K FLORENCE PGT BIOLOGY KKV1 NARIMEDU MADURAI CHENNAI

K. FLORENCE, PGT- BIOLOGY KKV-1, NARIMEDU, MADURAI CHENNAI REGION

Applications of Biotechnology in Medicine 1. To produce therapeutic drugs, vaccines, antibiotics, & hormones(Genetically engineered insulin) 2. To treat diseases(Gene therapy) 3. Molecular Diagnosis of diseases
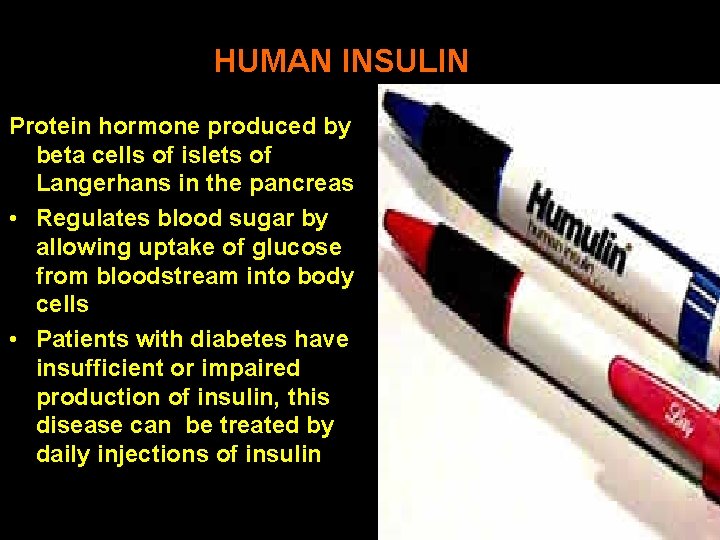
HUMAN INSULIN Protein hormone produced by beta cells of islets of Langerhans in the pancreas • Regulates blood sugar by allowing uptake of glucose from bloodstream into body cells • Patients with diabetes have insufficient or impaired production of insulin, this disease can be treated by daily injections of insulin
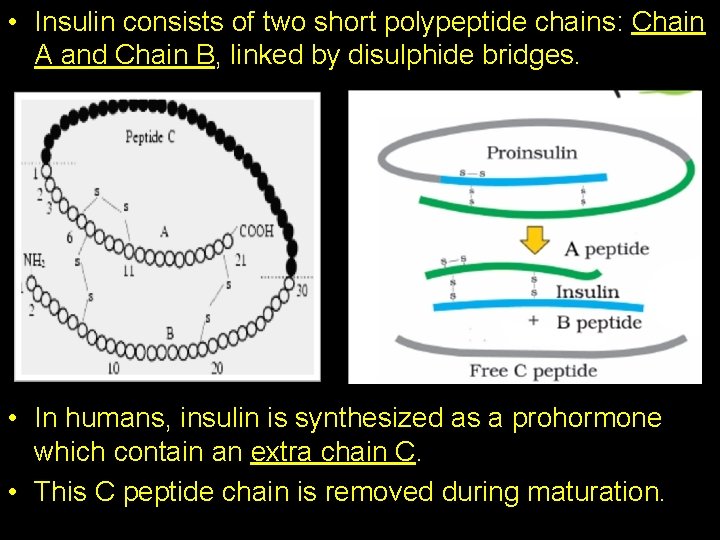
• Insulin consists of two short polypeptide chains: Chain A and Chain B, linked by disulphide bridges. • In humans, insulin is synthesized as a prohormone which contain an extra chain C. • This C peptide chain is removed during maturation.


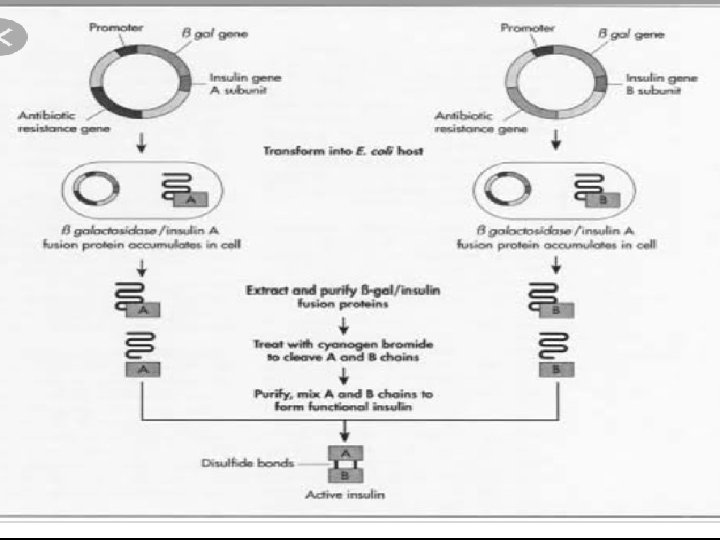
GENETICALLY ENGINEERED INSULIN • Genetically engineered insulin was produced using r-DNA techniques in 1983 by an American company named ELI LILLY. • They took the DNA sequences that code for polypeptide A and polypeptide B. • They introduced them in plasmids of E. coli and grown separately to produce insulin chain. • After growing they extracted them and combined by disulfide bonds to form human insulin. • Since it does not contain polypeptide c it is in active form and does not affect humans.

WHAT IS GENE THERAPY The most common form of gene therapy involves inserting a normal gene to replace an abnormal gene • Introducing a new gene into the body to help fight a disease. Give a chance of a normal life to baby born with genetic disease. Give hope of healthy life to cancer patient. For certain disease that do not have any cure except gene therapy, it could save many lives

Gene Therapy • Replace a gene by a normal healthy functional gene. • Example: ADA - Adenosine Deaminase Deficiency • Gene Therapy - In 1990, the first trial of actual gene therapy was conducted in USA. • A little (4 years old)' girl suffering with adenosine deaminase deficiency (ADA), a lethal disorder, was given genetically modified lymphocytes bearing the ADA gene. • This disease is caused due to deletion of the gene for the enzyme adenosine deaminase (ADA). • This enzyme is necessary for the immune system to function properly.

• ADA deficiency can be cured by: Gene Therapy ü Bone Marrow Transplantation ü Enzyme Replacement Therapy But they are not completely curative. • In gene therapy, lymphocytes are extracted from the blood of the patient. • A good copy of the human gene encoding ADA enzyme is introduced into these cells using a retroviral vector. • The cells thus treated are then re-injected to the patients. • Lymphocytes now produced have a fully functional ADA gene and they produce ADA to help the patients immune system.
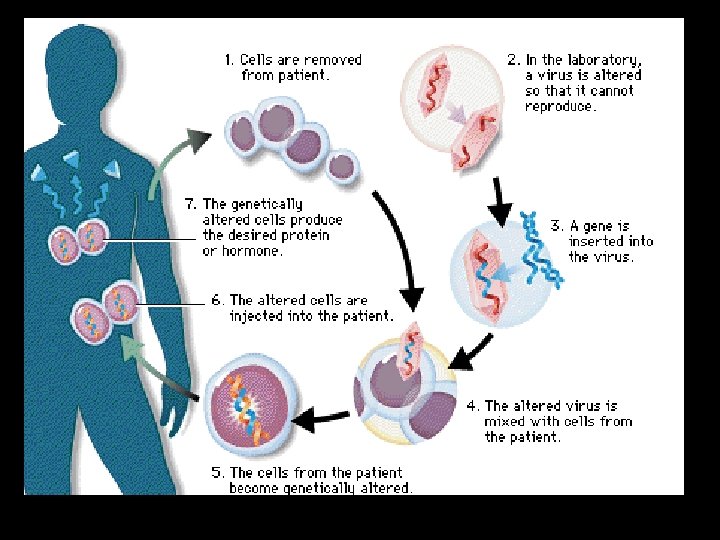

• As these cells are not immortal, the patient requires periodic infusion of such genetically engineered lymphocytes. • However, if the gene isolate from marrow cells producing ADA is introduced into cells at early embryonic stages, it could be a permanent cure.

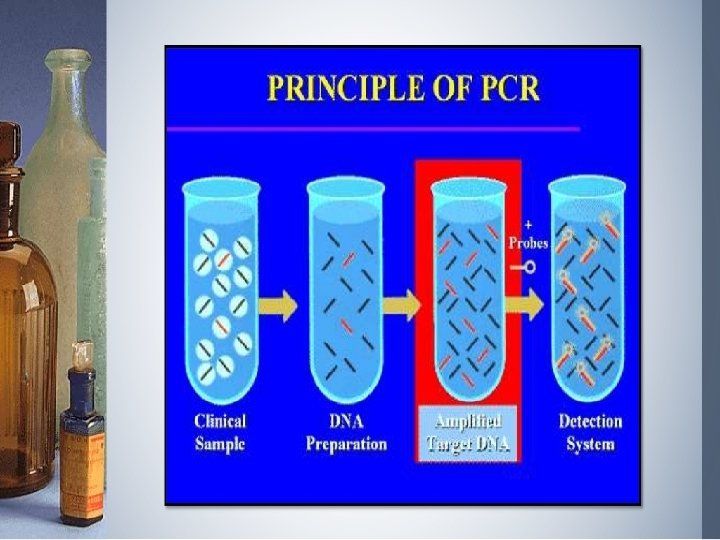
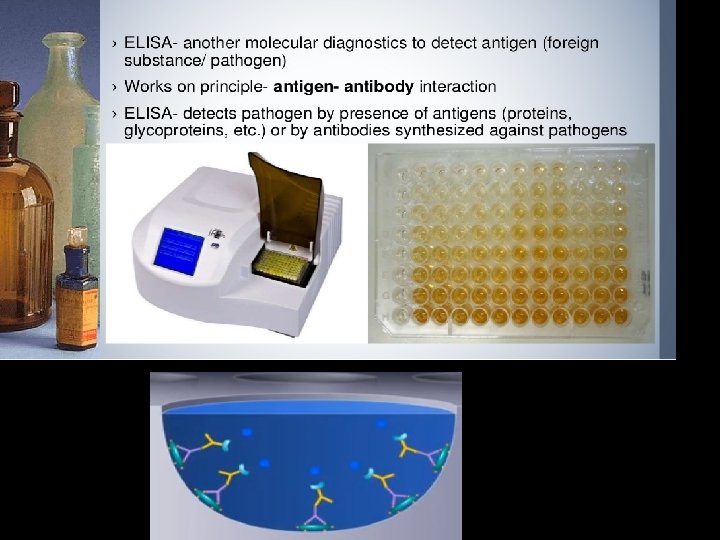
Molecular diagnosis of diseases • Using conventional methods of diagnosis (Serum and Urine analysis etc. ) early detection is not possible. • Now-a-days, many techniques are used to diagnose the presence of pathogen at very early stage such as: q. Recombinant DNA Technology, q. Polymerase Chain Reaction (PCR) q. Enzyme Linked Immuno - Sorbent Assay (ELISA)

Polymerase Chain Reaction (PCR) • Very low concentration of a bacteria or virus can be detected by amplification of their nucleic acid by PCR. • PCR can be used : To detect HIV in suspected AIDS patients To detect mutation in genes in cancer patients. Other genetic disorders

• ELISA • Enzyme Linked Immuno-sorbent Assay • https: //www. youtube. com/watch? v=l. UWp. WKVcmc 4 • https: //www. youtube. com/watch? v=YJ 0 -q. Qslqq. Q

Recombinant DNA Technology • A single stranded DNA or RNA, tagged with a radioactive molecule(probe) is allowed to hybridise to its complementary DNA in a clone of cells followed by detection using autoradiography. • The clone having the mutated gene will hence not appear on the photographic film, because the probe will not have complimentary with the mutated gene. • using autoradiography. • The clone having the mutated gene will hence not appear on the photographic film, because the probe will not have complimentary with the mutated gene.

RECAPITULATION 1. Mention two applications of biotechnology 2. PCR is a powerful diagnostic technique. justify 3. Name the diagnostic test for AIDS 4. What are the methods by which ADA deficiency can be cured Which method is used to cure the disease permanently. 5. In what way insulin produced by our body is different from the one produced by Eli lilly company 6. How many therapeutic drugs are approved in India? 7. What causes ADA deficiency? 8. How maturation of insulin takes place in our body

ho ASSIGNMENT Ø 1. Identify some approved therapeutic available in the market Ø 2. What are the diseases for which gene replacement therapy is carried out , other than ADA deficiency 3. Name some medically useful products of biotechnology other than insulin.

Replacement strategy Gene with defect Adenosine deaminase (ADA) a-1 -antitrypsin Disease/Disorder SCID Sever combined immunodeficiency Emphysema CF transmembrane regulator Cystic fibrosis Clotting factor VIII Hemophilia A Clotting factor IX Hemophilia B b-chain of hemoglobin Sickle cell anemia

Some Approved Therapeutics • Hormones – Growth hormone, Insulin, Calcitonin, FSH • Cytokines – Interferons (Interleukins), Neurotrophic factors EPO, CSF, • Clotting factors – Factor VIII, Factor IX • Vaccines – Hepatitis B, acellular pertussis vaccine (Bordatella pertussis, whooping cough) • Monoclonal antibodies

Therapeutic products approved in India v Granulocyte macrophage colony stimulating factor v Human Insulin v Alpha-interferon, v Streptokinase v Gamma-Interferon, v Erythropoietin v Blood factor VIII v Hepatitis B vaccine v Follicle stimulating hormone v Human growth v Granulocyte colony hormone v Human interleukin stimulating factor (GCSF), . Shantha Biotechnies Pvt. Ltd. , Bharat Biotech International, Wockhardt, Reddy Labs and Panacea Biotech, Biological E limited, Virchow Biotechniques etc. have initiated the production of several of these therapeutics and some growth factors in our cou ntry


REFERENCES Biotechnology in medicine & agriculture Written By—Charles Molnar Link www. ebe. biopharma. eu www. omicsonline. org

FLORENCE NO-1, MADURAI
- Slides: 26