Kathy Kokotis RN BS MBA Becton Dickinson Director

Kathy Kokotis RN BS MBA Becton Dickinson Director Global Clinical Development Vascular Access Change in Practice BAS/TSHW/0318/0058

Disclosure The speaker’s presentation today is on behalf of Becton Dickinson. Any discussion regarding Becton Dickinson products during the presentation today is limited to information that is consistent with Becton Dickinson labeling. Please consult Becton Dickinson product labels and inserts for any indications, contraindications, hazards, warnings, cautions and instructions for use. Results presented may not be predictive for all institutions or patients. Kathy Kokotis is an employee of Becton Dickinson and holds Becton Dickinson Stock as well as Johnson and Johnson Medical Stock. BAS/TSHW/0318/0058

My Grandmother (84) Why I started my quest for early assessment in vascular access needs Taken November 1995: Brain metastasis no chemotherapy Day 6 of a two week hospital stay Acute care CICC Jugular for TPN/antibiotics in the ICU (day 7) CICC was pulled for a CR-BSI on day 14 prior to her death (CLA-BSI definition did not exist in 1995) BAS/TSHW/0318/0058

Decision Change: Infusates 2011 versus 2017 Infusion Nursing Standards of Practice BAS/TSHW/0318/0058

Decision Making: INS 2011 � Therapies not appropriate for short peripheral catheters include continuous vesicant therapy, parenteral nutrition, infusates with p. H less than 5 or greater than 9, and infusates with an osmolality greater than 600 m. Osm/L. The nurse should collaborate with the pharmacist and the licensed independent practitioner (LIP) to assist in selection of the most appropriate vascular access device based on a projected treatment plan � Therapies not appropriate for midline catheters include continuous vesicant therapy, parenteral nutrition, infusates with p. H less than 5 or greater than 9, and infusates with an osmolality greater than 600 m. Osm/L. Infusion Nurses Society: (2011) Infusion Nursing Standards of Practice, JIN 34 (1 S)S p. 37 BAS/TSHW/0318/0058

Decision Making: INS 2016 �A. Choose a short peripheral catheter as follows: 1. Consider the infusate characteristics (eg, irritant, vesicant, osmolarity) in conjunction with anticipated duration of infusion therapy (eg, less than 6 days) and availability of peripheral vascular access sites. 2. Use vascular visualization technology (eg, near infrared, ultrasound) to increase success for patients with difficult venous access (refer to Standard 22, Vascular Visualization ). 3. Do not use peripheral catheters for continuous vesicant therapy, parenteral nutrition, or infusates with an osmolarity greater than 900. Infusion Nurses Society: (2016): Infusion Therapy Standards of Practice JIN (Jan/Feb) 39(1 S) BAS/TSHW/0318/0058

MAGIC: 2015 Appropriateness Guide �For peripherally compatible infusions, PICC use was rated as inappropriate when the proposed duration of use was 5 or fewer days. Midline catheters and ultrasonography-guided peripheral �Midline catheters and ultrasonography-guided peripheral intravenous catheters were preferred to PICCs for use between 6 and 14 days. Chopra V. Flanders S. Saint S. (Sept. 2015)The Michigan Appropriateness Guide for Intravenous Catheters (MAGIC): Results From a Multispecialty Panel Using the RAND/UCLA Appropriateness Method, Annals of Internal Medicine 163(6) p 1 -39 S BAS/TSHW/0318/0058

Definitions VESICANT EXTRAVASATION � An agent capable of � The inadvertent infiltration causing blistering, tissue sloughing, or necrosis when it escapes from the intended vascular pathway into the surrounding tissues of vesicant solution or medication into the surrounding tissue Gorski LA, Stranz M, Cook LS, et al. : (2017) Development of an evidence-based list of noncytotoxic vesicant medications And solutions JIN 40(1) Jan/Feb 26 -40 BAS/TSHW/0318/0058
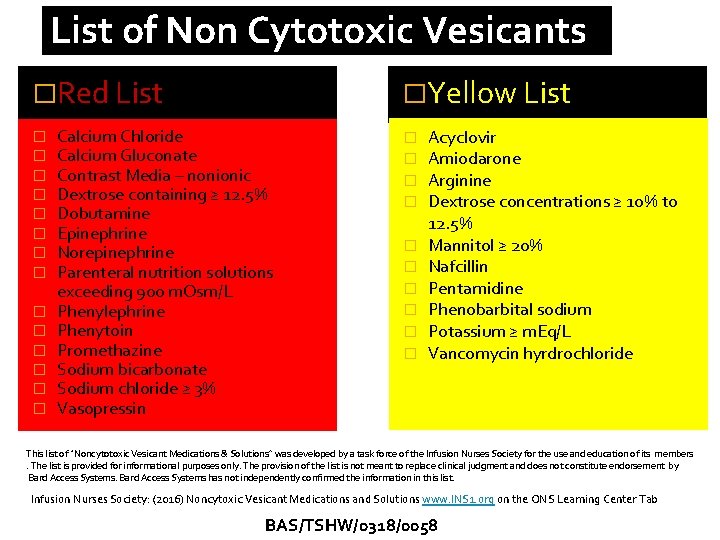
List of Non Cytotoxic Vesicants �Red List � � � �Yellow List Calcium Chloride Calcium Gluconate Contrast Media – nonionic Dextrose containing ≥ 12. 5% Dobutamine Epinephrine Norepinephrine Parenteral nutrition solutions exceeding 900 m. Osm/L Phenylephrine Phenytoin Promethazine Sodium bicarbonate Sodium chloride ≥ 3% Vasopressin � � � � � Acyclovir Amiodarone Arginine Dextrose concentrations ≥ 10% to 12. 5% Mannitol ≥ 20% Nafcillin Pentamidine Phenobarbital sodium Potassium ≥ m. Eq/L Vancomycin hyrdrochloride This list of “Noncytotoxic Vesicant Medications & Solutions” was developed by a task force of the Infusion Nurses Society for the use and education of its members. The list is provided for informational purposes only. The provision of the list is not meant to replace clinical judgment and does not constitute endorsement by Bard Access Systems has not independently confirmed the information in this list. Infusion Nurses Society: (2016) Noncytoxic Vesicant Medications and Solutions www. INS 1. org on the ONS Learning Center Tab BAS/TSHW/0318/0058
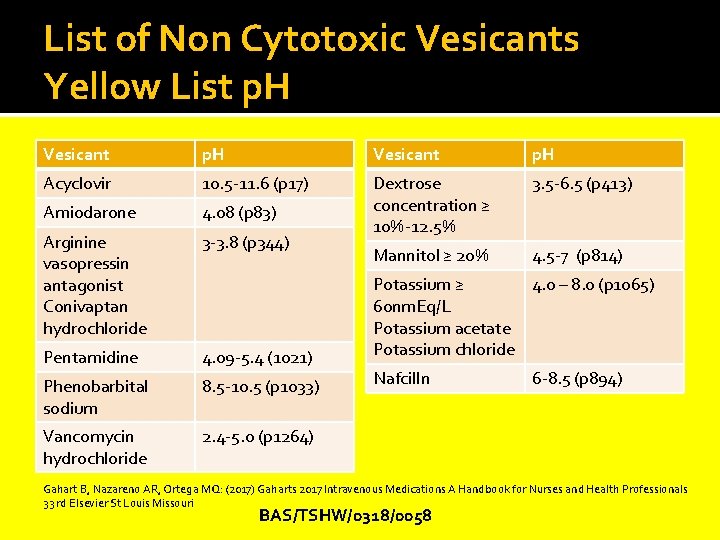
List of Non Cytotoxic Vesicants Yellow List p. H Vesicant p. H Acyclovir 10. 5 -11. 6 (p 17) 3. 5 -6. 5 (p 413) Amiodarone 4. 08 (p 83) Arginine vasopressin antagonist Conivaptan hydrochloride 3 -3. 8 (p 344) Dextrose concentration ≥ 10%-12. 5% Mannitol ≥ 20% 4. 5 -7 (p 814) Pentamidine 4. 09 -5. 4 (1021) Phenobarbital sodium 8. 5 -10. 5 (p 1033) Vancomycin hydrochloride 2. 4 -5. 0 (p 1264) Potassium ≥ 4. 0 – 8. 0 (p 1065) 60 nm. Eq/L Potassium acetate Potassium chloride Nafcilln 6 -8. 5 (p 894) Gahart B, Nazareno AR, Ortega MQ: (2017) Gaharts 2017 Intravenous Medications A Handbook for Nurses and Health Professionals 33 rd Elsevier St Louis Missouri BAS/TSHW/0318/0058
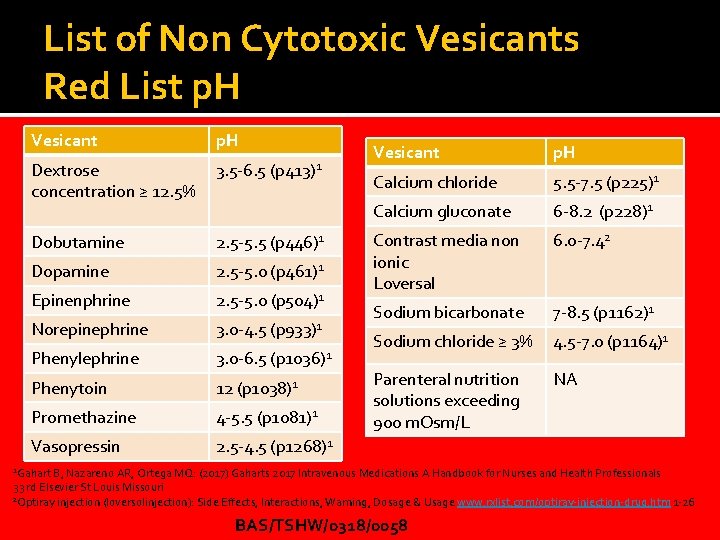
List of Non Cytotoxic Vesicants Red List p. H Vesicant p. H (p 413)1 Dextrose concentration ≥ 12. 5% 3. 5 -6. 5 Dobutamine 2. 5 -5. 5 (p 446)1 Dopamine 2. 5 -5. 0 (p 461)1 Epinenphrine 2. 5 -5. 0 (p 504)1 Norepinephrine 3. 0 -4. 5 (p 933)1 Phenylephrine 3. 0 -6. 5 (p 1036)1 Phenytoin 12 (p 1038)1 Promethazine 4 -5. 5 (p 1081)1 Vasopressin 2. 5 -4. 5 (p 1268)1 Vesicant p. H Calcium chloride 5. 5 -7. 5 (p 225)1 Calcium gluconate 6 -8. 2 (p 228)1 Contrast media non ionic Loversal 6. 0 -7. 42 Sodium bicarbonate 7 -8. 5 (p 1162)1 Sodium chloride ≥ 3% 4. 5 -7. 0 (p 1164)1 Parenteral nutrition solutions exceeding 900 m. Osm/L NA 1 Gahart B, Nazareno AR, Ortega MQ: (2017) Gaharts 2017 Intravenous Medications A Handbook for Nurses and Health Professionals 33 rd Elsevier St Louis Missouri 2 Optiray injection (loversolinjection): Side Effects, Interactions, Warning, Dosage & Usage www. rxlist. com/optiray-injection-drug. htm 1 -26 BAS/TSHW/0318/0058

“While some vesicant infusates possess extreme p. H levels (eg, acyclovir, pentobarbital, phenytoin) or are clearly hyperosmolar (eg, calcium chloride, high dextrose concentrations), many of the vesicants have neither property” Gorski LA, Stranz M, Cook LS, et al. : (2017) Development of an evidence-based list of noncytotoxic vesicant medications And solutions JIN 40(1) Jan/Feb 26 -40 BAS/TSHW/0318/0058
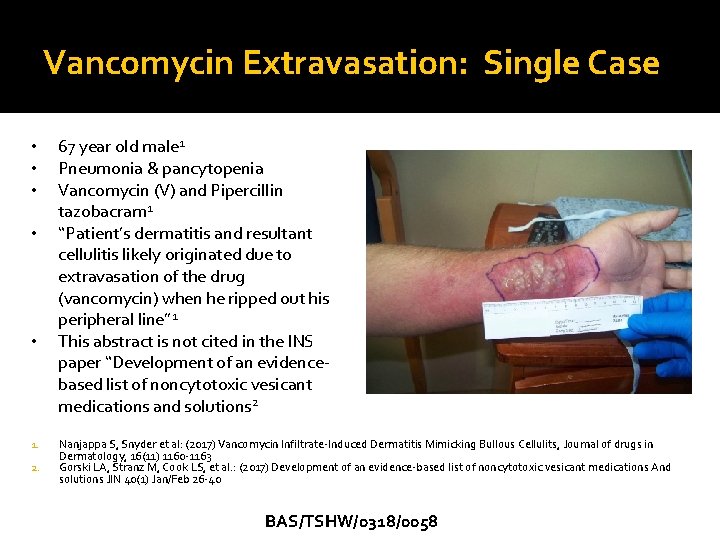
Vancomycin Extravasation: Single Case • • • 1. 2. 67 year old male 1 Pneumonia & pancytopenia Vancomycin (V) and Pipercillin tazobacram 1 “Patient’s dermatitis and resultant cellulitis likely originated due to extravasation of the drug (vancomycin) when he ripped out his peripheral line” 1 This abstract is not cited in the INS paper “Development of an evidencebased list of noncytotoxic vesicant medications and solutions 2 Nanjappa S, Snyder et al: (2017) Vancomycin Infiltrate-Induced Dermatitis Mimicking Bullous Cellulits, Journal of drugs in Dermatology, 16(11) 1160 -1163 Gorski LA, Stranz M, Cook LS, et al. : (2017) Development of an evidence-based list of noncytotoxic vesicant medications And solutions JIN 40(1) Jan/Feb 26 -40 BAS/TSHW/0318/0058

Vancomycin Extravasation: Single Case � 84 year old Female developed hospital acquired pneumonia Vancomycin (1, 000 mg daily) & cefepime (1, 000 mg BID) 220 pounds, anemia, asthma, afib. , CAD, hypertension, Diabetes Type 2, previous CABG 20 g right wrist Day 2 extravasation with blistering to right wrist Necrotic tissue developed (over week) Black eschar overlapping an ulcer developed Right thrombophelbitis mid forearm near the cephalic vein Wound healing 5 weeks with no surgical consult Author recommends central administration Peyko V, Saasson E: (2016) Vancomycin extravasation: evaluation, treatment, and avoidance of This adverse drug event, Case Reports in Internal Medicine 3(3) p. 40 -43 BAS/TSHW/0318/0058

Decision Making: INS 2016 �Use caution with intermittent vesicant administration due to risk of undetected extravasation. �The administration of vancomycin for less than 6 days through a midline catheter was found to be safe in 1 study (Level IV evidence) Infusion Nurses Society: (2016): Infusion Therapy Standards of Practice JIN (Jan/Feb) 39(1 S) BAS/TSHW/0318/0058

Definitions IRRITANT � An agent capable of producing discomfort (burning, stinging) or pain as a result of irritation in the internal lumen of the vein with or without immediate external signs of vein inflammation Gorski LA, Stranz M, Cook LS, et al. : (2017) Development of an evidence-based list of noncytotoxic vesicant medications And solutions JIN 40(1) Jan/Feb 26 -40 BAS/TSHW/0318/0058

Vesicant and Irritant �Vesicants cause severe damage when the agent escapes the vein �Irritants cause damage within the vein Gorski LA, Stranz M, Cook LS, et al. : (2017) Development of an evidence-based list of noncytotoxic vesicant medications And solutions JIN 40(1) Jan/Feb 26 -40 BAS/TSHW/0318/0058

Decision: Vesicant �VAD selection is a complex decision not based on a single factor �Peripheral administration of short term vesicant in an ER or small number of doses may or may not be appropriate given a patient’s vascular assessment Gorski LA, Stranz M, Cook LS, et al. : (2017) Development of an evidence-based list of noncytotoxic vesicant medications And solutions JIN 40(1) Jan/Feb 26 -40 BAS/TSHW/0318/0058

Vesicants in Yellow are still a Vesicant Patient Assessment is important BAS/TSHW/0318/0058

Decision Making Changes: ICU Patients BAS/TSHW/0318/0058

Decision Making ICU: INS 2016 �Use a PICC with caution in patients who have cancer or are critically ill due to venous thrombosis and infection risk. Infusion Nurses Society: (2016): Infusion Therapy Standards of Practice JIN (Jan/Feb) 39(1 S) BAS/TSHW/0318/0058

Audience participation BAS/TSHW/0318/0058
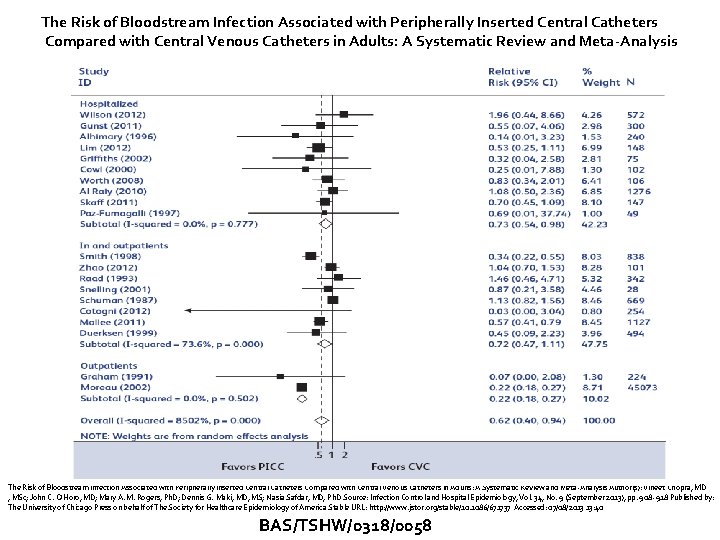
The Risk of Bloodstream Infection Associated with Peripherally Inserted Central Catheters Compared with Central Venous Catheters in Adults: A Systematic Review and Meta-Analysis Author(s): Vineet Chopra, MD , MSc; John C. O’Horo, MD; Mary A. M. Rogers, Ph. D; Dennis G. Maki, MD, MS; Nasia Safdar, MD, Ph. D Source: Infection Control and Hospital Epidemiology, Vol. 34, No. 9 (September 2013), pp. 908 - 918 Published by: The University of Chicago Press on behalf of The Society for Healthcare Epidemiology of America Stable URL: http: //www. jstor. org/stable/10. 1086/671737 Accessed: 07/08/2013 13: 40 BAS/TSHW/0318/0058
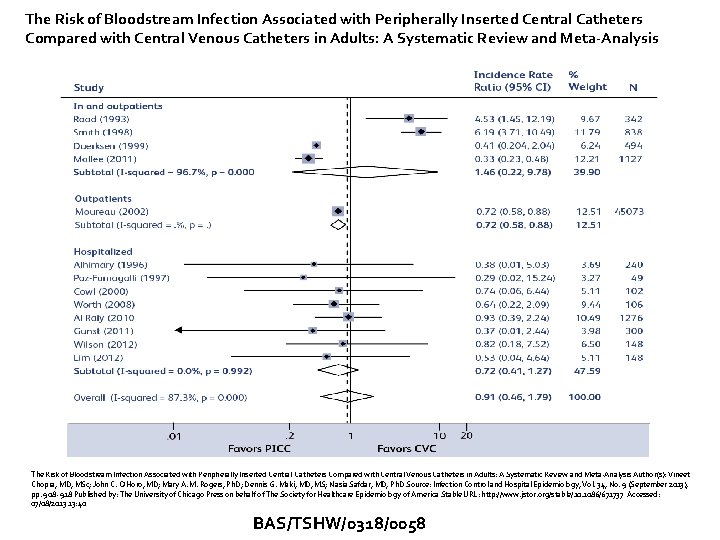
The Risk of Bloodstream Infection Associated with Peripherally Inserted Central Catheters Compared with Central Venous Catheters in Adults: A Systematic Review and Meta-Analysis Author(s): Vineet Chopra, MD, MSc; John C. O’Horo, MD; Mary A. M. Rogers, Ph. D; Dennis G. Maki, MD, MS; Nasia Safdar, MD, Ph. D Source: Infection Control and Hospital Epidemiology, Vol. 34, No. 9 (September 2013), pp. 908 - 918 Published by: The University of Chicago Press on behalf of The Society for Healthcare Epidemiology of America Stable URL: http: //www. jstor. org/stable/10. 1086/671737 Accessed: 07/08/2013 13: 40 BAS/TSHW/0318/0058

The Risk of Bloodstream Infection Associated with Peripherally Inserted Central Catheters Compared with Central Venous Catheters in Adults: A Systematic Review and Meta-Analysis � “risk of CLABSI associated with CVCs and PICCs appears to be similar in hospitalized patients, expansion of practices and campaigns such as hub decontamination and “scrub the hub” should specifically be targeted toward PICCs. ” � “hospitalized patients who underwent PICC placement experienced CLABSI rates that statistically paralleled that associated with CVCs” The Risk of Bloodstream Infection Associated with Peripherally Inserted Central Catheters Compared with Central Venous Catheters in Adults: A Systematic Review and Meta-Analysis Author(s): Vineet Chopra, MD, MSc; John C. O’Horo, MD; Mary A. M. Rogers, Ph. D; Dennis G. Maki, MD, MS; Nasia Safdar, MD, Ph. D Source: Infection Control and Hospital Epidemiology, Vol. 34, No. 9 (September 2013), pp. 908 - 918 Published by: The University of Chicago Press on behalf of The Society for Healthcare Epidemiology of America Stable URL: http: //www. jstor. org/stable/10. 1086/671737 Accessed: 07/08/2013 13: 40 BAS/TSHW/0318/0058
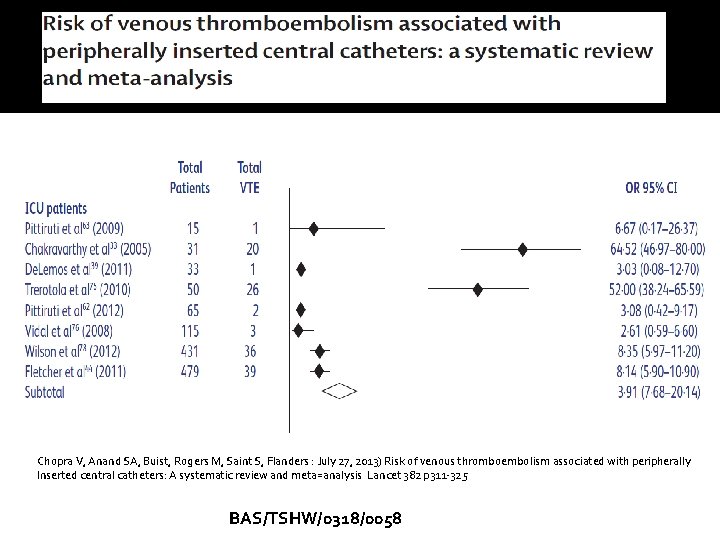
Chopra V, Anand SA, Buist, Rogers M, Saint S, Flanders : July 27, 2013) Risk of venous thromboembolism associated with peripherally Inserted central catheters: A systematic review and meta=analysis Lancet 382 p 311 -325 BAS/TSHW/0318/0058
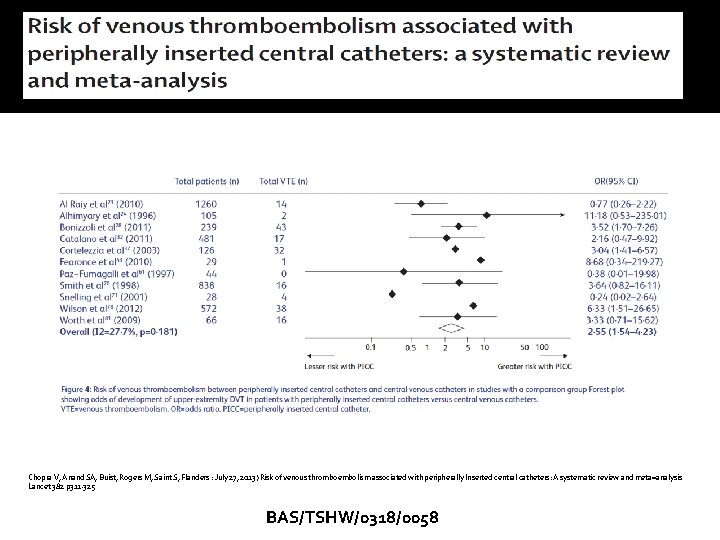
Chopra V, Anand SA, Buist, Rogers M, Saint S, Flanders : July 27, 2013) Risk of venous thromboembolism associated with peripherally Inserted central catheters: A systematic review and meta=analysis Lancet 382 p 311 -325 BAS/TSHW/0318/0058
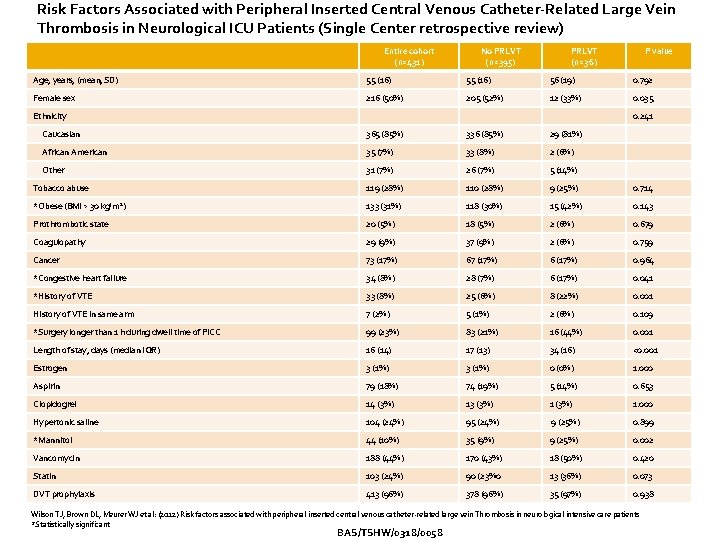
Risk Factors Associated with Peripheral Inserted Central Venous Catheter-Related Large Vein Thrombosis in Neurological ICU Patients (Single Center retrospective review) Entire cohort (n=431) No PRLVT (n=395) PRLVT (n=36) P value Age, years, (mean, SD) 55 (16) 56 (19) 0. 792 Female sex 216 (50%) 205 (52%) 12 (33%) 0. 035 Ethnicity 0. 241 Caucasian 365 (85%) 336 (85%) 29 (81%) African American 35 (7%) 33 (8%) 2 (6%) Other 31 (7%) 26 (7%) 5 (14%) Tobacco abuse 119 (28%) 110 (28%) 9 (25%) 0. 714 *Obese (BMI > 30 kg/m 2) 133 (31%) 118 (30%) 15 (42%) 0. 143 Prothrombotic state 20 (5%) 18 (5%) 2 (6%) 0. 679 Coagulopathy 29 (9%) 37 (9%) 2 (6%) 0. 759 Cancer 73 (17%) 67 (17%) 6 (17%) 0. 964 *Congestive heart failure 34 (8%) 28 (7%) 6 (17%) 0. 041 *History of VTE 33 (8%) 25 (6%) 8 (22%) 0. 001 History of VTE in same arm 7 (2%) 5 (1%) 2 (6%) 0. 109 *Surgery longer than 1 h during dwell time of PICC 99 (23%) 83 (21%) 16 (44%) 0. 001 Length of stay, days (median IQR) 16 (14) 17 (13) 34 (16) <0. 001 Estrogen 3 (1%) 0 (0%) 1. 000 Aspirin 79 (18%) 74 (19%) 5 (14%) 0. 653 Clopidogrel 14 (3%) 13 (3%) 1. 000 Hypertonic saline 104 (24%) 95 (24%) 9 (25%) 0. 899 *Mannitol 44 (10%) 35 (9%) 9 (25%) 0. 002 Vancomycin 188 (44%) 170 (43%) 18 (50%) 0. 420 Statin 103 (24%) 90 (23%0 13 (36%) 0. 073 DVT prophylaxis 413 (96%) 378 (96%) 35 (97%) 0. 938 Wilson TJ, Brown DL, Meurer WJ et al: (2012) Risk factors associated with peripheral inserted central venous catheter-related large vein Thrombosis in neurological intensive care patients *Statistically significant BAS/TSHW/0318/0058
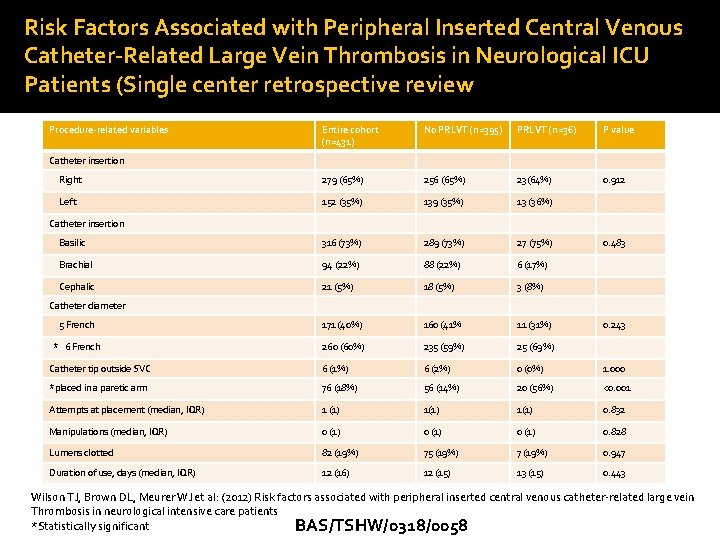
Risk Factors Associated with Peripheral Inserted Central Venous Catheter-Related Large Vein Thrombosis in Neurological ICU Patients (Single center retrospective review Procedure-related variables Entire cohort (n=431) No PRLVT (n=395) PRLVT (n=36) P value Right 279 (65%) 256 (65%) 23(64%) 0. 912 Left 152 (35%) 139 (35%) 13 (36%) Basilic 316 (73%) 289 (73%) 27 (75%) Brachial 94 (22%) 88 (22%) 6 (17%) Cephalic 21 (5%) 18 (5%) 3 (8%) 5 French 171 (40%) 160 (41% 11 (31%) * 6 French 260 (60%) 235 (59%) 25 (69%) Catheter tip outside SVC 6 (1%) 6 (2%) 0 (0%) 1. 000 *placed in a paretic arm 76 (18%) 56 (14%) 20 (56%) <0. 001 Attempts at placement (median, IQR) 1 (1) 1(1) 0. 832 Manipulations (median, IQR) 0 (1) 0. 828 Lumens clotted 82 (19%) 75 (19%) 7 (19%) 0. 947 Duration of use, days (median, IQR) 12 (16) 12 (15) 13 (15) 0. 443 Catheter insertion 0. 483 Catheter diameter 0. 243 Wilson TJ, Brown DL, Meurer WJ et al: (2012) Risk factors associated with peripheral inserted central venous catheter-related large vein Thrombosis in neurological intensive care patients *Statistically significant BAS/TSHW/0318/0058
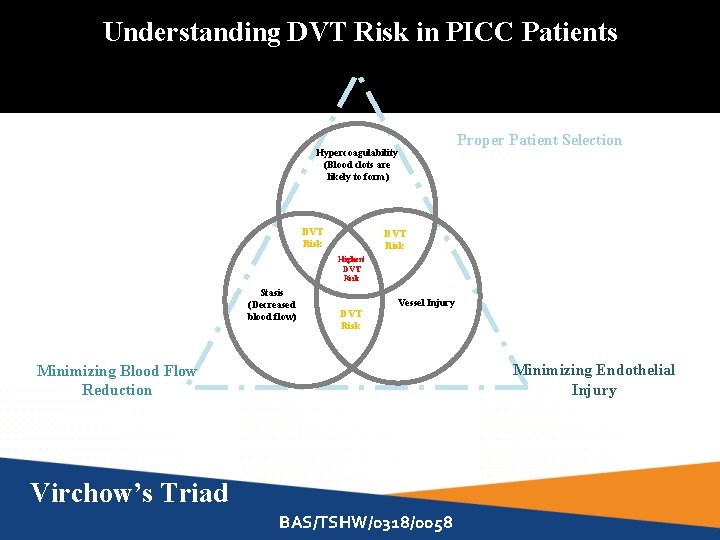
Understanding DVT Risk in PICC Patients Hypercoagulability (Blood clots are likely to form) DVT Risk Proper Patient Selection DVT Risk Highest DVT Risk Stasis (Decreased blood flow) DVT Risk Vessel Injury Minimizing Endothelial Injury Minimizing Blood Flow Reduction Virchow’s Triad BAS/TSHW/0318/0058
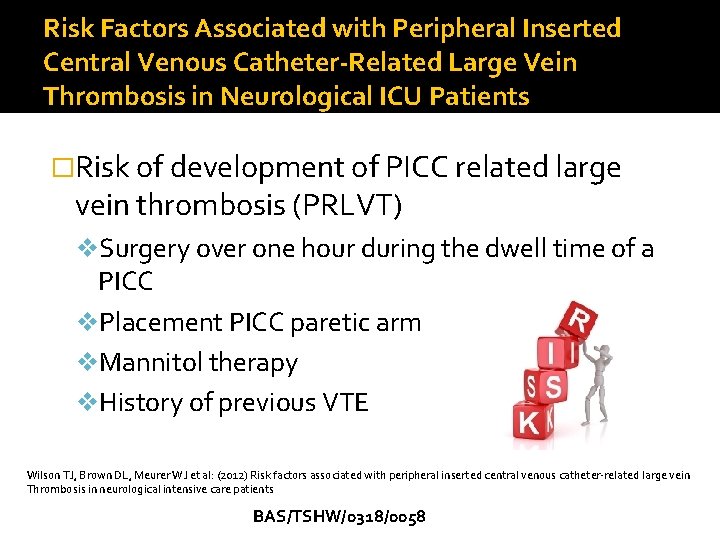
Risk Factors Associated with Peripheral Inserted Central Venous Catheter-Related Large Vein Thrombosis in Neurological ICU Patients �Risk of development of PICC related large vein thrombosis (PRLVT) v. Surgery over one hour during the dwell time of a PICC v. Placement PICC paretic arm v. Mannitol therapy v. History of previous VTE Wilson TJ, Brown DL, Meurer WJ et al: (2012) Risk factors associated with peripheral inserted central venous catheter-related large vein Thrombosis in neurological intensive care patients BAS/TSHW/0318/0058
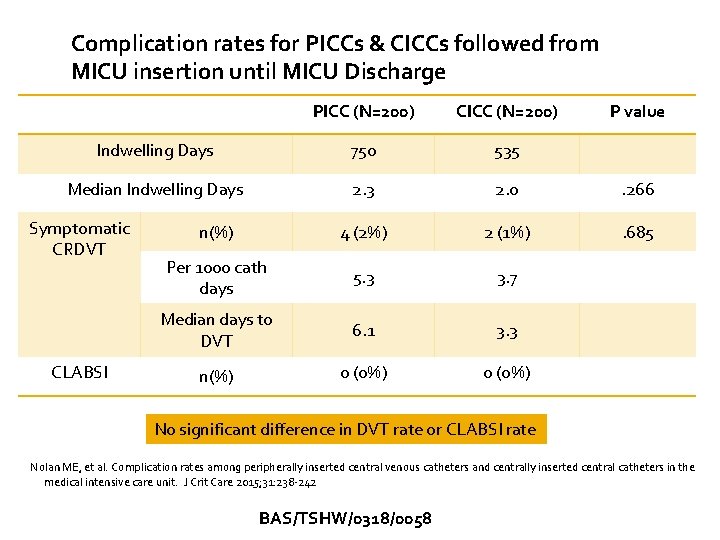
Complication rates for PICCs & CICCs followed from MICU insertion until MICU Discharge PICC (N=200) CICC (N=200) Indwelling Days 750 535 Median Indwelling Days 2. 3 2. 0 . 266 n(%) 4 (2%) 2 (1%) . 685 Per 1000 cath days 5. 3 3. 7 Median days to DVT 6. 1 3. 3 n(%) 0 (0%) Symptomatic CRDVT CLABSI P value No significant difference in DVT rate or CLABSI rate Nolan ME, et al. Complication rates among peripherally inserted central venous catheters and centrally inserted central catheters in the medical intensive care unit. J Crit Care 2015; 31: 238 -242 BAS/TSHW/0318/0058
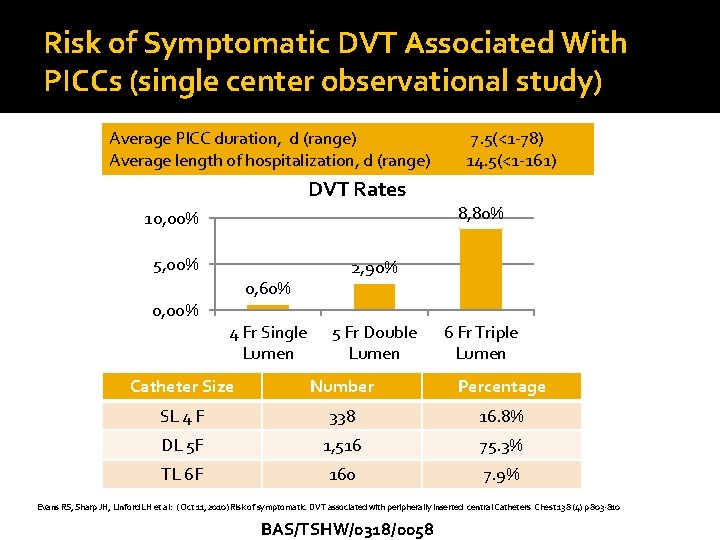
Risk of Symptomatic DVT Associated With PICCs (single center observational study) Average PICC duration, d (range) Average length of hospitalization, d (range) DVT Rates 10, 00% 5, 00% 0, 60% 4 Fr Single Lumen 7. 5(<1 -78) 14. 5(<1 -161) 8, 80% 2, 90% 5 Fr Double Lumen 6 Fr Triple Lumen Catheter Size Number Percentage SL 4 F 338 16. 8% DL 5 F 1, 516 75. 3% TL 6 F 160 7. 9% Evans RS, Sharp JH, Linford LH et al: ( Oct 11, 2010) Risk of symptomatic DVT associated with peripherally inserted central Catheters Chest 138 (4) p 803 -810 BAS/TSHW/0318/0058
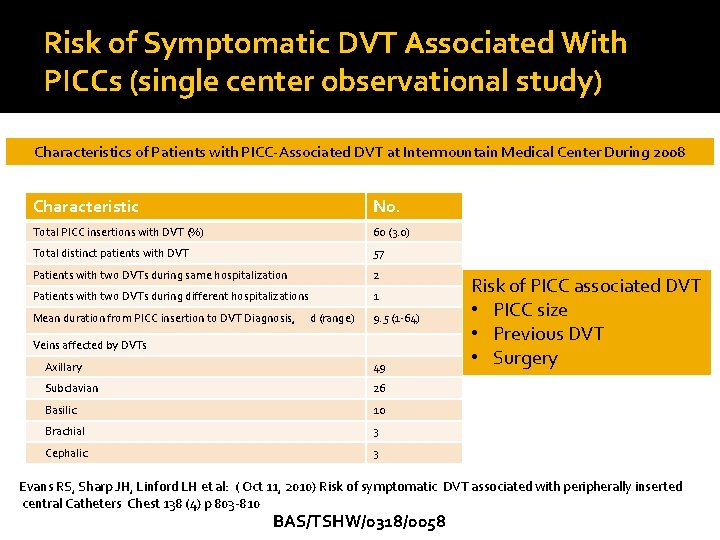
Risk of Symptomatic DVT Associated With PICCs (single center observational study) Characteristics of Patients with PICC-Associated DVT at Intermountain Medical Center During 2008 Characteristic No. Total PICC insertions with DVT (%) 60 (3. 0) Total distinct patients with DVT 57 Patients with two DVTs during same hospitalization 2 Patients with two DVTs during different hospitalizations 1 Mean duration from PICC insertion to DVT Diagnosis, d (range) 9. 5 (1 -64) Veins affected by DVTs Axillary 49 Subclavian 26 Basilic 10 Brachial 3 Cephalic 3 Risk of PICC associated DVT • PICC size • Previous DVT • Surgery Evans RS, Sharp JH, Linford LH et al: ( Oct 11, 2010) Risk of symptomatic DVT associated with peripherally inserted central Catheters Chest 138 (4) p 803 -810 BAS/TSHW/0318/0058
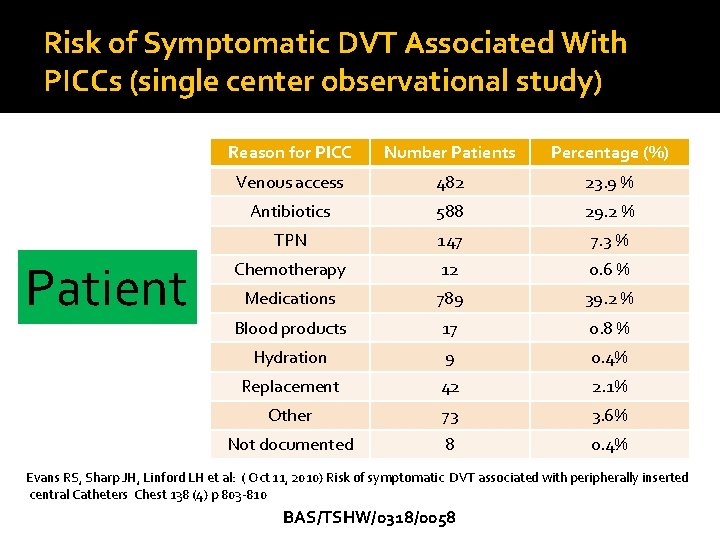
Risk of Symptomatic DVT Associated With PICCs (single center observational study) Patient Reason for PICC Number Patients Percentage (%) Venous access 482 23. 9 % Antibiotics 588 29. 2 % TPN 147 7. 3 % Chemotherapy 12 0. 6 % Medications 789 39. 2 % Blood products 17 0. 8 % Hydration 9 0. 4% Replacement 42 2. 1% Other 73 Who got a PICC Not documented 8 3. 6% 0. 4% Evans RS, Sharp JH, Linford LH et al: ( Oct 11, 2010) Risk of symptomatic DVT associated with peripherally inserted central Catheters Chest 138 (4) p 803 -810 BAS/TSHW/0318/0058
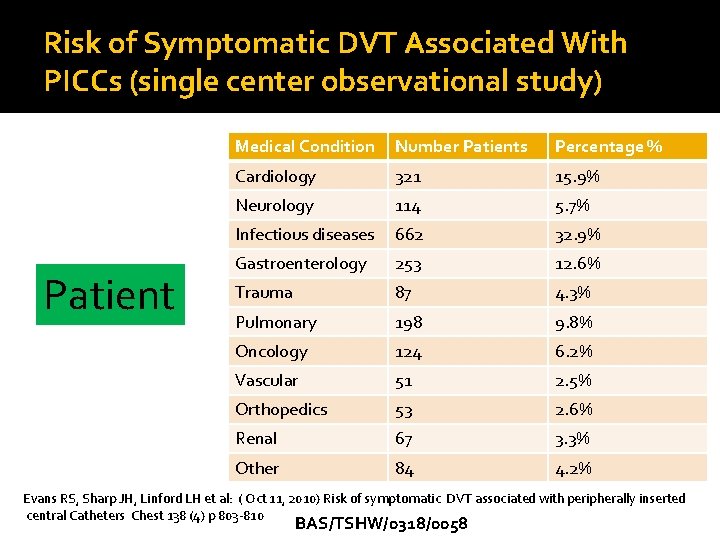
Risk of Symptomatic DVT Associated With PICCs (single center observational study) Patient Medical Condition Number Patients Percentage % Cardiology 321 15. 9% Neurology 114 5. 7% Infectious diseases 662 32. 9% Gastroenterology 253 12. 6% Trauma 87 4. 3% Pulmonary 198 9. 8% Oncology 124 6. 2% Vascular 51 2. 5% Orthopedics 53 2. 6% Renal 67 3. 3% Other 84 4. 2% Evans RS, Sharp JH, Linford LH et al: ( Oct 11, 2010) Risk of symptomatic DVT associated with peripherally inserted central Catheters Chest 138 (4) p 803 -810 BAS/TSHW/0318/0058

The ICU Patient Risk Evaluation BAS/TSHW/0318/0058

MAGIC: 2015 Appropriateness Guide �For peripherally compatible infusions, PICC use was rated as inappropriate when the proposed duration of use was 5 or fewer days �In critically ill patients, nontunneled central venous catheters were preferred over PICCs when 14 or fewer days of use were likely Chopra V. Flanders S. Saint S. (Sept. 2015)The Michigan Appropriateness Guide for Intravenous Catheters (MAGIC): Results From a Multispecialty Panel Using the RAND/UCLA Appropriateness Method, Annals of Internal Medicine 163(6) p 1 -39 S BAS/TSHW/0318/0058

Prevalent Risks of Placing and Removing PICCs & CICCs 7 PICC CLABSI 12 Brachial Arterial Injury Pneumothorax 12 Hematoma Air Embolism 12 Hemothorax Carotid/Subclavian/Femoral 2 Arterial Injury Hematoma Notes: Complications marked as “ 0%” are not included in lists. 7 Arterial injury expanded to include femoral artery based on Bowdle (2014) observations. 23 7 Dariushnia et al. Quality Improvement Guidelines for Central Venous Access. 2010. J Vasc Inter Radiol. 21. 976 -981 12 Hospital-Acquired Conditions and Present on Admission Indicator Reporting Provision. Centers for Medicare & Medicaid Services. ICN 901046 September 2014 2 Bowdle. Vascular Complications of Central Venous Catheter Placement: Evidence-Based Methods for Prevention and Treatment. 2014. J Cardiothoracic and Vascular Anesthesia. 21. 358 -368. BAS/TSHW/0318/0058

Patient Coagulation Considerations …and damaging the vein or artery Procedures with Low Risk of Bleeding, Easily Detected and Controllable (Category 1)20 Applicable Vascular Procedures Dialysis access intervention Central line removal PICC line placement Management related to line placement 20 � Platelet count: < 50 x 109 /L � International normalized ratio (INR): > 2. 0 CICC- large vein, large catheter PICC- small vein, small catheter Procedures with Moderate Risk of Bleeding (Category 2)20 Applicable Vascular Procedures Tunneled central venous catheter Subcutaneous port procedure � Platelet count: < 50 x 109 /L � International normalized ratio (INR): < 1. 5 20 Patel et al. Consensus Guidelines for Periprocedural Management of Coagulation Status and Hemostasis Risk in Percutaneous Image-guided Interventions. JVIR 2012. 23: 727 -736 BAS/TSHW/0318/0058
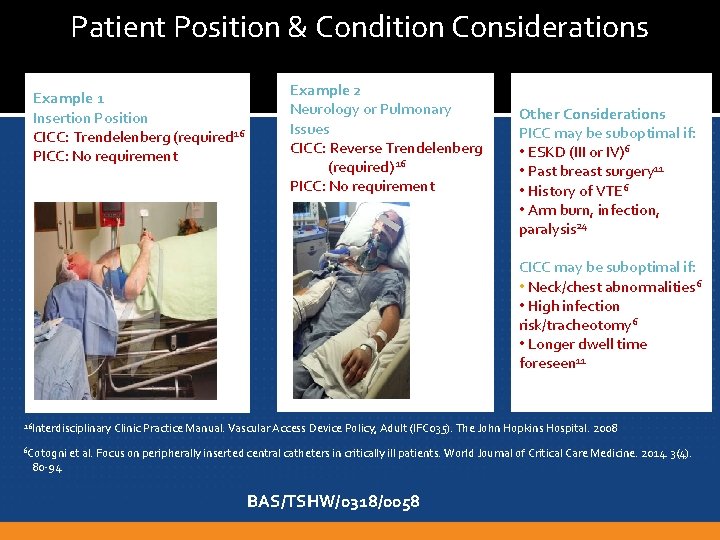
Patient Position & Condition Considerations Example 1 Insertion Position CICC: Trendelenberg (required 16 PICC: No requirement Example 2 Neurology or Pulmonary Issues CICC: Reverse Trendelenberg (required) 16 PICC: No requirement Other Considerations PICC may be suboptimal if: • ESKD (III or IV)6 • Past breast surgery 11 • History of VTE 6 • Arm burn, infection, paralysis 24 CICC may be suboptimal if: • Neck/chest abnormalities 6 • High infection risk/tracheotomy 6 • Longer dwell time foreseen 11 16 Interdisciplinary 6 Cotogni 80 -94 Clinic Practice Manual. Vascular Access Device Policy, Adult (IFC 035). The John Hopkins Hospital. 2008 et al. Focus on peripherally inserted central catheters in critically ill patients. World Journal of Critical Care Medicine. 2014. 3(4). BAS/TSHW/0318/0058

Required Life Supporting Devices: Competing for Real Estate Insertion Site Choice CICC • neck • clavicular triangle • groin PICC • either upper arm BAS/TSHW/0318/0058
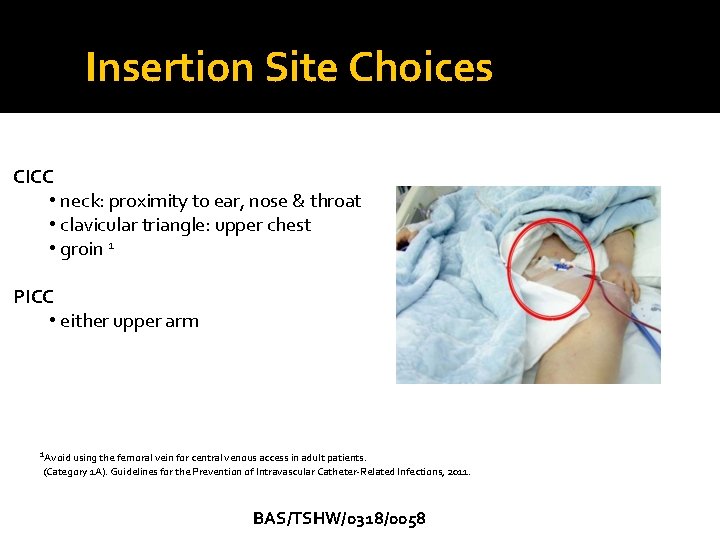
Insertion Site Choices CICC • neck: proximity to ear, nose & throat • clavicular triangle: upper chest • groin 1 PICC • either upper arm 1 Avoid using the femoral vein for central venous access in adult patients. (Category 1 A). Guidelines for the Prevention of Intravascular Catheter-Related Infections, 2011. BAS/TSHW/0318/0058
Insertion Site Choices CICC Jugular BAS/TSHW/0318/0058
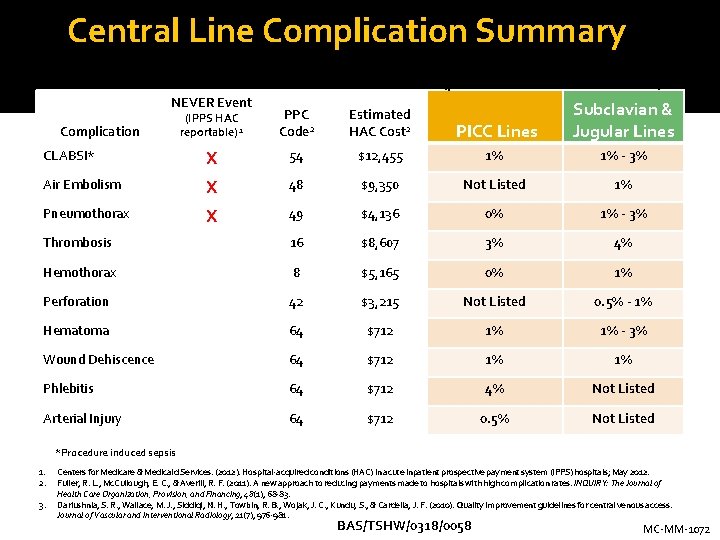
Central Line Complication Summary Incidence Rate (per SIR Central Venous Access CPG) 3 NEVER Event (IPPS HAC reportable) 1 PPC Code 2 Estimated HAC Cost 2 PICC Lines Subclavian & Jugular Lines CLABSI* X 54 $12, 455 1% 1% - 3% Air Embolism X 48 $9, 350 Not Listed 1% Pneumothorax X 49 $4, 136 0% 1% - 3% Thrombosis 16 $8, 607 3% 4% Hemothorax 8 $5, 165 0% 1% Perforation 42 $3, 215 Not Listed 0. 5% - 1% Hematoma 64 $712 1% 1% - 3% Wound Dehiscence 64 $712 1% 1% Phlebitis 64 $712 4% Not Listed Arterial Injury 64 $712 0. 5% Not Listed Complication *Procedure induced sepsis 1. 2. 3. Centers for Medicare & Medicaid Services. (2012). Hospital-acquired conditions (HAC) in acute inpatient prospective payment system (IPPS) hospitals; May 2012. Fuller, R. L. , Mc. Cullough, E. C. , & Averill, R. F. (2011). A new approach to reducing payments made to hospitals with high complication rates. INQUIRY: The Journal of Health Care Organization, Provision, and Financing, 48(1), 68 -83. Dariushnia, S. R. , Wallace, M. J. , Siddiqi, N. H. , Towbin, R. B. , Wojak, J. C. , Kundu, S. , & Cardella, J. F. (2010). Quality improvement guidelines for central venous access. Journal of Vascular and Interventional Radiology, 21(7), 976 -981. BAS/TSHW/0318/0058 MC-MM-1072

Key CVAD Patient Considerations q Status of bleeding q Status of ability to cooperate q Status of position for insertion and removal q Status of neck and chest q Status of breathing q Status of hemodynamics q Risk of infection with placement site BAS/TSHW/0318/0058
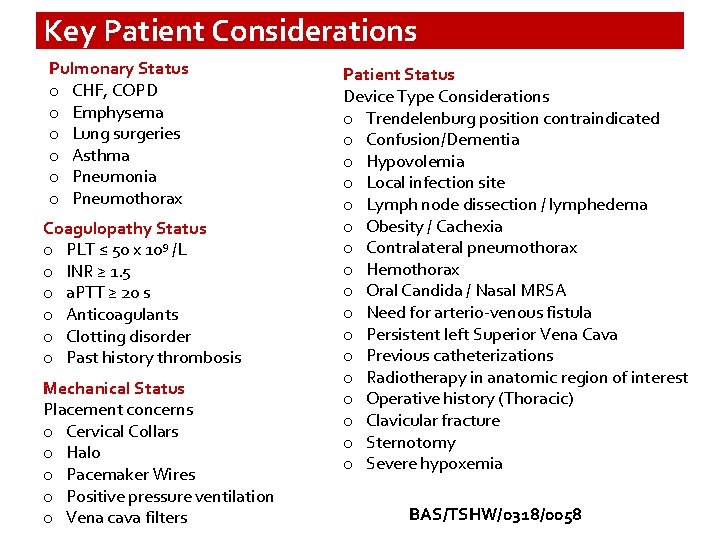
Key Patient Considerations Pulmonary Status o CHF, COPD o Emphysema o Lung surgeries o Asthma o Pneumonia o Pneumothorax Coagulopathy Status o PLT ≤ 50 x 109 /L o INR ≥ 1. 5 o a. PTT ≥ 20 s o Anticoagulants o Clotting disorder o Past history thrombosis Mechanical Status Placement concerns o Cervical Collars o Halo o Pacemaker Wires o Positive pressure ventilation o Vena cava filters Patient Status Device Type Considerations o Trendelenburg position contraindicated o Confusion/Dementia o Hypovolemia o Local infection site o Lymph node dissection / lymphedema o Obesity / Cachexia o Contralateral pneumothorax o Hemothorax o Oral Candida / Nasal MRSA o Need for arterio-venous fistula o Persistent left Superior Vena Cava o Previous catheterizations o Radiotherapy in anatomic region of interest o Operative history (Thoracic) o Clavicular fracture o Sternotomy o Severe hypoxemia BAS/TSHW/0318/0058

Annals of Internal Medicine (Sept. 2015) The Michigan Appropriateness Guide for Intravenous Catheters (MAGIC): From a Multidisciplinary Panel Using the RAND/UCLA Appropriateness Method Chopra V. Flanders S. Saint S. (Sept. 2015)The Michigan Appropriateness Guide for Intravenous Catheters (MAGIC): Results From a Multispecialty Panel Using the RAND/UCLA Appropriateness Method, Annals of Internal Medicine 163(6) p 1 -39 S BAS/TSHW/0318/0058

Decision-making: INS 2016 � Consider the infusate characteristics (eg, irritant, vesicant, osmolarity) in conjunction with anticipated duration of infusion therapy (eg, less than 6 days) and availability of peripheral vascular access sites. Irritant Vesicant osmolarity Peripheral access Duration Andrew Kokotis Infusion Nurses Society: (2016): Infusion Therapy Standards of Practice JIN (Jan/Feb) 39(1 S) BAS/TSHW/0318/0058

The following slides are based on personal real-world experience
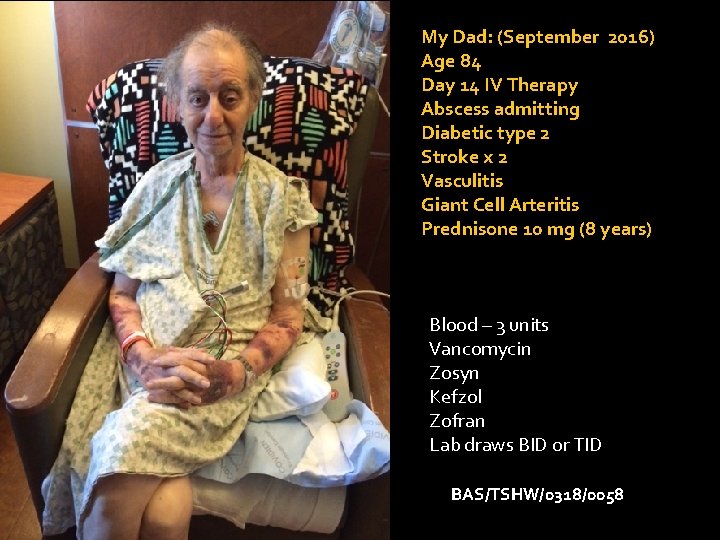
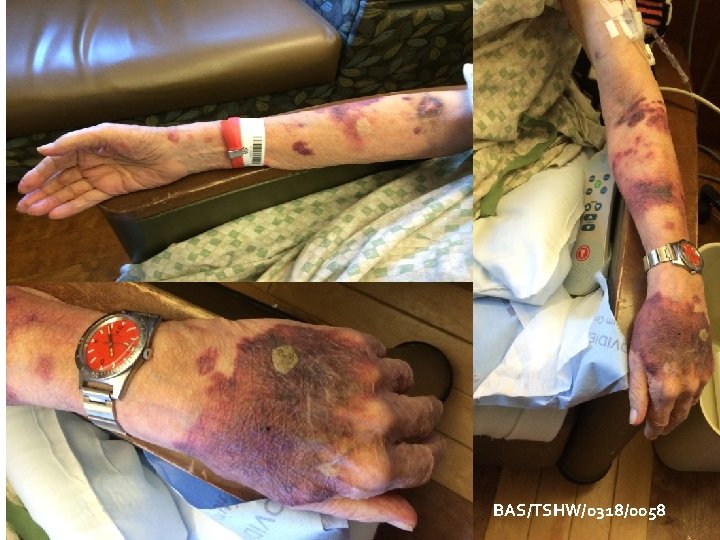
My Dad: (September 2016) Age 84 Day 14 IV Therapy Abscess admitting Diabetic type 2 Stroke x 2 Vasculitis Giant Cell Arteritis Prednisone 10 mg (8 years) Blood – 3 units Vancomycin Zosyn Kefzol Zofran Lab draws BID or TID BAS/TSHW/0318/0058
BAS/TSHW/0318/0058
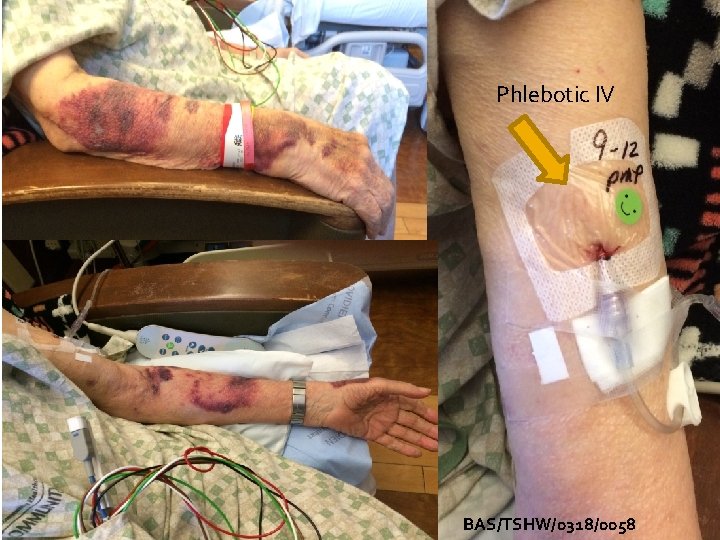
Phlebotic IV BAS/TSHW/0318/0058

My Mom Single lumen PICC for Sepsis BAS/TSHW/0318/0058 Did she get the right device?
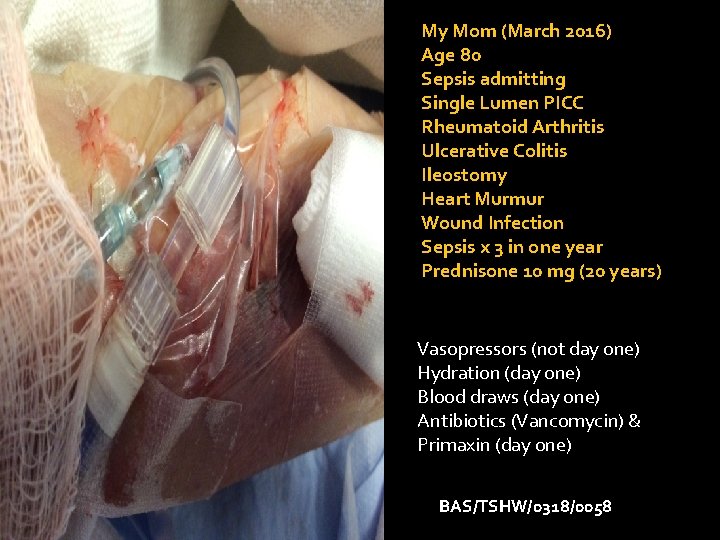
My Mom (March 2016) Age 80 Sepsis admitting Single Lumen PICC Rheumatoid Arthritis Ulcerative Colitis Ileostomy Heart Murmur Wound Infection Sepsis x 3 in one year Prednisone 10 mg (20 years) Vasopressors (not day one) Hydration (day one) Blood draws (day one) Antibiotics (Vancomycin) & Primaxin (day one) BAS/TSHW/0318/0058
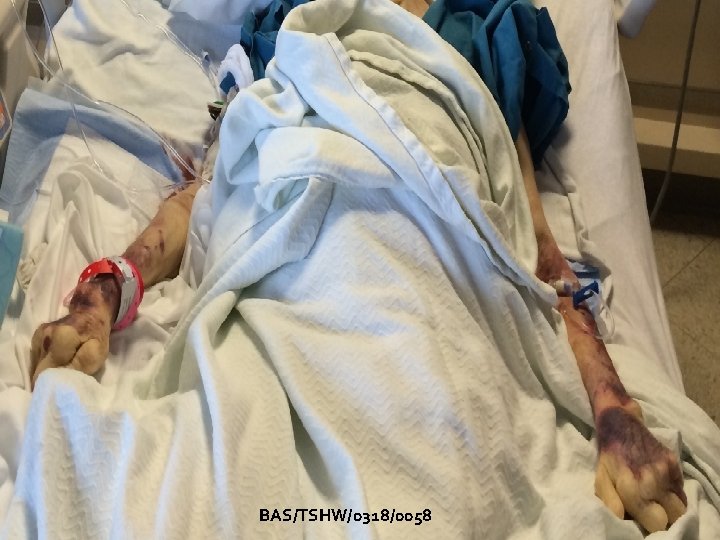
BAS/TSHW/0318/0058
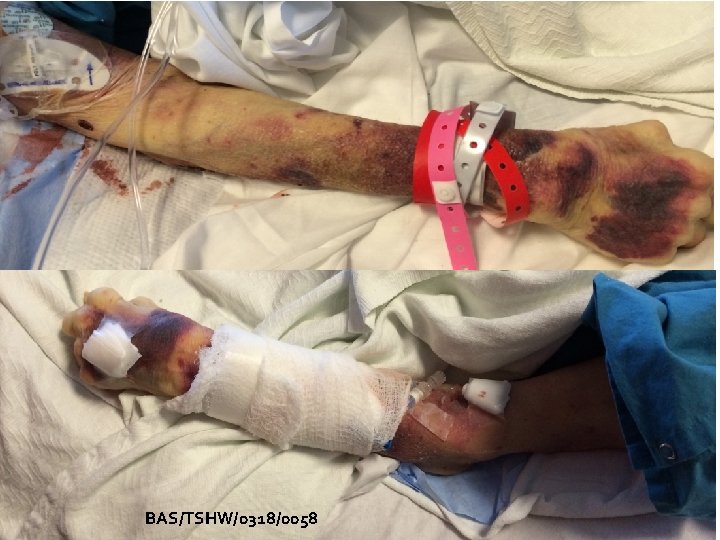
BAS/TSHW/0318/0058

Venous Access Device Recommendations for Infusion of Peripherally Compatible Infusate Chopra V. Flanders S. Saint S. (Sept. 2015)The Michigan Appropriateness Guide for Intravenous Catheters (MAGIC): Results From a Multispecialty Panel Using the RAND/UCLA Appropriateness Method, Annals of Internal Medicine 163(6) p 1 -39 S BAS/TSHW/0318/0058

Venous Access Device Recommendations for Patients with Difficult Venous Access Both my mom and dad were difficult access Chopra V. Flanders S. Saint S. (Sept. 2015)The Michigan Appropriateness Guide for Intravenous Catheters (MAGIC): Results From a Multispecialty Panel Using the RAND/UCLA Appropriateness Method, Annals of Internal Medicine 163(6) p 1 -39 S BAS/TSHW/0318/0058

Venous Access Device Recommendations for Infusion of Peripherally Non-Compatible Infusate My Dad received Vancomycin a Vesicant Drug Chopra V. Flanders S. Saint S. (Sept. 2015)The Michigan Appropriateness Guide for Intravenous Catheters (MAGIC): Results From a Multispecialty Panel Using the RAND/UCLA Appropriateness Method, Annals of Internal Medicine 163(6) p 1 -39 S BAS/TSHW/0318/0058

Venous Access Device Recommendations for Patients Who Require Frequent Phlebotomy My dad and mom both Had frequent blood draws My mom was not in the ICU on admit My mom and dad Chopra V. Flanders S. Saint S. (Sept. 2015)The Michigan Appropriateness Guide for Intravenous Catheters (MAGIC): Results From a Multispecialty Panel Using the RAND/UCLA Appropriateness Method, Annals of Internal Medicine 163(6) p 1 -39 S BAS/TSHW/0318/0058

Potential Alternatives MOM – DIFFICULT ACCESS DAD – DIFFICULT ACCESS � Vancomycin - Vesicant � Sepsis – potential for ICU � Repeated blood draws � Single lumen PICC was � Vancomycin – Vesicant � Repeated blood draws � Original length of inappropriate? � Dual or Triple Lumen PICC should have been placed? � My mom ended in ICU on vasoconstrictors treatment was supposed to be < 6 days � Single lumen PICC Line should have been placed? BAS/TSHW/0318/0058
Decision Making Basics Length of Therapy Infusates Location of Therapy Vascular Access Decision BAS/TSHW/0318/0058 Patient

Conclusion: Decision Making INS (2016) �Assess the patient’s condition age diagnosis comorbidities condition of the vasculature at the insertion �site and proximal to the intended insertion site; condition of skin at intended insertion site; history of previous venipunctures and access devices; type and �duration of infusion therapy � patient preference for VAD site selection. Infusion Nurses Society: (2016): Infusion Therapy Standards of Practice JIN (Jan/Feb) 39(1 S BAS/TSHW/0318/0058

BAS/TSHW/0318/0058

References Avoid using the femoral vein for central venous access in adult patients. (Category 1 A). Guidelines for the Prevention of Intravascular Catheter-Related Infections, 2011. 2. Bowdle. Vascular Complications of Central Venous Catheter Placement: Evidence-Based Methods for Prevention and Treatment. 2014. J Cardiothoracic and Vascular Anesthesia. 21. 358 -368 Centers for Medicare & Medicaid Services. (2012). Hospital-acquired conditions (HAC) in acute inpatient prospective payment system (IPPS) hospitals; May 2012. 3. Chopra V. Flanders S. Saint S. (Sept. 2015)The Michigan Appropriateness Guide for Intravenous Catheters (MAGIC): Results From a Multispecialty Panel Using the RAND/UCLA Appropriateness Method, Annals of Internal Medicine 163(6) p 1 -39 S 4. Chopra V, O’Horo JC. Rogers M (Sept. 2013) The Risk of Bloodstream Infection Associated with Peripherally Inserted Central Catheters Compared with Central Venous Catheters in Adults: A Systematic Review and Meta-Analysis Author(s): Source: Infection Control and Hospital Epidemiology, Vol. 34, No. 9 (September 2013), pp. 908 - 918 Published by: The University of Chicago Press on behalf of The Society for Healthcare Epidemiology of America Stable URL: http: //www. jstor. org/stable/10. 1086/671737 Accessed: 07/08/2013 13: 40 5. Chopra V, Anand SA, Buist, Rogers M, Saint S, Flanders : July 27, 2013) Risk of venous thromboembolism associated with peripherally Inserted central catheters: A systematic review and meta=analysis Lancet 382 p 311 -325 1. BAS/TSHW/0318/0058

References 6. 7. 8. 9. 10. 11. 12. 13. 14. 15. 16. Cotogni et al. Focus on peripherally inserted central catheters in critically ill patients. World Journal of Critical Care Medicine. 2014. 3(4). 80 -94 Dariushnia et al. Quality Improvement Guidelines for Central Venous Access. 2010. J Vasc Inter Radiol. 21. 976 -981 Fuller, R. L. , Mc. Cullough, E. C. , & Averill, R. F. (2011). A new approach to reducing payments made to hospitals with high complication rates. INQUIRY: The Journal of Health Care Organization, Provision, and Financing, 48(1), 68 -83. Gahart B, Nazareno AR, Ortega MQ: (2017) Gaharts 2017 Intravenous Medications A Handbook for Nurses and Health Professionals 33 rd Elsevier St Louis Missouri Gorski LA, Stranz M, Cook LS, et al. : (2017) Development of an evidence-based list of noncytotoxic vesicant medications And solutions JIN 40(1) Jan/Feb 26 -40 Helen Hamilton and Andrew R. Bodenham. Central Venous Catheters. (United Kingdom. John Wiley & Sons Ltd. 2009) 38 -84 Hospital-Acquired Conditions and Present on Admission Indicator Reporting Provision. Centers for Medicare & Medicaid Services. ICN 901046 September 2014 Infusion Nurses Society: (2011) Infusion Nursing Standards of Practice, JIN 34 (1 S)S p. 37 Infusion Nurses Society: (2016): Infusion Therapy Standards of Practice JIN (Jan/Feb) 39(1 S Infusion Nurses Society: (2016) Noncytoxic Vesicant Medications and Solutions www. INS 1. org on the ONS Learning Center Tab Clinic Practice Manual. Vascular Access Device Interdisciplinary Clinic Practice Manual. Vascular Access Device Policy, Adult (IFC 035). The John Hopkins Hospital. 2008 BAS/TSHW/0318/0058

References 17. 18. 19. 20. 21. 22. Nanjappa S, Snyder et al: (2017) Vancomycin Infiltrate-Induced Dermatitis Mimicking Bullous Cellulits, Journal of drugs in Dermatology, 16(11) 1160 -1163 Nolan ME, et al. Complication rates among peripherally inserted central venous catheters and centrally inserted central catheters in the medical intensive care unit. J Crit Care 2015; 31: 238 -242 Optiray injection (loversolinjection): Side Effects, Interactions, Warning, Dosage & Usage www. rxlist. com/optiray-injection-drug. htm 1 -26 Patel et al. Consensus Guidelines for Periprocedural Management of Coagulation Status and Hemostasis Risk in Percutaneous Image-guided Interventions. JVIR 2012. 23: 727 -736 Peyko V, Saasson E: (2016) Vancomycin extravasation: evaluation, treatment, and avoidance of This adverse drug event, Case Reports in Internal Medicine 3(3) p. 40 -43 Wilson TJ, Brown DL, Meurer WJ et al: (2012) Risk factors associated with peripheral inserted central venous catheter-related large vein Thrombosis in neurological intensive care patient BAS/TSHW/0318/0058

Questions © 2018 BD. BD and the BD Logo are trademarks of Becton, Dickinson and Company. Please consult product labels and inserts for any indications, contraindications, hazards, warnings, precautions, and directions for use. BAS/TSHW/0318/0058
- Slides: 69