K S SCHOOL OF ENGINEERING AND MANAGEMENT Basic

K. S. SCHOOL OF ENGINEERING AND MANAGEMENT Basic Thermodynamics (18 ME 33) Module 3 ‘Second Law of Thermodynamics’ Session delivered by: - Vinod A Assistant Professor Department of Mechanical Engineering K. S. S. E. M
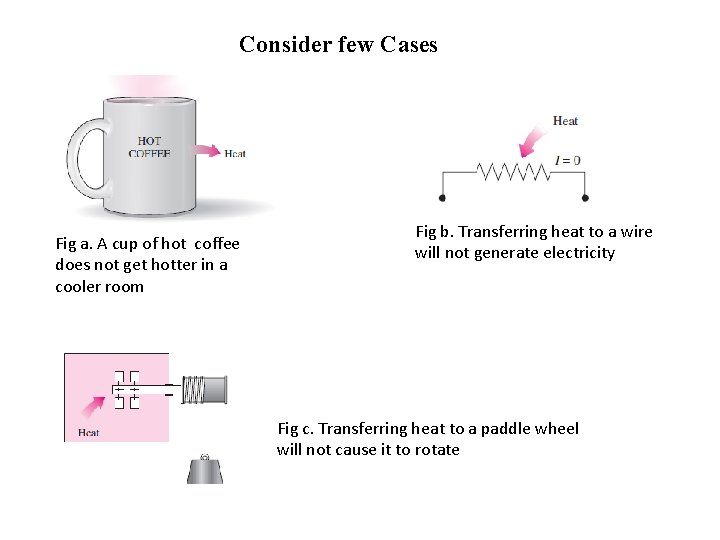
Consider few Cases Fig a. A cup of hot coffee does not get hotter in a cooler room Fig b. Transferring heat to a wire will not generate electricity Fig c. Transferring heat to a paddle wheel will not cause it to rotate

Observations from the above examples • It is clear that the process proceeds in certain direction and not in reverse direction. • Satisfying first law of thermodynamics does not ensure that the process can actually occur. • A process cannot occur unless it satisfies both the First law and second law of thermodynamics
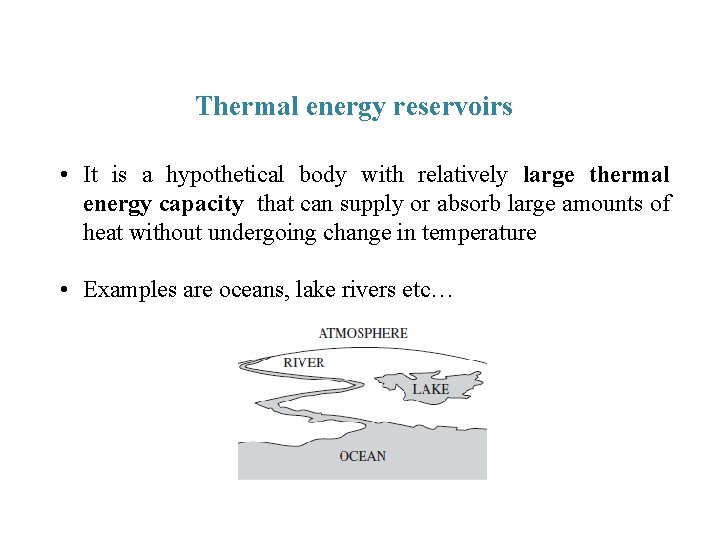
Thermal energy reservoirs • It is a hypothetical body with relatively large thermal energy capacity that can supply or absorb large amounts of heat without undergoing change in temperature • Examples are oceans, lake rivers etc…

• A reservoir which supplies energy in the form of heat is called Source • A reservoir which absorbs energy in the form heat is called Sink
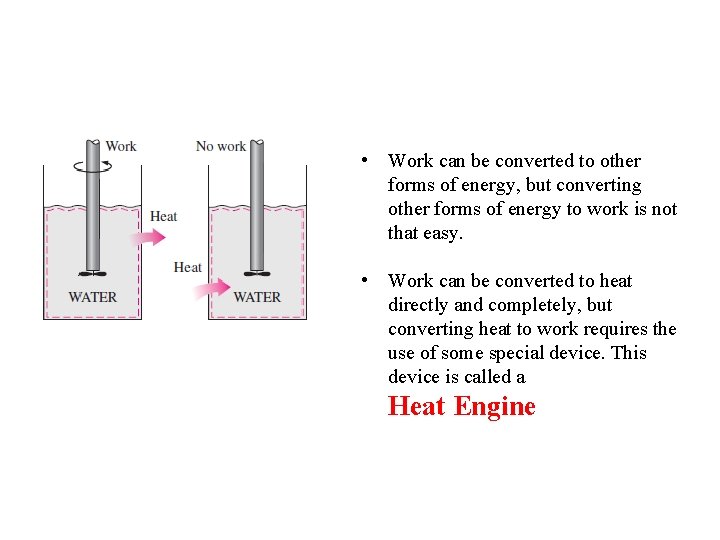
• Work can be converted to other forms of energy, but converting other forms of energy to work is not that easy. • Work can be converted to heat directly and completely, but converting heat to work requires the use of some special device. This device is called a Heat Engine
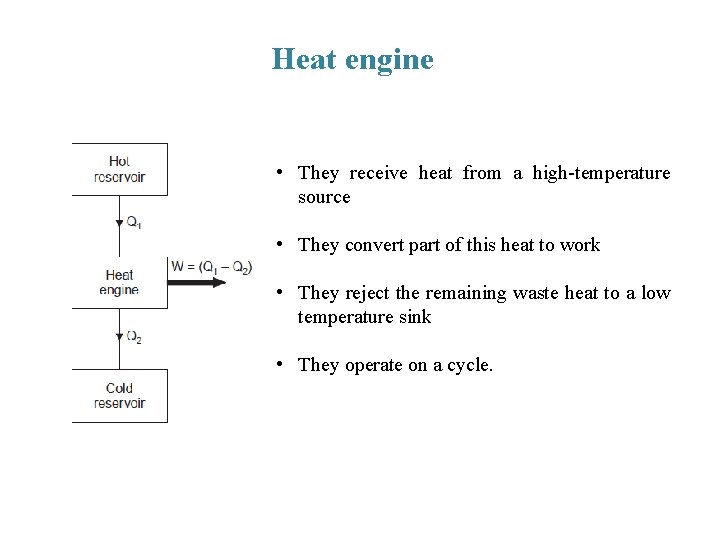
Heat engine • They receive heat from a high-temperature source • They convert part of this heat to work • They reject the remaining waste heat to a low temperature sink • They operate on a cycle.
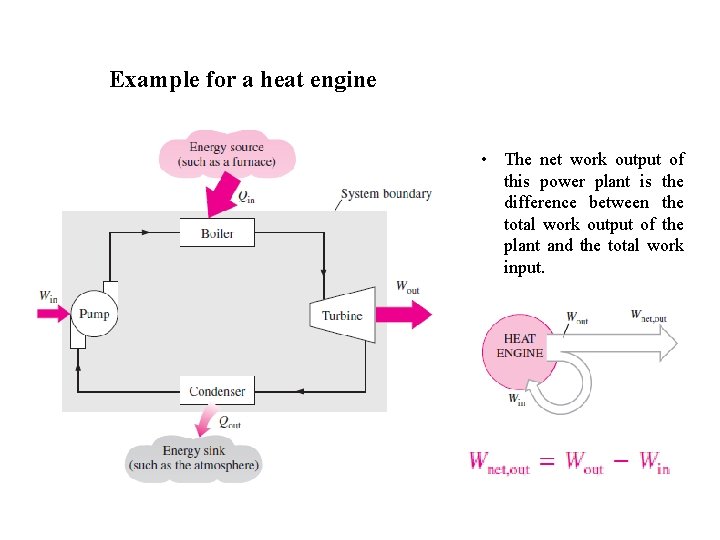
Example for a heat engine • The net work output of this power plant is the difference between the total work output of the plant and the total work input.
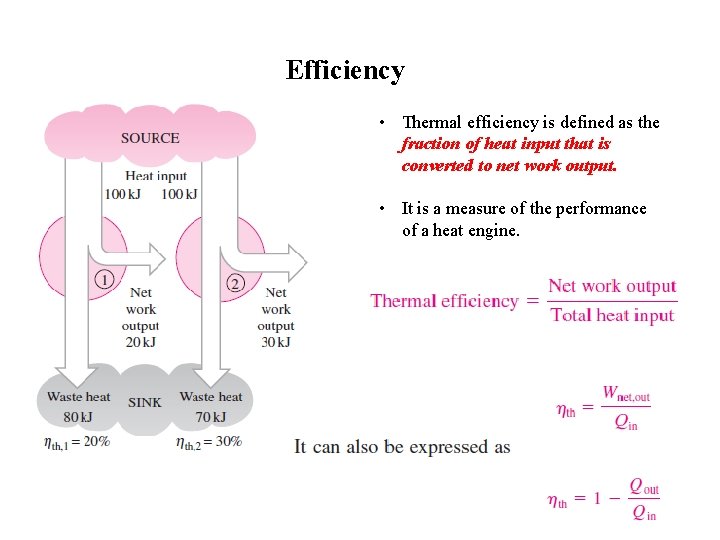
Efficiency • Thermal efficiency is defined as the fraction of heat input that is converted to net work output. • It is a measure of the performance of a heat engine.
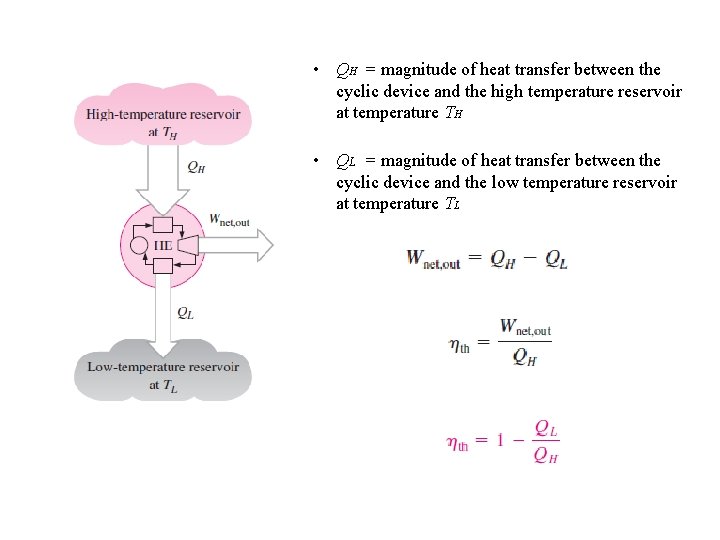
• QH = magnitude of heat transfer between the cyclic device and the high temperature reservoir at temperature TH • QL = magnitude of heat transfer between the cyclic device and the low temperature reservoir at temperature TL

Important points on thermal efficiency • Thermal efficiency is a measure of how efficiently a heat engine converts the heat that it receives to work. • Engineers are constantly trying to improve the efficiencies of these devices since increased efficiency means less fuel consumption and thus lower fuel bills and less pollution. • The thermal efficiencies of work producing devices are relatively low. • Spark ignition engine have efficiency of about 25% • Diesel engines have about 40%. • Gas-steam plants have about 60%
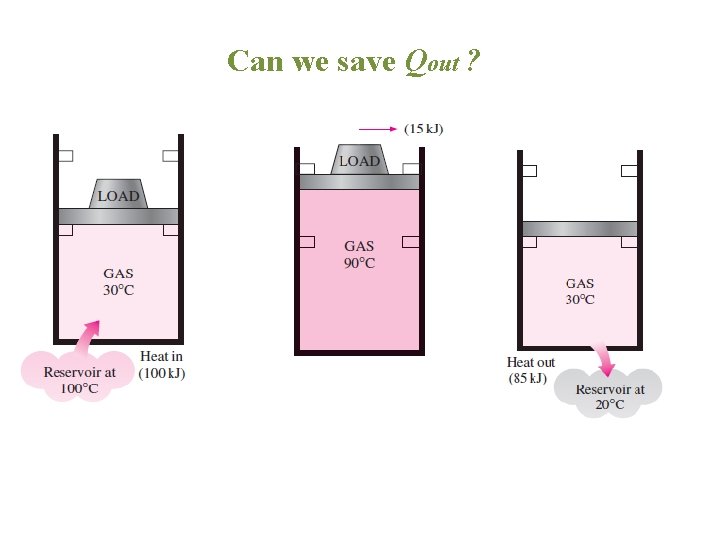
Can we save Qout ?

THE SECOND LAW OF THERMODYNAMICS KELVIN-PLANCK STATEMENT “ It is impossible for any device that operates on a cycle to receive heat from a single reservoir and produce a net amount of work” Fig:

Refrigerators and Heat pumps • Heat is transferred in the direction of decreasing temperature. This heat transfer process occurs in nature without requiring any devices. • The reverse process however, cannot occur by itself. The transfer of heat from a low temperature medium to a high temperature one requires special devices called Refrigerators and Heat pump.
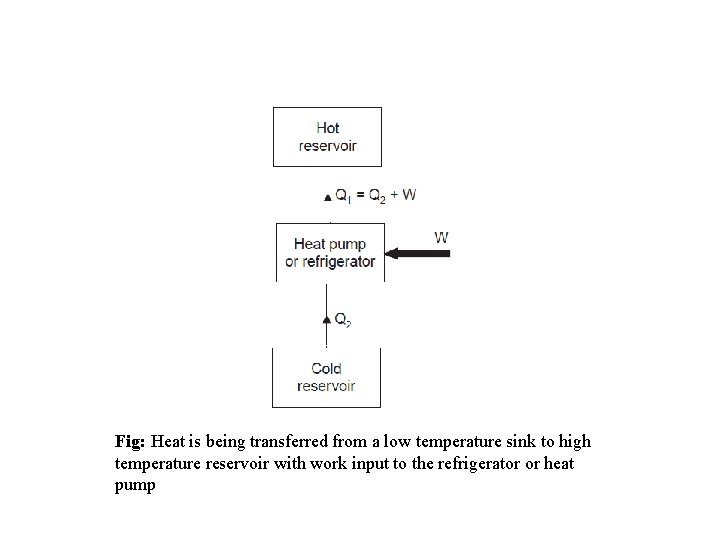
Fig: Heat is being transferred from a low temperature sink to high temperature reservoir with work input to the refrigerator or heat pump
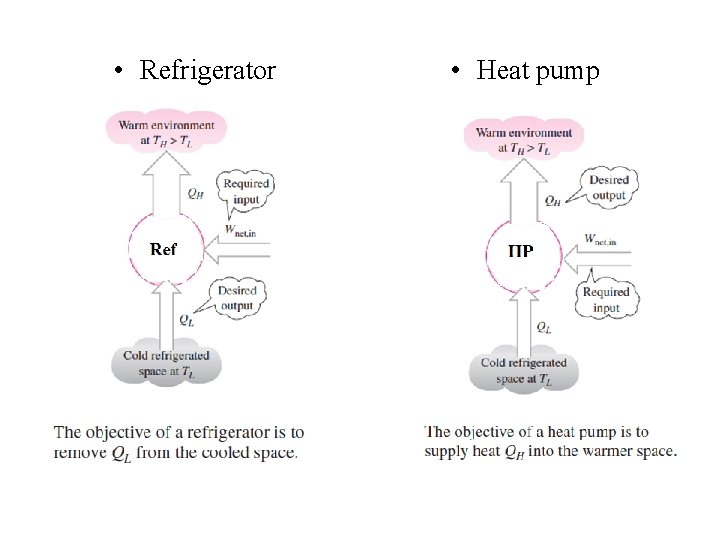
• Refrigerator • Heat pump

Coefficient of Performance • The performance of a refrigerator or a heat pump is expressed in terms of the coefficient of performance (COP).

THE SECOND LAW OF THERMODYNAMICS CLAUSIUS STATEMENT “It is impossible to construct a device that operates in a cycle and produces no effect other than the transfer of heat from a lower temperature body to a higher temperature body” Fig:

Equivalence of two statements Although the Clausius and Kelvin-planck statememt appear to be different they are really equivalent in the sense that a violation of either statement implies violation of other
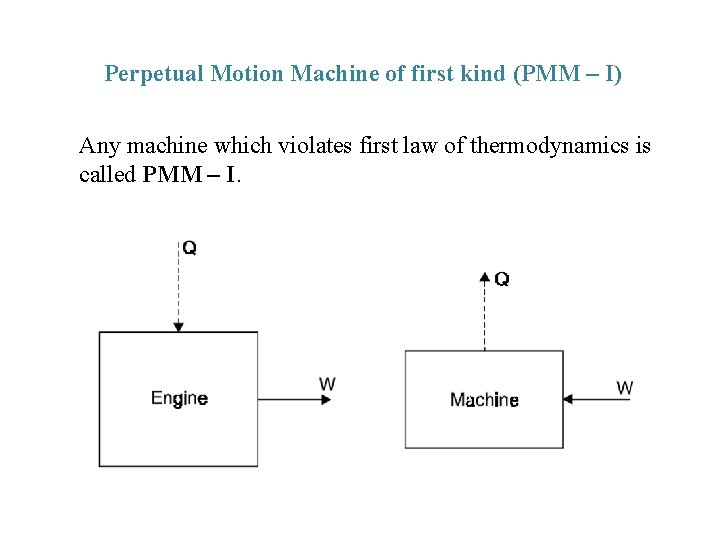
Perpetual Motion Machine of first kind (PMM – I) Any machine which violates first law of thermodynamics is called PMM – I.

Perpetual Motion Machine of second kind (PMM – II) • Any machine which violates second law of thermodynamics is called PMM – II.
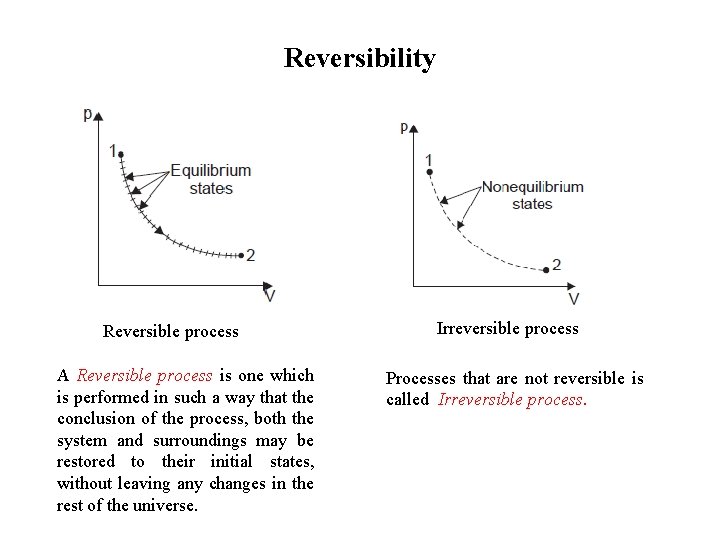
Reversibility Reversible process A Reversible process is one which is performed in such a way that the conclusion of the process, both the system and surroundings may be restored to their initial states, without leaving any changes in the rest of the universe. Irreversible process Processes that are not reversible is called Irreversible process.

Causes of Irreversibility The irreversibility of a process may be due to either one or both of the following: 1. Lack of Equilibrium during the process. a. Heat transfer through a finite temperature difference b. Lack of pressure equilibrium within the interior of the system or between the system and the surroundings c. Free expansion 2. Irreversibility due to dissipative effects a. Friction b. paddle-wheel work transfer c. Transfer of electricity through a resistor

Carnot Cycle The cycle was first suggested by a French Engineer Sadi Carnot in 1824 which works on reversible cycle and is known as Carnot Cycle. The Assumptions are: 1. The piston moving in a cylinder does not develop any friction during motion 2. The walls of piston and cylinder are considered as perfect insulators of heat 3. The cylinder head is so arranged that it can be perfect heat conductor or perfect heat insulator 4. Working medium is a perfect gas and has constant specific heat 5. Compression and expansion are reversible

Carnot Cycle A Reversible Cycle or Carnot Cycle is an ideal cycle in which all the processes constituting the cycle are reversible. Carnot cycle is a reversible cycle Process 1 -2: Isothermal Expansion Process 2 -3: Isentropic Expansion Process 3 -4: Isothermal compression Process 4 -1: Isentropic Compression

Reversed Heat Engine Or Reversed Carnot Cycle In this, the cycle remains exactly same, except that the directions of any heat and work interactions are reversed: Heat in the amount (QL) is absorbed from the low temperature reservoir Heat in the amount (QH) is rejected to a high temperature reservoir Wnet, in is required to accomplish all this
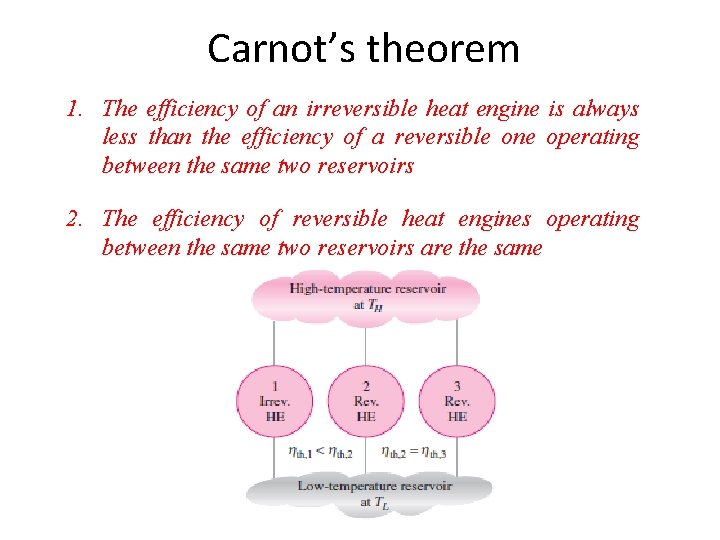
Carnot’s theorem 1. The efficiency of an irreversible heat engine is always less than the efficiency of a reversible one operating between the same two reservoirs 2. The efficiency of reversible heat engines operating between the same two reservoirs are the same
- Slides: 27