Kent Kirshenbaum kentnyu edu 212 998 8486 Waverly

Kent Kirshenbaum kent@nyu. edu (212) 998 -8486 Waverly 866

Agenda • • The structure of water Why oil and water don’t mix Altering water composition in cooking Phase changes of water – Reduction of a stock: water to vapor (P. Gadsby) – Rapid freezing of oil/water emulsions (D. Arnold) – Lyophilization: sublimating ice to vapor (Y. Utku) • Why a souffle rises (Y. Utku, inspired by H. This) • Reception & tour of the research lab
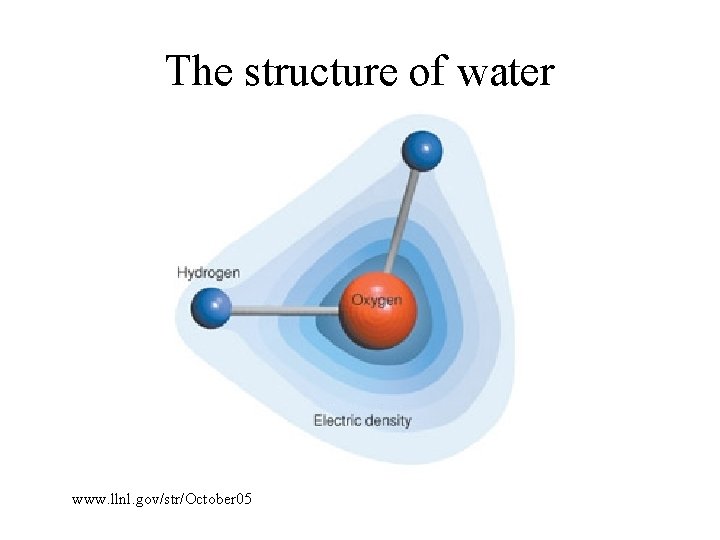
The structure of water www. llnl. gov/str/October 05

Charge distribution in water
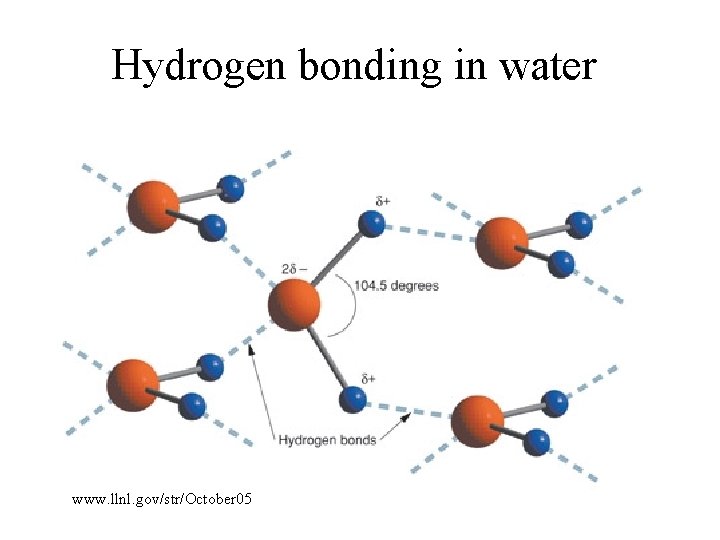
Hydrogen bonding in water www. llnl. gov/str/October 05
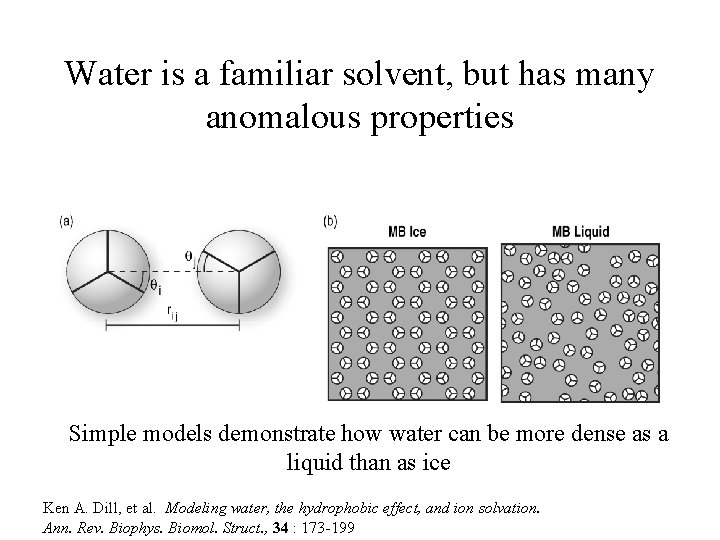
Water is a familiar solvent, but has many anomalous properties Simple models demonstrate how water can be more dense as a liquid than as ice Ken A. Dill, et al. Modeling water, the hydrophobic effect, and ion solvation. Ann. Rev. Biophys. Biomol. Struct. , 34 : 173 -199
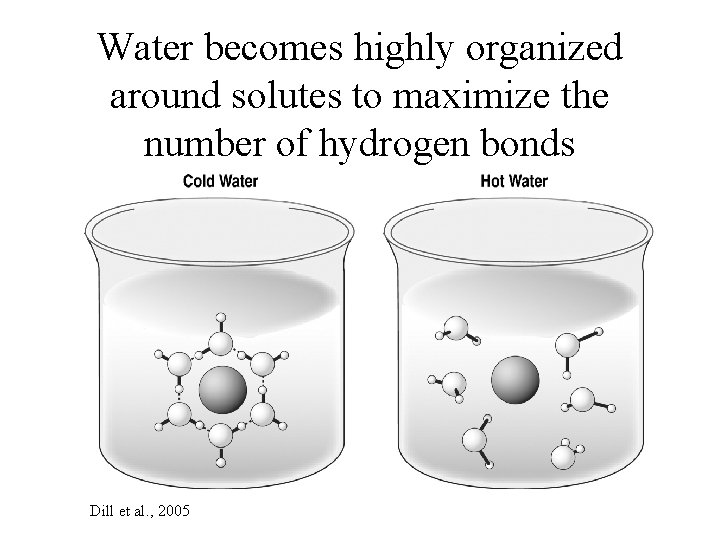
Water becomes highly organized around solutes to maximize the number of hydrogen bonds Dill et al. , 2005
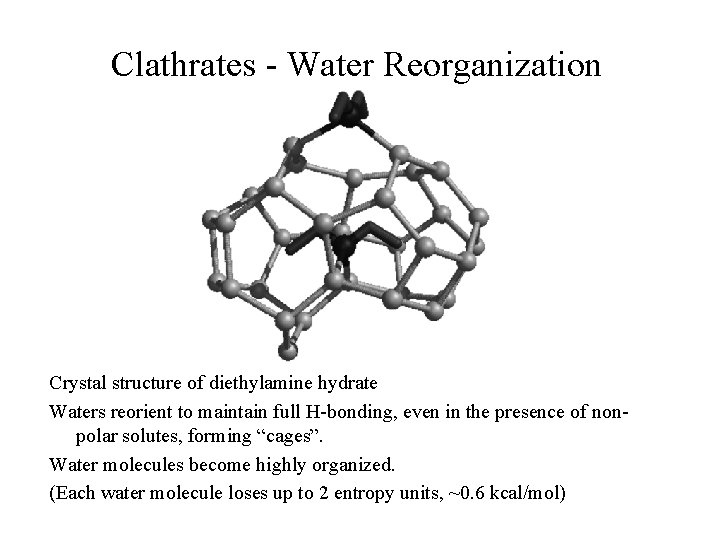
Clathrates - Water Reorganization Crystal structure of diethylamine hydrate Waters reorient to maintain full H-bonding, even in the presence of nonpolar solutes, forming “cages”. Water molecules become highly organized. (Each water molecule loses up to 2 entropy units, ~0. 6 kcal/mol)

Figure showing ordered water in proximity to lipids

Figure showing lipids dispersed in water

Figure showing favorable packing of lipids in bulk water

Understanding The Hydrophobic Effect • By self-associating, hydrophobic molecules reduce the amount of non-polar surface exposed to solvent. • This explains the separation of oil and water. Restrict oil to one phase, water to another, and minimize the extent of water reorganization • Association of hydrophobes is the result of an increase in the disorder of water, not merely due to “sticking interactions” between the hydrophobes
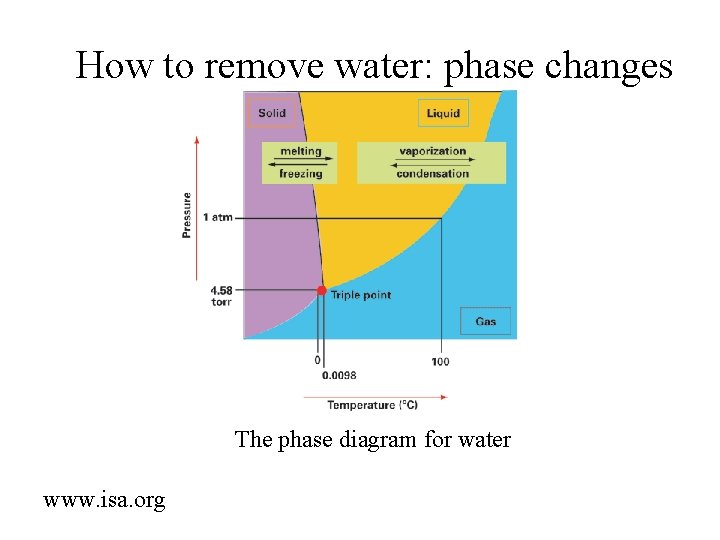
How to remove water: phase changes The phase diagram for water www. isa. org
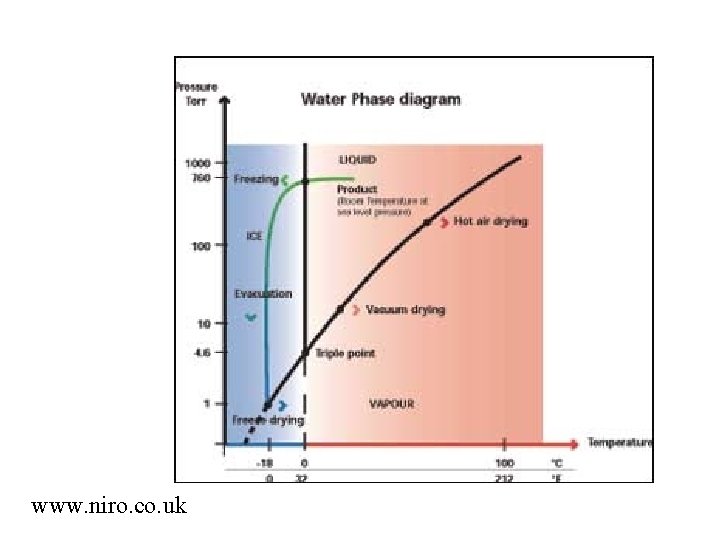
www. niro. co. uk

The volume change of a souffle: heating of a liquid • The thermal coefficient of expansion of water is 0. 00021 per 1° Celsius at 20° Celsius.

The volume change of a souffle: expansion of a gas “As it bakes, the air bubbles we've incorporated into the batter will start to expand, causing the entire souffle to rise. ” www. cookingforengineers. com/recipe/160/Dark-Chocolate-Souffle
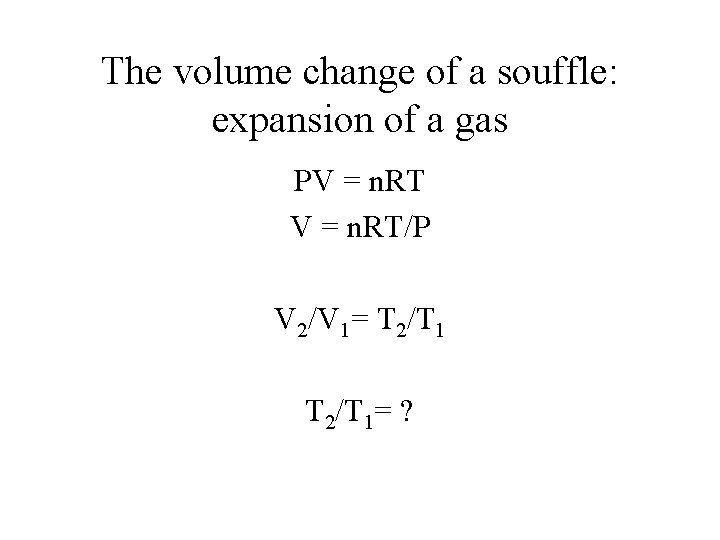
The volume change of a souffle: expansion of a gas PV = n. RT/P V 2/V 1= T 2/T 1= ?

The volume change of a souffle: expansion of a gas PV = n. RT/P V 2/V 1= T 2/T 1= 370/300 23% increase

The volume change of a souffle: liquid/vapor phase transitions PV = n. RT/P V 2/V 1= n 2/n 1 Is it reasonable to get the expected volume changes by converting a plausible amount of water to steam?

The volume change of a souffle: liquid/vapor phase transitions PV = n. RT/P V 2/V 1= n 2/n 1 One mole of an ideal gas is 22. 4 liters at 273 K. To double the size of a one liter souffle, we will need to convert 1/22. 4 moles of water to steam and retain this in the matrix of the souffle. This is 0. 045 moles x 18 g/mole = 0. 81 g of water.

- Slides: 21