Kesuburan tanah Lanjutan Sub Topik Besi Fe dan

Kesuburan tanah Lanjutan Sub Topik: Besi (Fe) dan Seg (Zn) Oleh: Dr. Ir. Hamidah Hanum, MP Sekolah Pascasarjana USU

UNSUR MIKRO TANAH § § § Fungsi fisiologis : dlm reaksi enzimatik Dlm tanaman : < 100 mg/g Diserap tanaman dlm btk ion dan senyawa kompleks organik alami/sintetik Bentuk dlm tnh: ion dlm larutan tnh, senyawa garam (mineral primer dan sekunder), kompleks dgn senyawa organik, bentuk organik dlm biomass mikroba Sumber : litosfer, batuan, mineral, pupuk, bahan organik Kadar dlm tnh : rendah Fe : 104 – 105 ppm Cl : 50 ppm Zn : 10 – 300 ppm Mo : 10 ppm Mn : 200 -300 ppm B : 7 – 10 ppm Cu : 10 -80 ppm
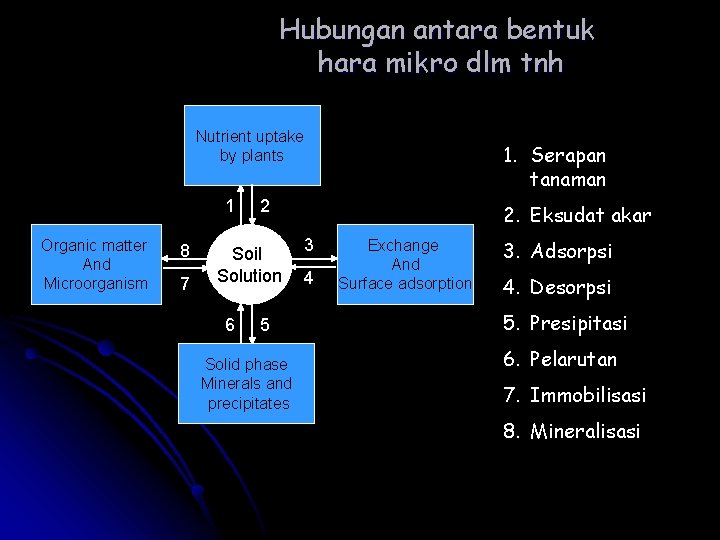
Hubungan antara bentuk hara mikro dlm tnh Nutrient uptake by plants 1 2 Organic matter And Microorganism 8 7 Exchange 3 Soil And Solution 4 Surface adsorption 6 5 Solid phase Minerals and precipitates 1. Serapan tanaman 2. Eksudat akar 3. Adsorpsi 4. Desorpsi 5. Presipitasi 6. Pelarutan 7. Immobilisasi 8. Mineralisasi
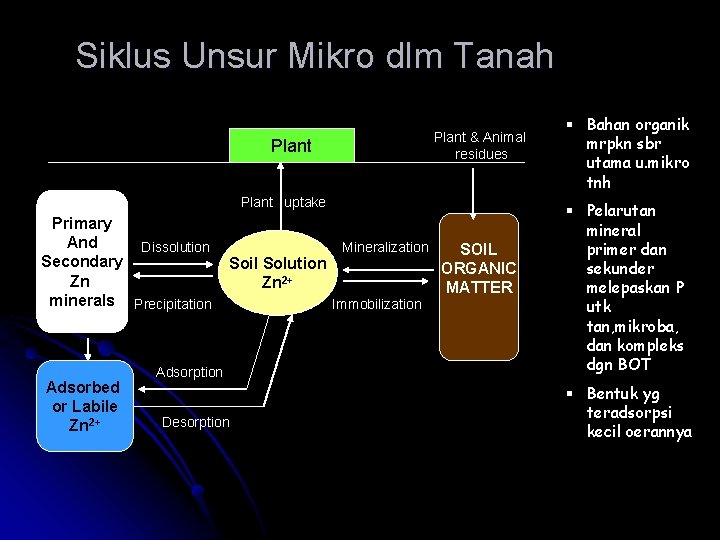
Siklus Unsur Mikro dlm Tanah Plant & Animal residues Plant uptake Primary And Dissolution Secondary Zn minerals Precipitation Adsorbed or Labile Zn 2+ Soil Solution Zn 2+ Adsorption Desorption Mineralization Immobilization SOIL ORGANIC MATTER § Bahan organik mrpkn sbr utama u. mikro tnh § Pelarutan mineral primer dan sekunder melepaskan P utk tan, mikroba, dan kompleks dgn BOT § Bentuk yg teradsorpsi kecil oerannya

Peranan Fe Function and mobility of Fe l l Fe-utake as Fe 2+ and Fe 3+ Iron is required for electron transport in photosynthesis and is a constituent of iron porphyrins and ferredoxins, both of which are essential components in the light phase of photosynthesis. Fe is an important electron acceptor in redox reactions and an activator for several enzymes (e. g. , catalase, succinic dehydrogenase, and aconitase), but inhibits K absorption. On alkaline soils, immobilization of Fe in plant roots occurs because of Fe precipitation. Because Fe is not mobile within rice plants, young leaves are affected first. Fe deficiency symptoms and effects on growth Interveinal yellowing and chlorosis of emerging leaves. § Whole leaves become chlorotic and then very pale. § The entire plant becomes chlorotic and dies if Fe deficiency is very severe. § Fe deficiency is very important on dryland soils but often disappears one month after planting. § Fe deficiency results in decreased dry matter production, reduced chlorophyll concentration in leaves, and reduced activity of enzymes involved in sugar metabolism.

(b) (a) (c) Kadar Fe-sufficient : 50 -250 ppm Kadar Fedefisiensi : < 50 ppm Kadar Fe-toxic : > 300 ppm Iron deficiency symptoms in rice (a) Deficiency is mainly a problem on upland soils. (b) Under conditions of severe Fe deficiency, plants are stunted with narrow leaves. (c) Symptoms appears as interveinal yellowing of emerging leaves
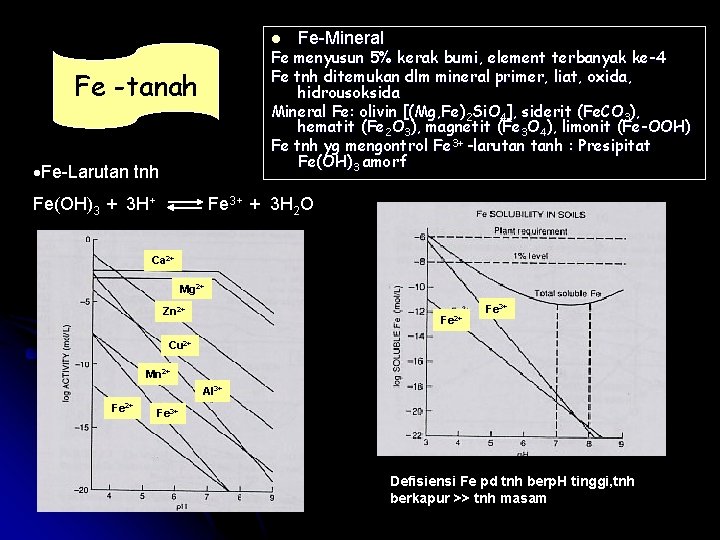
l Fe-Mineral Fe menyusun 5% kerak bumi, element terbanyak ke-4 Fe tnh ditemukan dlm mineral primer, liat, oxida, hidrousoksida Mineral Fe: olivin [(Mg, Fe)2 Si. O 4], siderit (Fe. CO 3), hematit (Fe 2 O 3), magnetit (Fe 3 O 4), limonit (Fe-OOH) Fe tnh yg mengontrol Fe 3+ -larutan tanh : Presipitat Fe(OH)3 amorf Fe -tanah ·Fe-Larutan tnh Fe(OH)3 + 3 H+ Fe 3+ + 3 H 2 O Ca 2+ Mg 2+ Zn 2+ Fe 3+ Cu 2+ Mn 2+ Al 3+ Fe 2+ Fe 3+ Defisiensi Fe pd tnh berp. H tinggi, tnh berkapur >> tnh masam
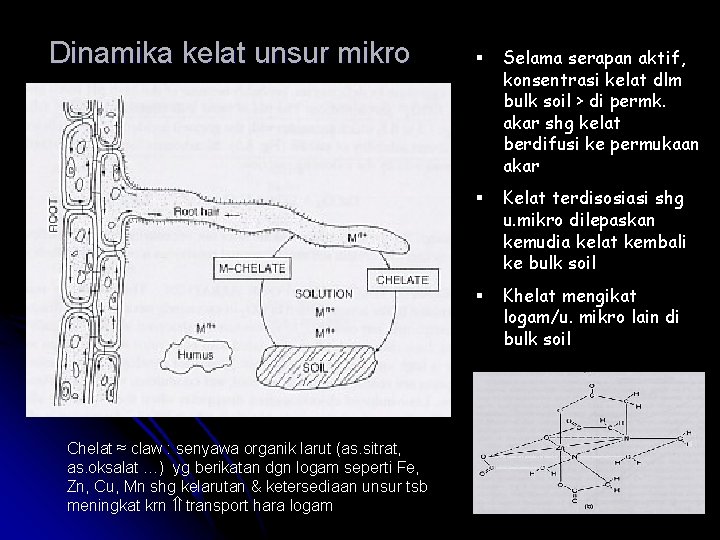
Dinamika kelat unsur mikro Chelat ≈ claw : senyawa organik larut (as. sitrat, as. oksalat …) yg berikatan dgn logam seperti Fe, Zn, Cu, Mn shg kelarutan & ketersediaan unsur tsb meningkat krn transport hara logam § Selama serapan aktif, konsentrasi kelat dlm bulk soil > di permk. akar shg kelat berdifusi ke permukaan akar § Kelat terdisosiasi shg u. mikro dilepaskan kemudia kelat kembali ke bulk soil § Khelat mengikat logam/u. mikro lain di bulk soil

Faktor yg mempengaruhi ketersediaan Fe 1. p. H dan Bicarbonat terbtk pd tnh berkapur. p. H yg tinggi (> 7) berkaitan dgn akumulasi HCO 3 Ca. CO 3 + CO 2 + H 2 O Ca 2+ + 2 HCO 3 - 2. Air berlebih dan aerase yg jelek Tanah yg kompak, bertekstur berat, berkapur berpotensi defisiensi Fe. 3. Bahan organik Aplikasi bahan organik pd tanah berdrainase baik meningkatkan ketersediaan Fe karena: Tersedianya agen pengkelat Meningkatnya struktur tnh bertekstur halus shg aerase menjadi baik 4. Interaksi dgn hara lain Defisiensi Cu dpt disebabkan kelebihan Cu, Mn, Zn dan Mo, interaksi Fe-P. Jk tanaman menyerap NH 4+ mk kelarutan dan ketersediaan Fe meningkat, krn terbtknya kondisi masam Jk tanaman menyerap NO 3 - mk terjadi stress Fe, krn NO 3 - mengganti HCO 3 mk p. H rhizosfer meningkat shg kelarutan Fe menurun 5. Faktor tanaman Setiap tanaman berbeda (faktor genetik) kemampuannya dlm uptake dan translokasi Fe Mekanisme toleransi dan adaptasi Fe : Ekskresi H+ dari akar Ekskresi senyawa kelat dari akar Tingkat reduksi meningkat di akar Meningkatnya anion organik as. Sitrat pd tudung akar. Transfor Fe dari akar ke bagian atas tan. Kurangnya akumulasi P di akar dan tajuk atau kada. P yg sgt tinggi pd media tumbuh

• Competition between nutrients for uptake: Suppression: l l l Mg K, Ca, Mn Ca K, Mg K N, Mg, Ca, B N Fe, Cu NH 4 -N K, Ca, Mg NO 3 -N P, S P Zn, Ca S B, Mo, Fe Cu Zn Zn Fe B, Cu, Mn, Fe compete IOWA STATE UNIVERSITY University Extension Enhancement: l l l NO 3 -N Ca P Mo K Fe S N B Ca

Causes of Fe deficiency § Low concentration of soluble Fe 2+ in upland soils. § Inadequate soil reduction under submerged conditions (e. g. , low organic matter status soils). § High p. H of alkaline or calcareous soils following submergence (i. e. , decreased solubility and uptake of Fe because of large bicarbonate concentration). § Wide P: Fe ratio in the soil (i. e. , Fe bound in Fe phosphates, possibly because of excess application of P fertilizer). § Excessive concentrations of Mn, Cu, Zn, Mo, Ni, and Al. § In upland soils, cultivars with low potential for excretion of organic acids to solubilize Fe. § Increased rhizosphere p. H after the application of large amounts of NO 3 -N fertilizer (rare case and is relevant for upland crops only). Preventive strategies for Fe management Varieties: Screen and breed for tolerance for low soil Fe availability. Grow Fe-efficient cultivars. Selection of high-Fe rice cultivars is in progress to improve Fe nutrition in children and pregnant women in developing countries. Soil management: Apply organic matter (e. g. , crop residues, manure). Apply waste materials from mining and other industrial operations provided that they do not contain other pollutants at toxic concentrations. Fertilizer management: Use acidifying fertilizers (e. g. , ammonium sulfate instead of urea) on high-p. H soils. Use fertilizers containing Fe as a trace element
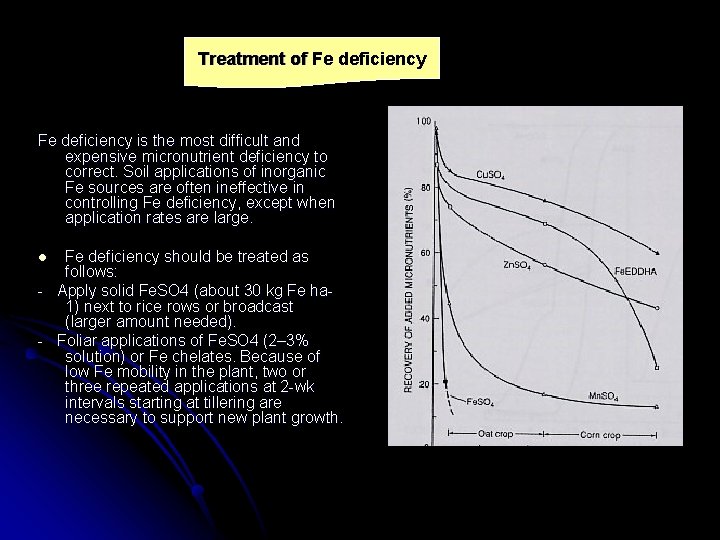
Treatment of Fe deficiency is the most difficult and expensive micronutrient deficiency to correct. Soil applications of inorganic Fe sources are often ineffective in controlling Fe deficiency, except when application rates are large. Fe deficiency should be treated as follows: - Apply solid Fe. SO 4 (about 30 kg Fe ha 1) next to rice rows or broadcast (larger amount needed). - Foliar applications of Fe. SO 4 (2– 3% solution) or Fe chelates. Because of low Fe mobility in the plant, two or three repeated applications at 2 -wk intervals starting at tillering are necessary to support new plant growth. l

Function and mobility Peranan Zn Plant roots absorbs Zn as Zn 2+ and as a componento synthetic Description of Zn deficiency symptoms and natural organic effects on growth complexes. l Cytochrome and nucleotide Dusty brown spots on upper leaves of stunted plants synthesis starting 2– 4 wk after planting. l Auxin metabolism l Chlorophyll production l Symptoms appear between two to four weeks l Enzyme activation after transplanting, with uneven plant growth and l Membrane integrity patches of poorly established hills in the field, but the crop may recover without intervention. l Under severe Zn deficiency, tillering decreases l Zn accumulates in roots and can stop completely and time to crop maturity and can be translocated increases. from roots to developing plant parts. Because little l Zn deficiency can also increase spikelet sterility in retranslocation of Zn rice. occurs within the leaf l Midribs, particularly near the leaf base of younger canopy, particularly in N- leaves, become chlorotic and leaves lose turgor and deficient plants, Zn deficiency turn brown as brown blotches and streaks appear symptoms are more common on lower leaves, enlarge, and coalesce. A white line on young or middle-aged leaves. sometimes appears along the leaf midrib. l Plant growth is stunted and leaf blade size is reduced. In Japan, Zn deficiency is the cause of the "Akagare Type II" disorder in rice l

a) (b) (c) Zinc deficiency symptoms in rice (a) Uneven field growth, plant stunting (foreground). (b) Tillering is reduced. (b), (c) Appearance of dusty brown spots on upper leaves.
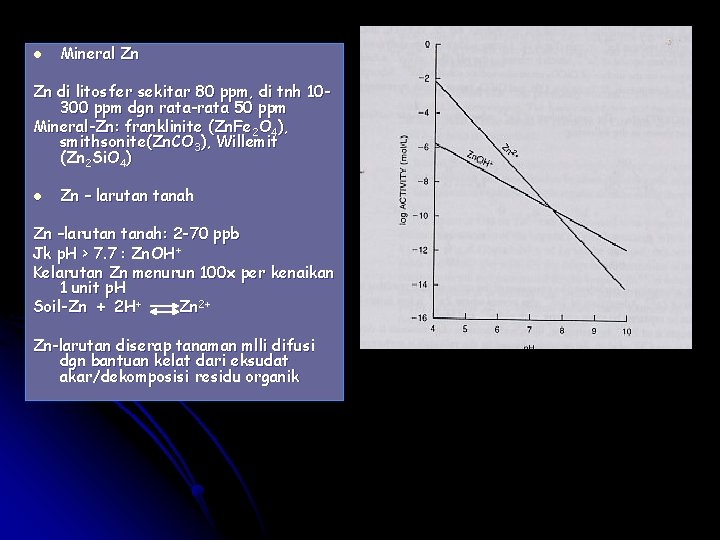
l Mineral Zn Zn di litosfer sekitar 80 ppm, di tnh 10300 ppm dgn rata-rata 50 ppm Mineral-Zn: franklinite (Zn. Fe 2 O 4), smithsonite(Zn. CO 3), Willemit (Zn 2 Si. O 4) l Zn – larutan tanah Zn –larutan tanah: 2 -70 ppb Jk p. H > 7. 7 : Zn. OH+ Kelarutan Zn menurun 100 x per kenaikan 1 unit p. H Soil-Zn + 2 H+ Zn 2+ Zn-larutan diserap tanaman mlli difusi dgn bantuan kelat dari eksudat akar/dekomposisi residu organik
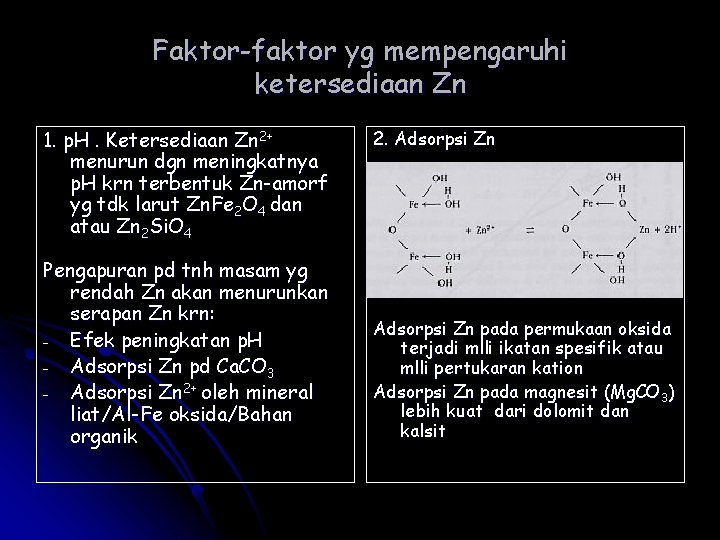
Faktor-faktor yg mempengaruhi ketersediaan Zn 1. p. H. Ketersediaan Zn 2+ menurun dgn meningkatnya p. H krn terbentuk Zn-amorf yg tdk larut Zn. Fe 2 O 4 dan atau Zn 2 Si. O 4 Pengapuran pd tnh masam yg rendah Zn akan menurunkan serapan Zn krn: - Efek peningkatan p. H - Adsorpsi Zn pd Ca. CO 3 - Adsorpsi Zn 2+ oleh mineral liat/Al-Fe oksida/Bahan organik 2. Adsorpsi Zn pada permukaan oksida terjadi mlli ikatan spesifik atau mlli pertukaran kation Adsorpsi Zn pada magnesit (Mg. CO 3) lebih kuat dari dolomit dan kalsit

Faktor yg mempengaruhi ketersediaan Zn 4. Interaksi dgn hara lain Ketersediaan P yg tinggi menyebabkan defisiensi Zn karena terjadi reaksi P-Zn membtk Zn 3(PO 4)2 4 H 2 O Mikoriza meningkatkan serapan u. mikro 5. Penggenangan Jk tnh masam digenangi ketersediaan hara meningkat kecuali Zn Jk tnh berkapur digenangi meningkatkan ketersediaan Zn 6. Kondisi Iklim Defisiensi Zn sering terjadi pd musim hujan dan hilang pd musim panas. Jk temperatur tnh meningkat mk ketersediaan Zn meningkat krn meningkatnya kelarutan difusi Zn 2+ 7. Faktor tanaman Terdapat perbedaan toleransi tanaman thdp ketersediaan Zn yg rendah kn perbedaan kemampuan setiap kultivar dlm menyerap Zn, translokasi dan penggunaan Zn, perbedaan akumulasi hara yng berinteraksi dgn Zn, perbedaan eksploitasi akar terhadap Zn-tanah

Causes of Zn deficiency §Small amount of available Zn in the soil. §Planted varieties are susceptible to Zn deficiency §High p. H (close to 7 or alkaline under anaerobic conditions). Solubility of Zn decreases by two orders of magnitude for each unit increase in p. H. Zn is precipitated as sparingly soluble Zn(OH)2 when p. H increases in acid soil following flooding. §High HCO 3 - concentration because of reducing conditions in calcareous soils with high organic matter content or because of large concentrations of HCO 3 - in irrigation water. §Depressed Zn uptake because of an increase in Fe, Ca, Mg, Cu, Mn, and P after flooding. § Formation of Zn-phosphates following large applications of P fertilizer. High P content in irrigation water (only in areas with polluted water). § Formation of complexes between Zn and organic matter in soils with high p. H and high organic matter content or because of large applications of organic manures and crop residues. § Precipitation of Zn as Zn. S when p. H decreases in alkaline soil following flooding. § Excessive liming. § Wide Mg: Ca ratio (i. e. , >1) and adsorption of Zn by Ca. CO 3 and Mg. CO 3. Excess Mg in soils derived from ultrabasic rocks.

Treatment of Zn deficiency § Zn deficiencies are most § § effectively corrected by soil Zn application. Surface application is more effective than soil incorporation on high p. H soils. If Zn deficiency symptoms are observed in the field, immediately apply 10– 25 kg ha-1 Zn. SO 4. 7 H 2 O. Uptake of Zn. SO 4 is more efficient when broadcast over the soil surface (compared with incorporated) particularly in direct -sown rice. To facilitate more homogeneous application, mix the Zn sulfate (25%) with sand (75%). Apply 0. 5– 1. 5 kg Zn ha-1 as a foliar spray (e. g. , a 0. 5% Zn. SO 4 solution at about 200 L water ha-1) for emergency treatment of Zn deficiency in growing plants. Start at tillering (25– 30 DAT), two or three repeated applications at intervals of 10– 14 d may be necessary. Zn chelates (e. g. , Zn. EDTA) can be used for foliar application, Preventive strategies for Zn management Varieties: Screen and breed for tolerance for low soil Fe availability. Grow Fe-efficient cultivars. Selection of high-Fe rice cultivars is in progress to improve Fe nutrition in children and pregnant women in developing countries. Soil management: Apply organic matter (e. g. , crop residues, manure). Apply waste materials from mining and other industrial operations provided that they do not contain other pollutants at toxic concentrations. Fertilizer management: Use acidifying fertilizers (e. g. , ammonium sulfate instead of urea) on high-p. H soils. Use fertilizers containing Fe as a trace element
- Slides: 19