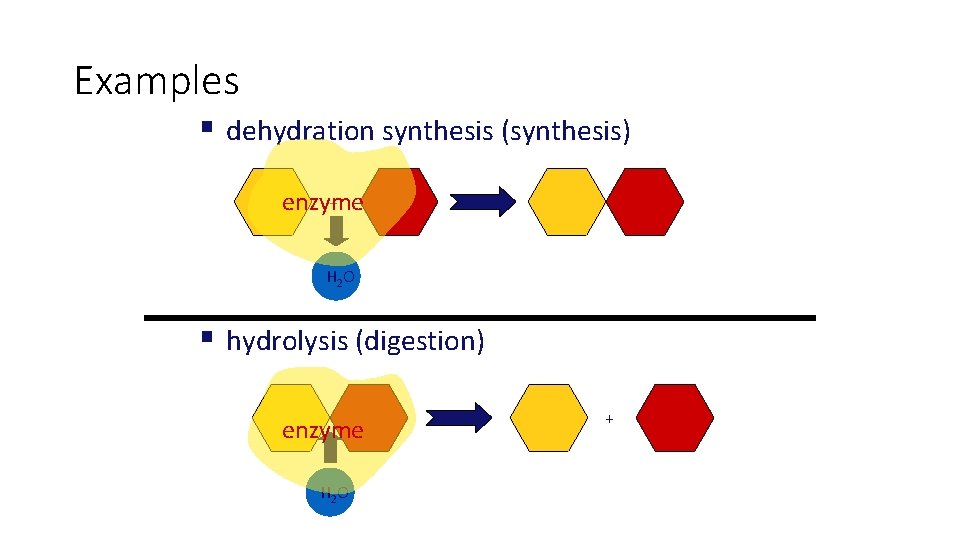
Examples § dehydration synthesis (synthesis) + enzyme H 2 O § hydrolysis (digestion) enzyme H 2 O +
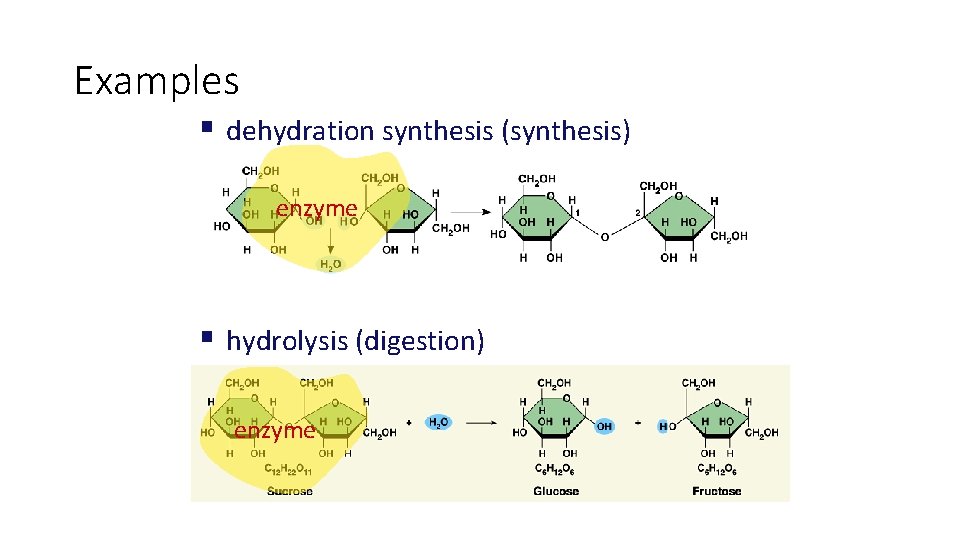
Examples § dehydration synthesis (synthesis) enzyme § hydrolysis (digestion) enzyme

The study of the energy transformations that occur in a collection of matter is called thermodynamics. • According to the first law of thermodynamics, the energy of the universe is constant: Energy can be transferred and transformed, but it cannot be created or destroyed. If energy cannot be created or destroyed, why can’t organisms simply recycle their energy over and over again? • It turns out that during every energy transfer or transformation, some energy becomes unavailable to do work. In most energy transformations, energy is converted to thermal energy and released as heat.

• The second law of thermodynamics: Every energy transfer or transformation increases the entropy of the universe. For example, entropy is evident in the gradual decay of an unmaintained building. Much of the increasing entropy in the universe is less obvious, however, because it takes the form of increasing amounts of heat and less ordered forms of matter. • 144

• Gibbs free energy of a system, symbolized by the letter G, is the portion of a system’s energy that can perform work when temperature and pressure are uniform throughout the system, as in a living cell. • When a process occurs spontaneously, we can be sure that G is negative. This is because the process involves a loss of free energy during the change from initial state to final state. G = G final state - G initial state • We can think of free energy as a measure of a system’s instability – it’s tendency to change to a more stable state. Unstable systems (higher G) tend to change so that they become more stable (lower G).

Chemical reactions & energy § Some chemical reactions release energy uexergonic digesting molecules= udigesting polymers LESS organization= lower energy state uhydrolysis = catabolism u“Spontaneous” = energetically favorable § Some chemical reactions require input of energy uendergonic ubuilding polymers udehydration synthesis = anabolism u“non-spontaneous” building molecules= MORE organization= higher energy state
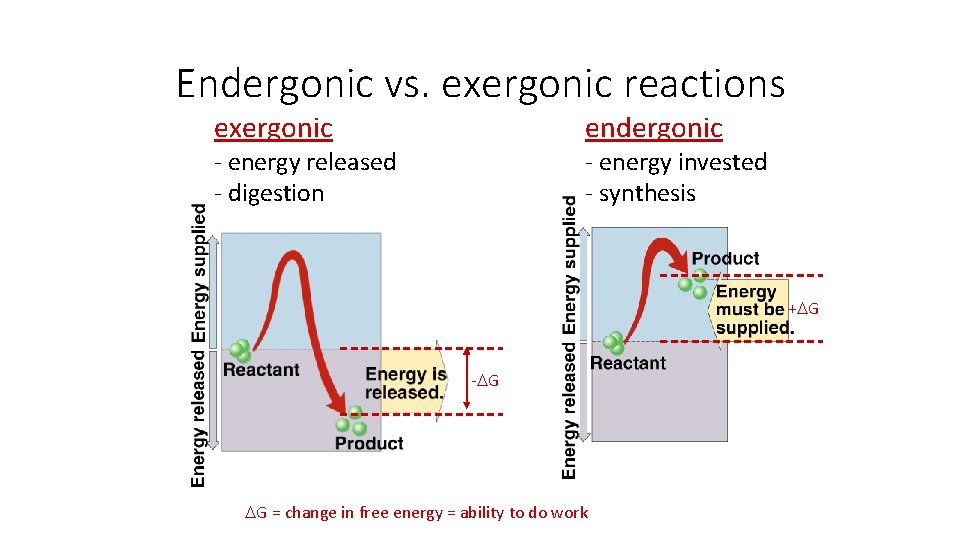
Endergonic vs. exergonic reactions exergonic endergonic - energy invested - synthesis - energy released - digestion + G - G G = change in free energy = ability to do work

• For every ole of glucose broken down by respiration, 686 kcal of energy are made available for work. • If G = -686 kcal/mol for respiration, which converts glucose and oxygen to carbon dioxide and water, then the reverse process – the conversion of carbon dioxide and water to glucose and oxygen – must be strongly endergonic, with G = +686 kcal/mol. Plants get the required energy from the environment by capturing light and converting its energy to chemical energy. Next, in a long series of exergonic steps, they gradually spend that chemical energy to assemble glucose molecules.

A cell does three main kinds of work: • Mechanical work, such as beating of cilia, the contraction of muscle cells, and the movement of chromosomes during cell reproduction. • Transport work, the pumping of substances across membranes against the direction of spontaneous movement • Chemical work, the pushing of endergonic reactions that would not occur spontaneously, such as the synthesis of polymers from monomers • A key feature in the way cells manage their energy resources to do this work is energy coupling, the use of an exergonic process to drive an endergonic one. ATP is responsible for mediating most energy coupling in cells, and in most cases, it is the immediate source of energy that powers cellular work.
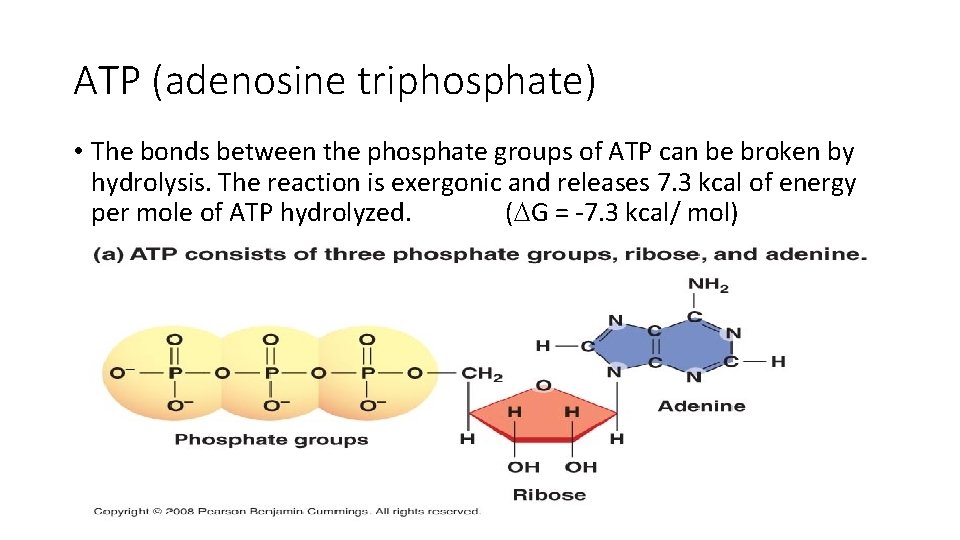
ATP (adenosine triphosphate) • The bonds between the phosphate groups of ATP can be broken by hydrolysis. The reaction is exergonic and releases 7. 3 kcal of energy per mole of ATP hydrolyzed. ( G = -7. 3 kcal/ mol)

• With the help of specific enzymes, the cell is able to use the energy released by ATP hydrolysis directly to drive chemical reactions that, by themselves, are endergonic. • If the G of an endergonic reaction is less than the amount of energy released by ATP hydrolysis, then the two reactions can be coupled so that, overall, the coupled reactions are exergonic. This usually involves phosphorylation, the transfer of a phosphate group from ATP to some other molecule, such as the reactant. • Phosphorylation and dephosphorylation promote crucial protein shape changes during many other important cellular processes as well.
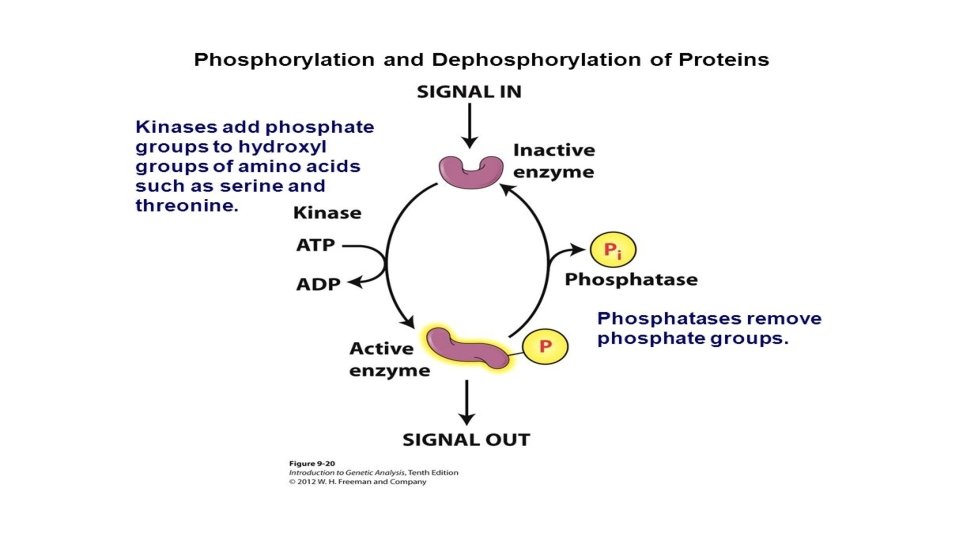
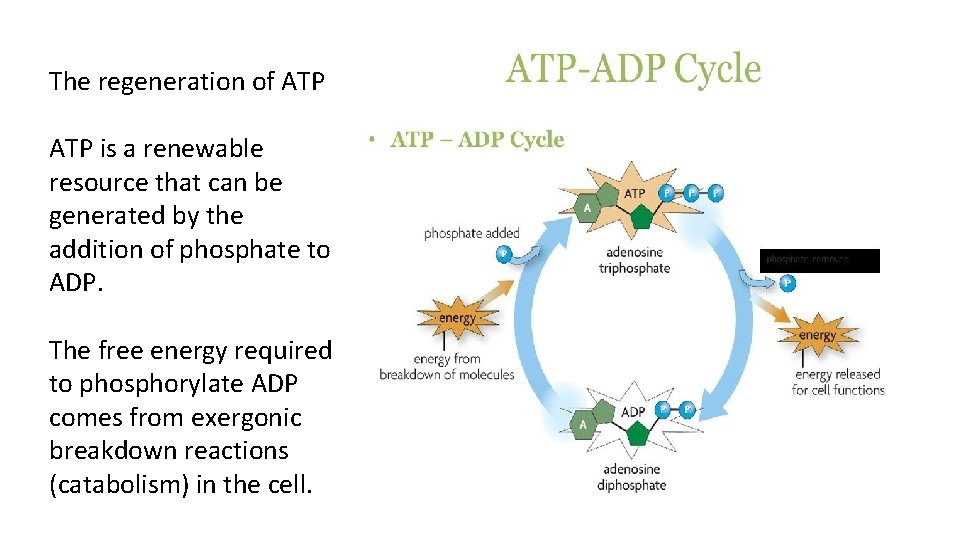
The regeneration of ATP is a renewable resource that can be generated by the addition of phosphate to ADP. The free energy required to phosphorylate ADP comes from exergonic breakdown reactions (catabolism) in the cell.
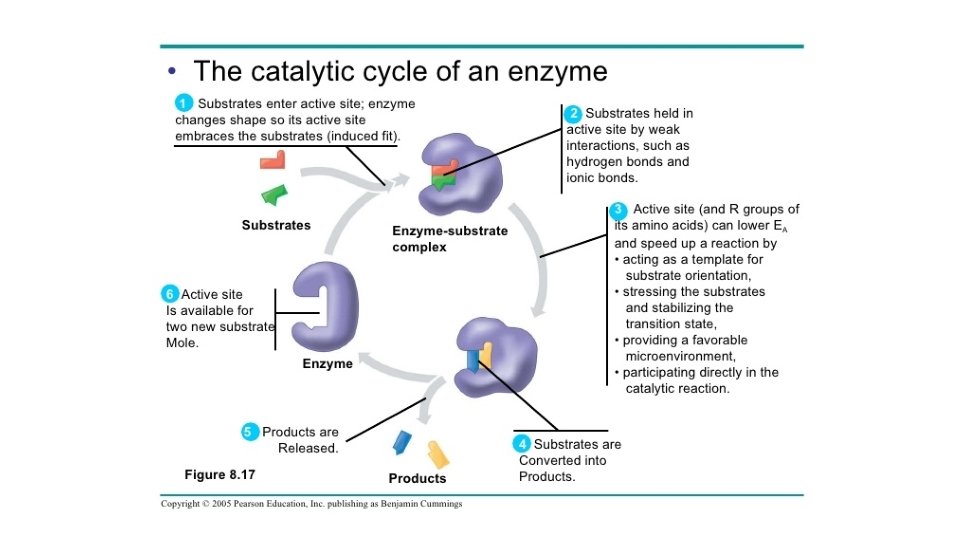
• A spontaneous chemical reaction occurs without any requirement for outside energy, but it may occur so slowly that it will sit for years without any noticeable change. Enzymes speed up metabolic reactions by lowering energy barriers • An enzyme is a macromolecule that acts as a catalyst, a chemical agent that speeds up a reaction without being consumed by the reaction. For now, we are focusing on proteins that are enzymes (Some RNA molecules, called ribozymes, can function as enzymes)
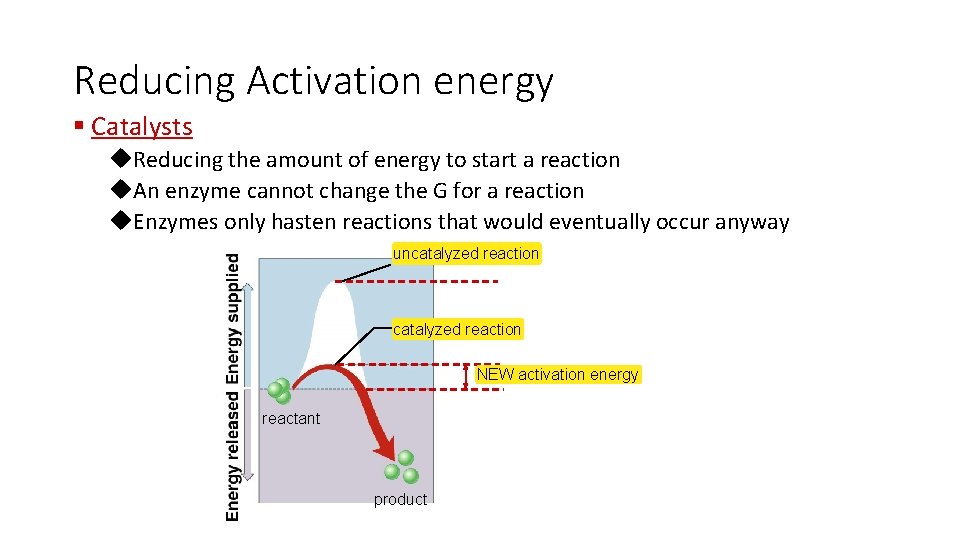
Reducing Activation energy § Catalysts u. Reducing the amount of energy to start a reaction u. An enzyme cannot change the G for a reaction u. Enzymes only hasten reactions that would eventually occur anyway uncatalyzed reaction NEW activation energy reactant product
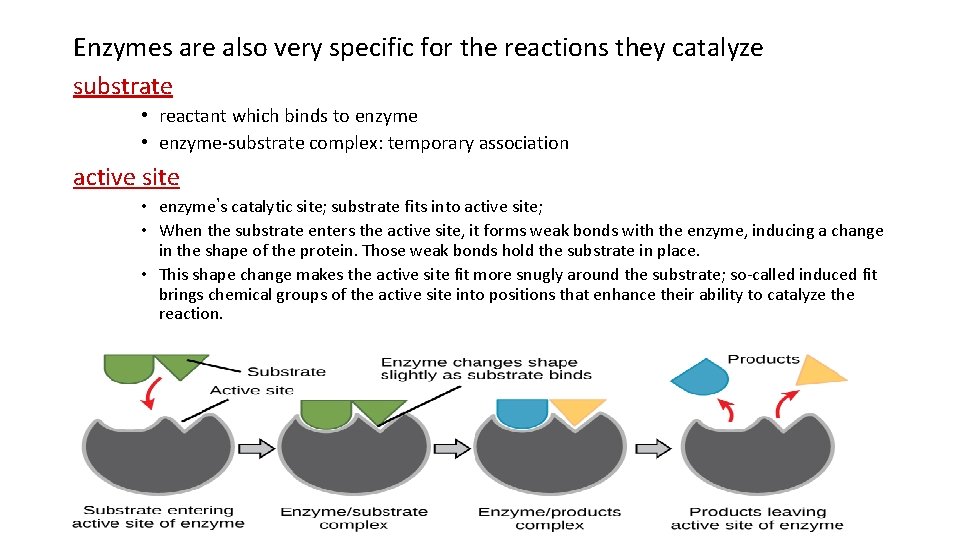
Enzymes are also very specific for the reactions they catalyze substrate • reactant which binds to enzyme • enzyme-substrate complex: temporary association active site • enzyme’s catalytic site; substrate fits into active site; • When the substrate enters the active site, it forms weak bonds with the enzyme, inducing a change in the shape of the protein. Those weak bonds hold the substrate in place. • This shape change makes the active site fit more snugly around the substrate; so-called induced fit brings chemical groups of the active site into positions that enhance their ability to catalyze the reaction.

Properties of enzymes § Reaction specific ueach enzyme works with a specific substrate § chemical fit between active site & substrate w H bonds & ionic bonds § Not consumed in reaction usingle enzyme molecule can catalyze thousands or more reactions per second § enzymes unaffected by the reaction § Affected by cellular conditions uany condition that affects protein structure § temperature, p. H, salinity
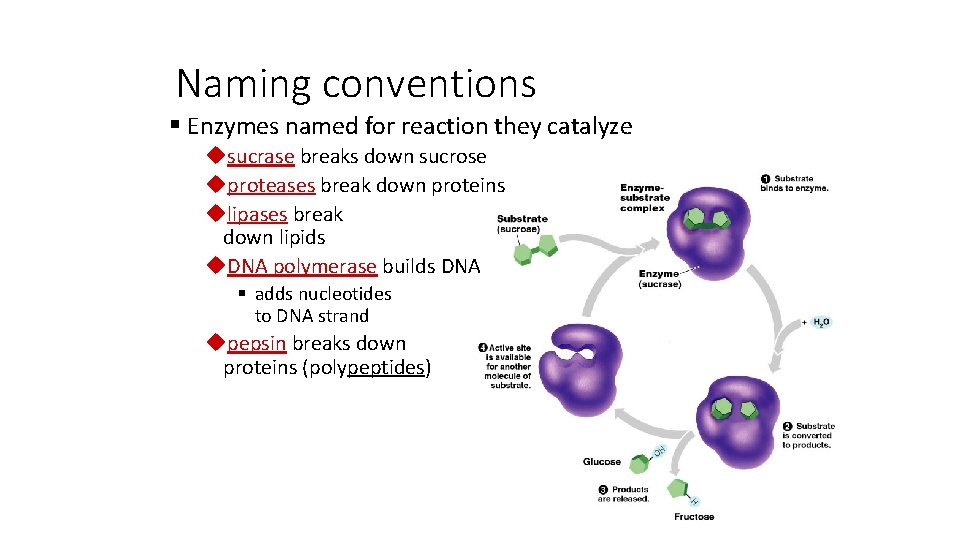
Naming conventions § Enzymes named for reaction they catalyze usucrase breaks down sucrose uproteases break down proteins ulipases break down lipids u. DNA polymerase builds DNA § adds nucleotides to DNA strand upepsin breaks down proteins (polypeptides)

Induced fit model • More accurate model of enzyme action (more accurate than lock & key model) • 3 -D structure of enzyme fits substrate • substrate binding cause enzyme to change shape leading to a tighter fit • “conformational change” • bring chemical groups in position to catalyze reaction
Factors Affecting Enzyme Function § Enzyme concentration § Substrate concentration § Temperature § p. H § Salinity § Activators § Inhibitors catalase
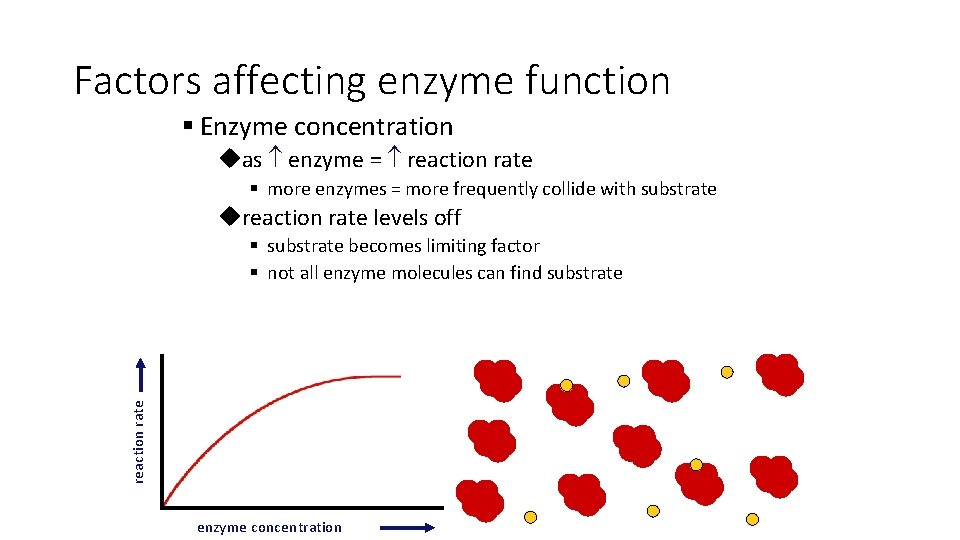
Factors affecting enzyme function § Enzyme concentration uas enzyme = reaction rate § more enzymes = more frequently collide with substrate ureaction rate levels off reaction rate § substrate becomes limiting factor § not all enzyme molecules can find substrate enzyme concentration
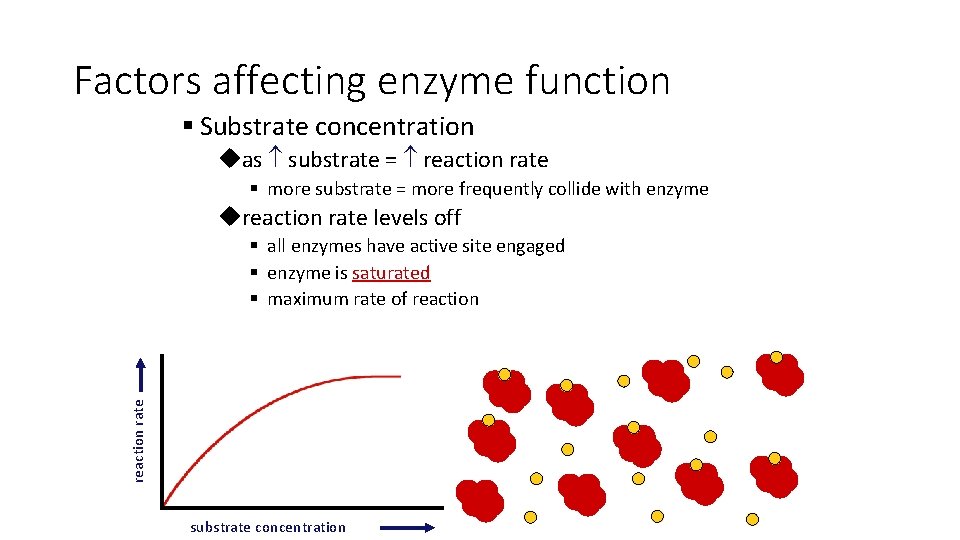
Factors affecting enzyme function § Substrate concentration uas substrate = reaction rate § more substrate = more frequently collide with enzyme ureaction rate levels off reaction rate § all enzymes have active site engaged § enzyme is saturated § maximum rate of reaction substrate concentration

Factors affecting enzyme function • Temperature • Optimum T° • greatest number of molecular collisions • human enzymes = 35°- 40°C • body temp = 37°C • Heat: increase beyond optimum T° • increased energy level of molecules disrupts bonds in enzyme & between enzyme & substrate • H, ionic = weak bonds • denaturation = lose 3 D shape (3° structure) • Cold: decrease T° • molecules move slower • decrease collisions between enzyme & substrate
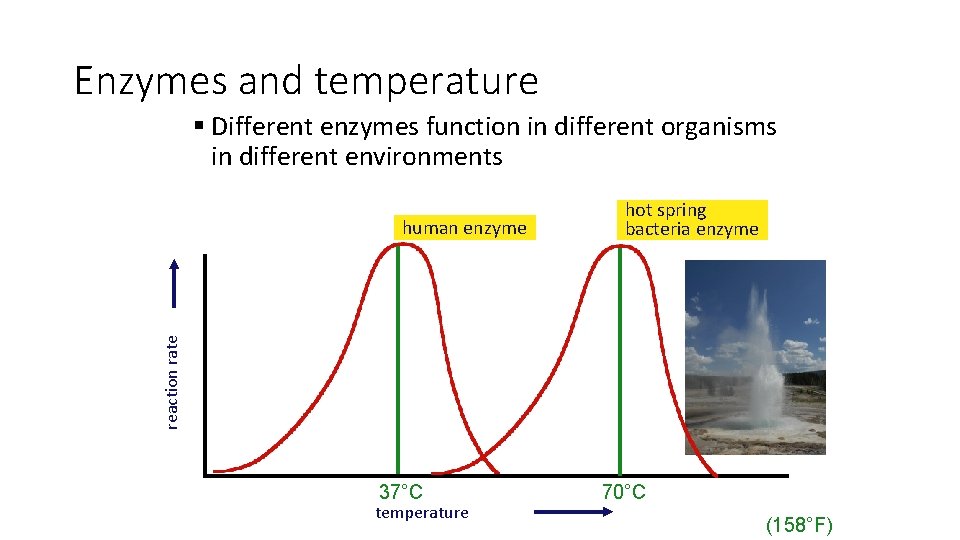
Enzymes and temperature § Different enzymes function in different organisms in different environments reaction rate human enzyme hot spring bacteria enzyme 37°C temperature 70°C (158°F)
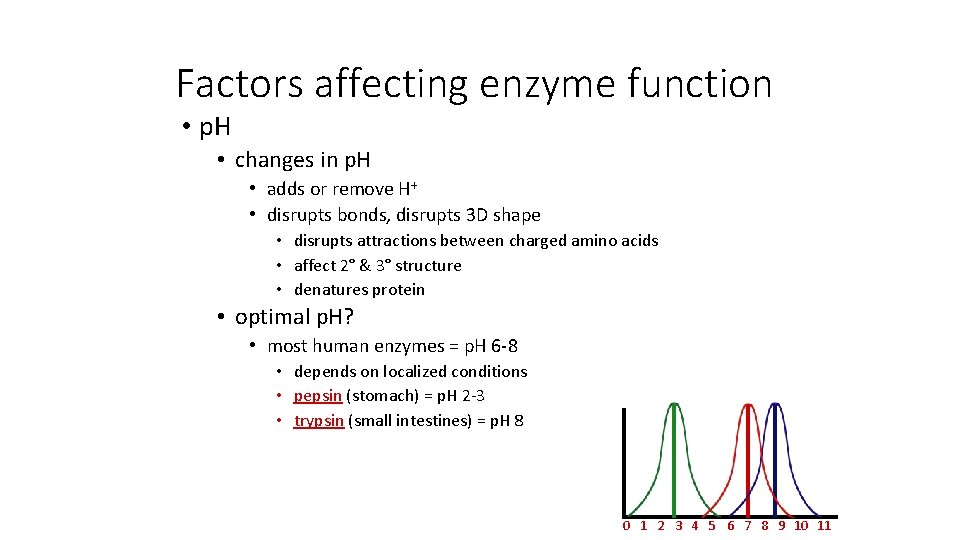
Factors affecting enzyme function • p. H • changes in p. H • adds or remove H+ • disrupts bonds, disrupts 3 D shape • disrupts attractions between charged amino acids • affect 2° & 3° structure • denatures protein • optimal p. H? • most human enzymes = p. H 6 -8 • depends on localized conditions • pepsin (stomach) = p. H 2 -3 • trypsin (small intestines) = p. H 8 0 1 2 3 4 5 6 7 8 9 10 11

Factors affecting enzyme function • Salt concentration • changes in salinity • adds or removes cations (+) & anions (–) • disrupts bonds, disrupts 3 D shape • disrupts attractions between charged amino acids • affect 2° & 3° structure • denatures protein • enzymes intolerant of extreme salinity • Dead Sea is called dead for a reason!

Compounds which help enzymes § Activators ucofactors Fe in hemoglobin § non-protein, small inorganic compounds & ions w Mg, K, Ca, Zn, Fe, Cu w bound within enzyme molecule ucoenzymes § non-protein, organic molecules w bind temporarily or permanently to enzyme near active site § many vitamins w NAD (niacin; B 3) w FAD (riboflavin; B 2) w Coenzyme A Mg in chlorophyll

Compounds which regulate enzymes § Inhibitors umolecules that reduce enzyme activity ucompetitive inhibition unoncompetitive inhibition uirreversible inhibition ufeedback inhibition
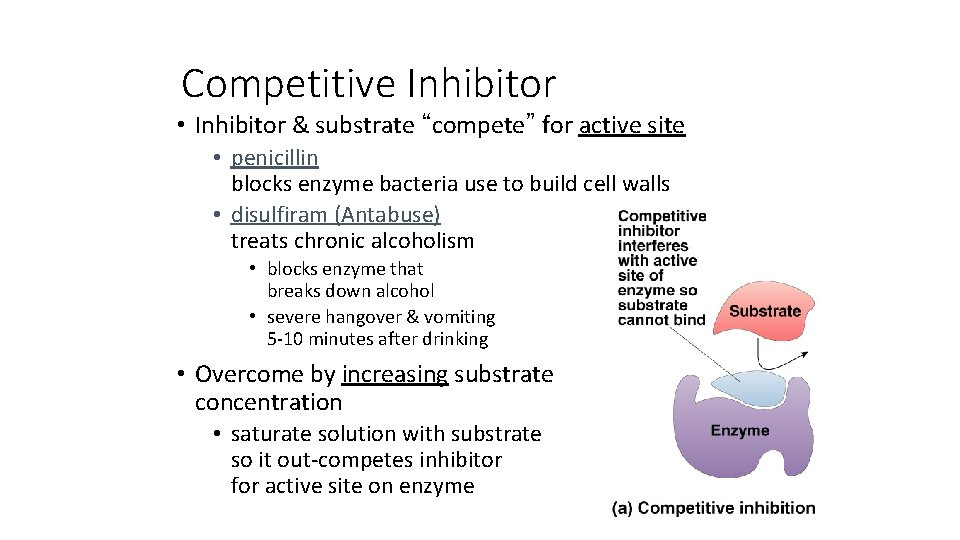
Competitive Inhibitor • Inhibitor & substrate “compete” for active site • penicillin blocks enzyme bacteria use to build cell walls • disulfiram (Antabuse) treats chronic alcoholism • blocks enzyme that breaks down alcohol • severe hangover & vomiting 5 -10 minutes after drinking • Overcome by increasing substrate concentration • saturate solution with substrate so it out-competes inhibitor for active site on enzyme

Non-Competitive Inhibitor • Inhibitor binds to site other than active site • allosteric inhibitor binds to allosteric site • causes enzyme to change shape • conformational change • active site is no longer functional binding site • keeps enzyme inactive • some anti-cancer drugs inhibit enzymes involved in DNA synthesis • stop DNA production • stop division of more cancer cells • cyanide poisoning irreversible inhibitor of Cytochrome C, an enzyme in cellular respiration • stops production of ATP

Irreversible inhibition • Inhibitor permanently binds to enzyme • competitor • permanently binds to active site • allosteric • permanently binds to allosteric site • permanently changes shape of enzyme • nerve gas, sarin, many insecticides (malathion, parathion…) • cholinesterase inhibitors • doesn’t breakdown the neurotransmitter, acetylcholine

Allosteric regulation § Conformational changes by regulatory molecules uinhibitors § keeps enzyme in inactive form uactivators § keeps enzyme in active form Conformational changes Allosteric regulation
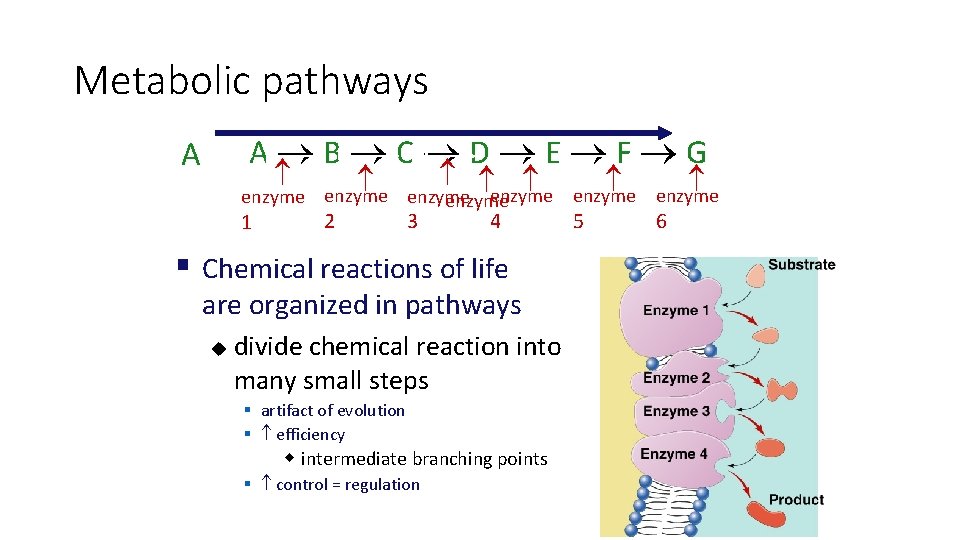
Metabolic pathways A AB BC CD DE EF F G enzyme enzyme 1 2 3 4 § Chemical reactions of life are organized in pathways u divide chemical reaction into many small steps § artifact of evolution § efficiency w intermediate branching points § control = regulation 5 6
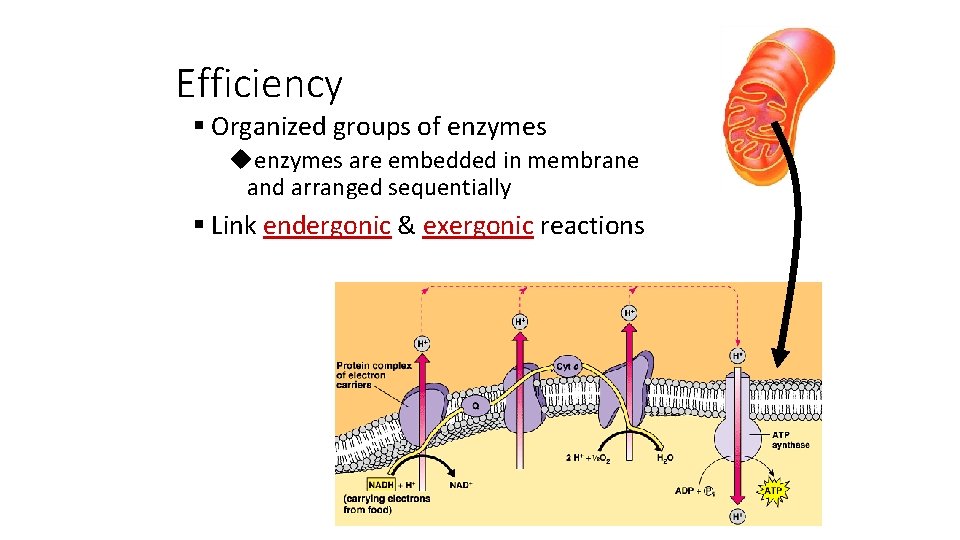
Efficiency § Organized groups of enzymes uenzymes are embedded in membrane and arranged sequentially § Link endergonic & exergonic reactions
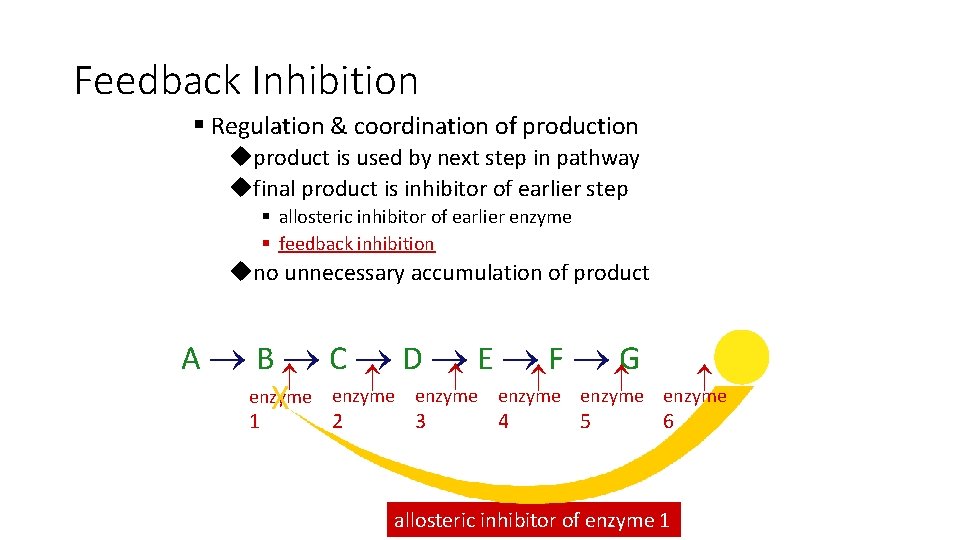
Feedback Inhibition § Regulation & coordination of production uproduct is used by next step in pathway ufinal product is inhibitor of earlier step § allosteric inhibitor of earlier enzyme § feedback inhibition uno unnecessary accumulation of product X A B C D E F G enzyme enzyme 1 2 3 4 5 6 allosteric inhibitor of enzyme 1
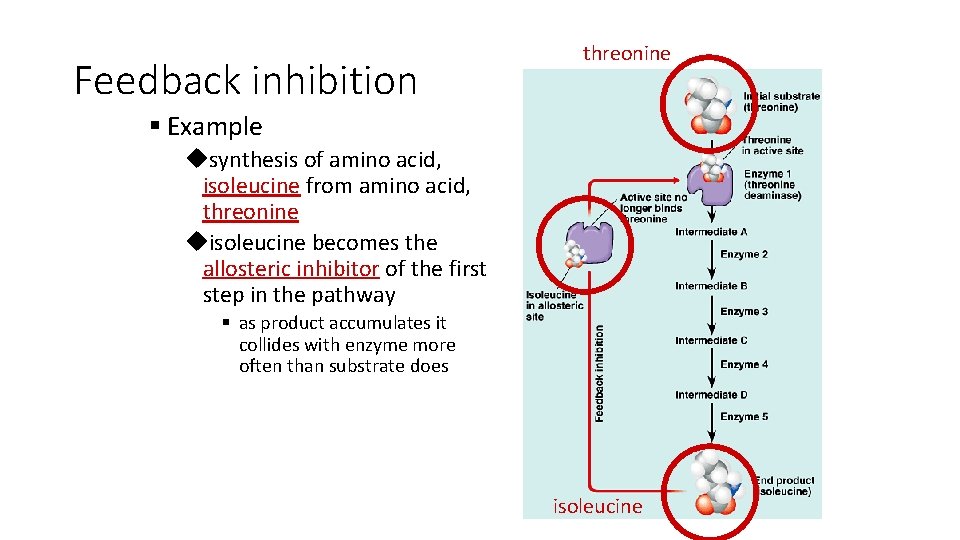
Feedback inhibition threonine § Example usynthesis of amino acid, isoleucine from amino acid, threonine uisoleucine becomes the allosteric inhibitor of the first step in the pathway § as product accumulates it collides with enzyme more often than substrate does isoleucine

The Evolution of Enzymes • Most enzymes are proteins and proteins are encoded by genes. • A permanent change in a gene, known as a mutation, can result in a protein with one or more changed amino acids. • In the case of an enzyme, if the changed amino acids are in the active site or some other crucial region, the altered enzyme might have different activity or might bind to a different substrate. Under environmental conditions where the new function benefits the organism, natural selection would tend to favor the mutated form of the gene causing it to persist in the population.

Localization of Enzymes Within the Cell • The cell is not just a bag of chemicals with thousands of different enzymes and substrates in a random mix. • The cell is compartmentalized, and cellular structures help bring order to metabolic pathways. • Some enzymes and enzyme complexes have fixed locations within the cell and act as structural components of particular membranes. Others are in solution within particular membrane-enclosed eukaryotic organelles, each with its own internal chemical environment. For example, in eukaryotic cells, the enzymes for cellular respiration reside in specific locations within mitochondria.

1. Choose the pair of terms that correctly completes this sentence: Catabolism is to anabolism as _______ is to _______. a. exergonic; spontaneous b. exergonic; endergonic c. free energy; entropy d. work; energy
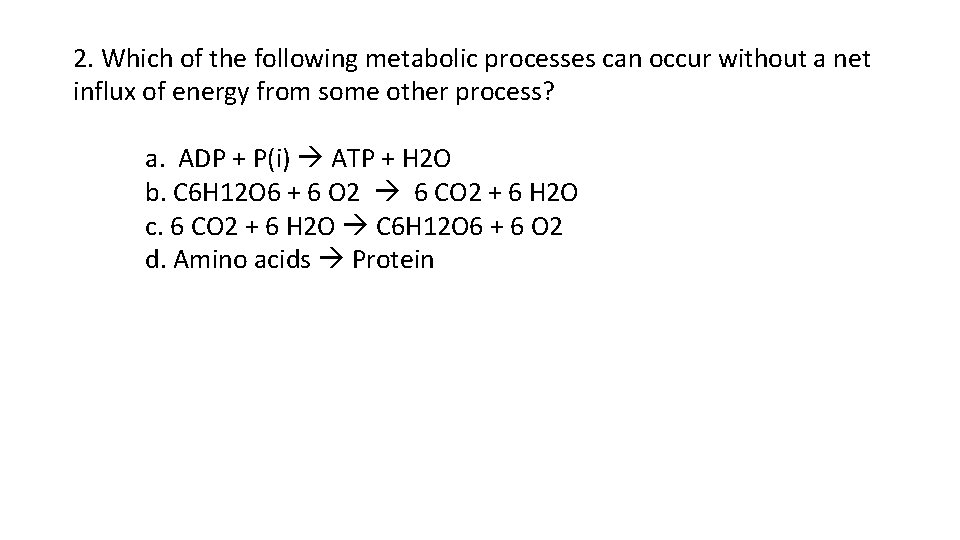
2. Which of the following metabolic processes can occur without a net influx of energy from some other process? a. ADP + P(i) ATP + H 2 O b. C 6 H 12 O 6 + 6 O 2 6 CO 2 + 6 H 2 O c. 6 CO 2 + 6 H 2 O C 6 H 12 O 6 + 6 O 2 d. Amino acids Protein

3. If an enzyme in solution is saturated with substrate, the most effective way to obtain a faster yield of products is to a. add more of the enzyme b. heat the solution to 90 degrees Celsius c. add more substrate d. add a noncompetitive inhibitor

4. Some bacteria are metabolically active in hot springs because a. they are able to maintain a lower internal temperature b. high temperatures make catalysis unnecessary c. their enzymes have high optimal temperatures d. their enzymes are completely insensitive to temperature

5. If an enzyme is added to a solution where its substrate and product are in equilibrium, what will occur? a. additional substrate will be formed b. the reaction will change from endergonic to exergonic c. the free energy of the system will change d. nothing; the reaction will stay at equilibrium
- Slides: 47