Journal Club Whitmer RA Karter AJ Yaffe K

Journal Club Whitmer RA, Karter AJ, Yaffe K, Quesenberry CP Jr, Selby JV. Hypoglycemic episodes and risk of dementia in older patients with type 2 diabetes mellitus. JAMA. 2009 Apr 15; 301(15): 1565 -72. Young LH, Wackers FJ, Chyun DA, Davey JA, Barrett EJ, Taillefer R, Heller GV, Iskandrian AE, Wittlin SD, Filipchuk N, Ratner RE, Inzucchi SE; DIAD Investigators. Cardiac outcomes after screening for asymptomatic coronary artery disease in patients with type 2 diabetes: the DIAD study: a randomized controlled trial. JAMA. 2009 Apr 15; 301(15): 1547 -55. 2009年 5月7日 8: 30 -8: 55 8階 医局 埼玉医科大学 総合医療センター 内分泌・糖尿病内科 Department of Endocrinology and Diabetes, Saitama Medical Center, Saitama Medical University 松田 昌文 Matsuda, Masafumi

Diabetes Center, Massachusetts General Hospital, Harvard Medical School, Boston, Massachusetts. Couri et al describe long-term follow-up and insulin secretion status of patients from their original study, in which they performed nonmyeloablative autologous stem cell transplantation to treat very recent–onset type 1 diabetes. Clinicians are left with trying to achieve an Hb. A 1 c goal of less than 7%, which reduces microvascular and cardiovascular disease over time, but no rationale to try to achieve an Hb. A 1 c of less than 6%if more than lifestyle changes are required. Based on an analysis of registry data from a large ongoing epidemiologic cohort, the authors report that among elderly patients with type 2 diabetes who had no documented prior diagnoses of dementia, cognitive impairment, or memory complaints, episodes of hypoglycemia severe enough to result in emergency department or hospital treatment were associated with increased risk of being diagnosed with dementia. The DIAD study results suggest that more aggressive screening for coronary artery disease does not appear to improve the outcome of asymptomatic individuals with type 2 diabetes. In the observational study by Kosiborod et al, the occurrence of hypoglycemia was associated with higher mortality among patients who did not receive insulin (compared with patients who did not experience hypoglycemia), but not in patients who were treated with insulin.

Hypoglycemic Episodes and Risk of Dementia in Older Patients With Type 2 Diabetes Mellitus JAMA. 2009; 301(15): 1565 -1572. JAMA Report Video This JAMA Report summarizes key findings from the article in video format and includes an interview with one of the authors. The video script and links to the video are available at http: //pubs. ama-assn. org/media/2009 j/0414. dtl#vnrscript.
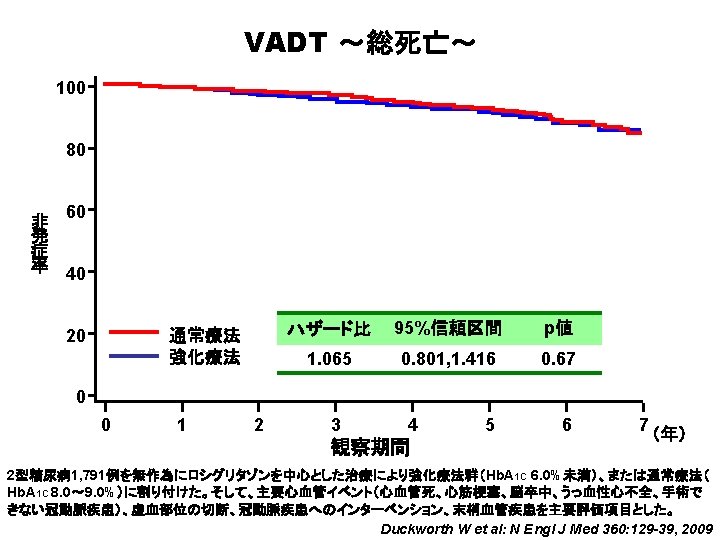
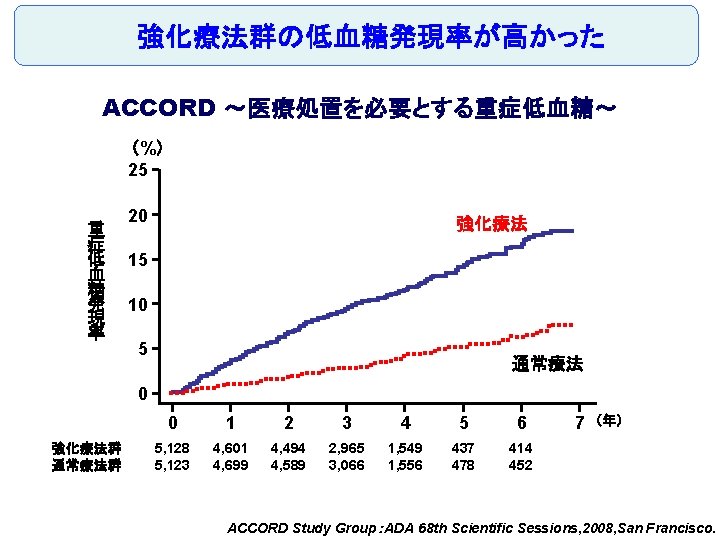
強化療法群の低血糖発現率が高かった ACCORD ~医療処置を必要とする重症低血糖~ (%) 25 重 症 低 血 糖 発 現 率 20 強化療法 15 10 5 通常療法 0 強化療法群 通常療法群 0 1 2 3 4 5 6 5, 128 5, 123 4, 601 4, 699 4, 494 4, 589 2, 965 3, 066 1, 549 1, 556 437 478 414 452 7 (年) ACCORD Study Group:ADA 68 th Scientific Sessions, 2008, San Francisco.
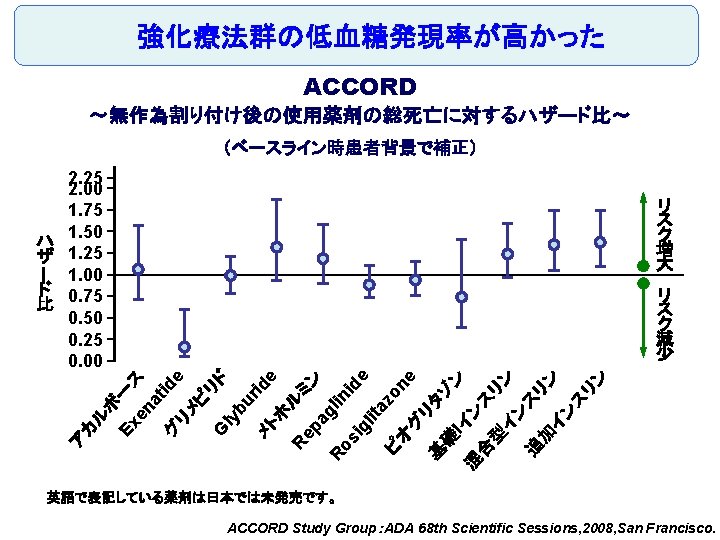
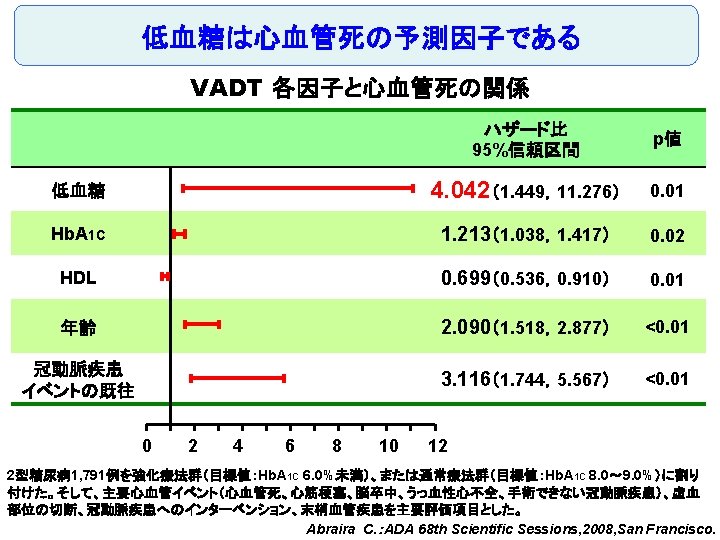

Original Article Long-Term Effect of Diabetes and Its Treatment on Cognitive Function The Diabetes Control and Complications Trial/Epidemiology of Diabetes Interventions and Complications (DCCT/EDIC) Study Research Group The members of the writing committee — Alan M. Jacobson, M. D. , and Gail Musen, Ph. D. , Joslin Diabetes Center and Harvard Medical School, Boston; Christopher M. Ryan, Ph. D. , and Nancy Silvers, R. N. , University of Pittsburgh School of Medicine, Pittsburgh; Patricia Cleary, M. S. , and Barbara Waberski, M. S. , George Washington University, Rockville, MD; Amanda Burwood, B. S. , and Katie Weinger, Ed. D. , Joslin Diabetes Center, Boston; Meg Bayless, R. N. , University of Iowa College of Medicine, Iowa City; William Dahms, M. D. (deceased), Case Western Reserve University, Cleveland; and Judith Harth, R. N. , University of Western Ontario Schulich School of Medicine, London, ON, Canada — and the DCCT/EDIC Study Research Group assume responsibility for the overall content and integrity of the article. N Engl J Med Volume 356(18): 1842 -1852 May 3, 2007 No evidence of substantial long-term declines in cognitive function
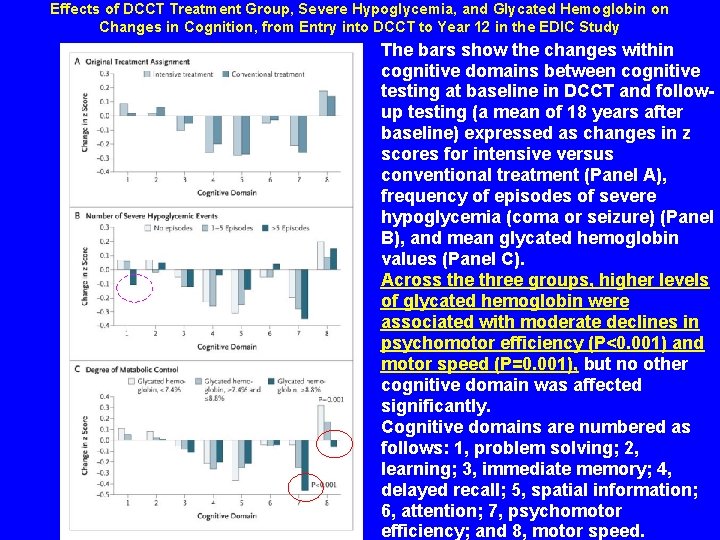
Effects of DCCT Treatment Group, Severe Hypoglycemia, and Glycated Hemoglobin on Changes in Cognition, from Entry into DCCT to Year 12 in the EDIC Study The bars show the changes within cognitive domains between cognitive testing at baseline in DCCT and followup testing (a mean of 18 years after baseline) expressed as changes in z scores for intensive versus conventional treatment (Panel A), frequency of episodes of severe hypoglycemia (coma or seizure) (Panel B), and mean glycated hemoglobin values (Panel C). Across the three groups, higher levels of glycated hemoglobin were associated with moderate declines in psychomotor efficiency (P<0. 001) and motor speed (P=0. 001), but no other cognitive domain was affected significantly. Cognitive domains are numbered as follows: 1, problem solving; 2, learning; 3, immediate memory; 4, delayed recall; 5, spatial information; 6, attention; 7, psychomotor efficiency; and 8, motor speed.

Division of Research, Section of Etiology and Prevention, Kaiser Permanente, Oakland, California (Drs Whitmer, Karter, Quesenberry, and Selby); Departments of Psychiatry, Neurology and Epidemiology, University of California, San Francisco (Dr Yaffe). JAMA. 2009; 301(15): 1565 -1572

Background and Aim Although acute hypoglycemia may be associated with cognitive impairment in children with type 1 diabetes, no studies to date have evaluated whether hypoglycemia is a risk factor for dementia in older patients with type 2 diabetes. To determine if hypoglycemic episodes severe enough to require hospitalization are associated with an increased risk of dementia in a population of older patients with type 2 diabetes followed up for 27 years.

Methods Design, Setting, and Patients: A longitudinal cohort study from 1980 -2007 of 16, 667 patients with a mean age of 65 years and type 2 diabetes who are members of an integrated health care delivery system in northern California. Main Outcome Measure: Hypoglycemic events from 19802002 were collected and reviewed using hospital discharge and emergency department diagnoses. Cohort members with no prior diagnoses of dementia, mild cognitive impairment, or general memory complaints as of January 1, 2003, were followed up for a dementia diagnosis through January 15, 2007. Dementia risk was examined using Cox proportional hazard regression models, adjusted for age, sex, race/ ethnicity, education, body mass index, duration of diabetes, 7 -year mean glycated hemoglobin, diabetes treatment, duration of insulin use, hyperlipidemia, hypertension, cardiovascular disease, stroke, transient cerebral ischemia, and end-stage renal disease.

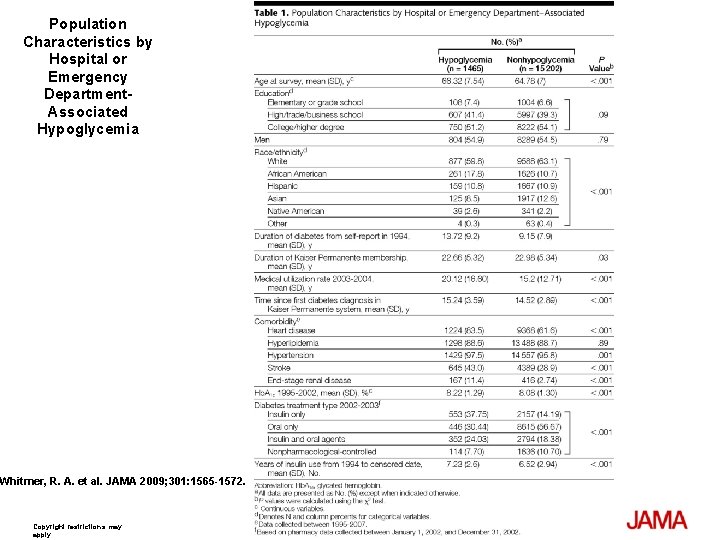
Population Characteristics by Hospital or Emergency Department. Associated Hypoglycemia Whitmer, R. A. et al. JAMA 2009; 301: 1565 -1572. Copyright restrictions may apply.
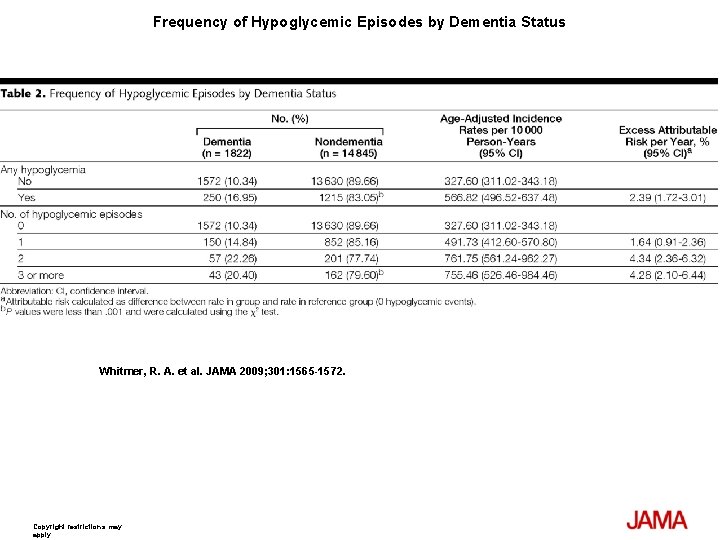
Frequency of Hypoglycemic Episodes by Dementia Status Whitmer, R. A. et al. JAMA 2009; 301: 1565 -1572. Copyright restrictions may apply.
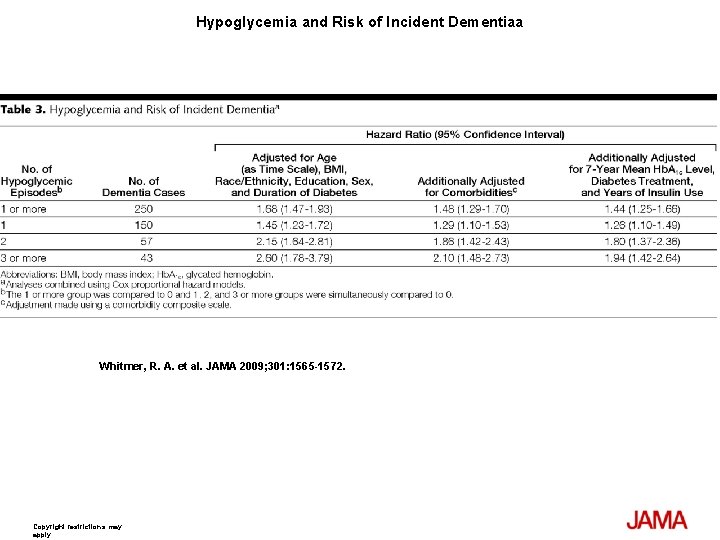
Hypoglycemia and Risk of Incident Dementiaa Whitmer, R. A. et al. JAMA 2009; 301: 1565 -1572. Copyright restrictions may apply.
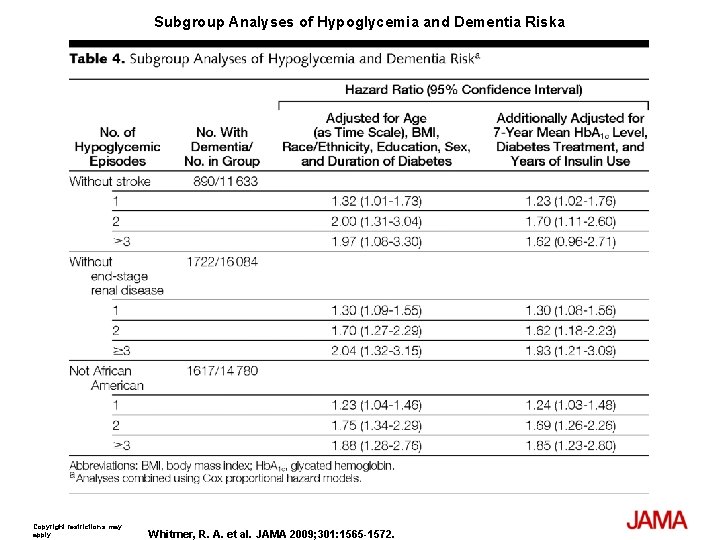
Subgroup Analyses of Hypoglycemia and Dementia Riska Copyright restrictions may apply. Whitmer, R. A. et al. JAMA 2009; 301: 1565 -1572.
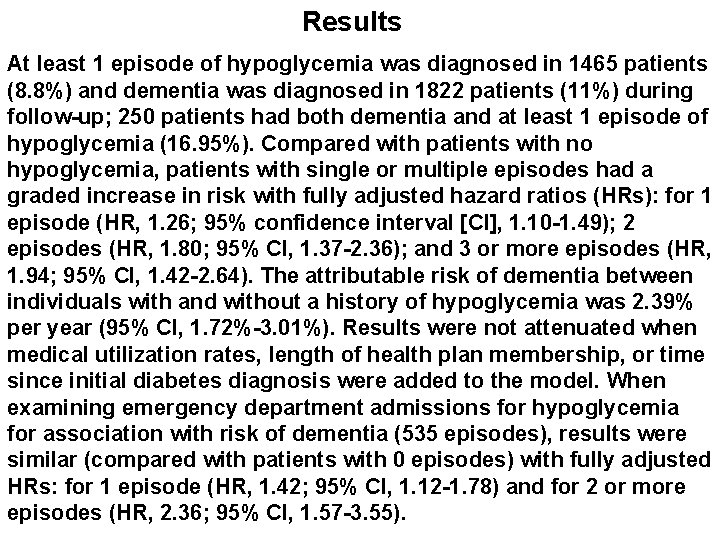
Results At least 1 episode of hypoglycemia was diagnosed in 1465 patients (8. 8%) and dementia was diagnosed in 1822 patients (11%) during follow-up; 250 patients had both dementia and at least 1 episode of hypoglycemia (16. 95%). Compared with patients with no hypoglycemia, patients with single or multiple episodes had a graded increase in risk with fully adjusted hazard ratios (HRs): for 1 episode (HR, 1. 26; 95% confidence interval [CI], 1. 10 -1. 49); 2 episodes (HR, 1. 80; 95% CI, 1. 37 -2. 36); and 3 or more episodes (HR, 1. 94; 95% CI, 1. 42 -2. 64). The attributable risk of dementia between individuals with and without a history of hypoglycemia was 2. 39% per year (95% CI, 1. 72%-3. 01%). Results were not attenuated when medical utilization rates, length of health plan membership, or time since initial diabetes diagnosis were added to the model. When examining emergency department admissions for hypoglycemia for association with risk of dementia (535 episodes), results were similar (compared with patients with 0 episodes) with fully adjusted HRs: for 1 episode (HR, 1. 42; 95% CI, 1. 12 -1. 78) and for 2 or more episodes (HR, 2. 36; 95% CI, 1. 57 -3. 55).

Limitation A possible weakness is that our dementia diagnoses are based on clinical diagnoses obtained from electronic medical records, rather than the results of standardized neurological assessments administered periodically to all cohort members. Another potential concern is that due to the observational nature of our cohort study, we cannot be certain of the temporality of our findings, and cognitive problems due to undiagnosed dementia may have contributed to the occurrence of hypoglycemia. However, individuals with diagnoses of dementia, mild cognitive impairment, or general memory impairment before 2003 were excluded. In addition, we designed the study to increase the temporal separation of earlier hypoglycemic episodes from later occurrences of dementia and also conducted analyses with further lags between exposure to hypoglycemia and the beginning of observation for incident dementia. These lagged-model findings demonstrated similar associations. Even when considering only hypoglycemic episodes during the first 5 years of the study, when the patients were between the ages of 52 and 57 years (when dementia is highly unlikely), there was still an association with an elevated risk of dementia more than 2 decades later. Finally, our study involves the association between severe hypoglycemic episodes and risk of dementia; implications from our study do not address the role of less severe but more frequent episodes of hypoglycemia on dementia risk. The clinical significance of minor hypoglycemic episodes on dementia risk is unknown.

Conclusions Among older patients with type 2 diabetes, a history of severe hypoglycemic episodes was associated with a greater risk of dementia. Whether minor hypoglycemic episodes increase risk of dementia is unknown.


Department of Internal Medicine, Section of Cardiovascular Medicine (Drs Young and Wackers and Ms Davey) and Section of Endocrinology (Dr Inzucchi), Yale University School of Medicine, New Haven, Connecticut; College of Nursing at the College of Dentistry, New York University, New York (Dr Chyun); Department of Endocrinology, University of Virginia, Charlottesville (Dr Barrett); Medecine Nucleaire, University of Montreal, Quebec, Canada (Dr Taillefer); Department of Cardiology, Hartford Hospital, Hartford, Connecticut (Dr Heller); Department of Cardiology, University of Alabama, Birmingham (Dr Iskandrian); Department of Endocrinology, University of Rochester, New York (Dr JAMA. 2009; 301(15): 1547 -1555

Background and Aim Coronary artery disease (CAD) is the major cause of mortality and morbidity in patients with type 2 diabetes. But the utility of screening patients with type 2 diabetes for asymptomatic CAD is controversial. To assess whether routine screening for CAD identifies patients with type 2 diabetes as being at high cardiac risk and whether it affects their cardiac outcomes.

Methods The Detection of Ischemia in Asymptomatic Diabetics (DIAD) study is a randomized controlled trial in which 1123 participants with type 2 diabetes and no symptoms of CAD were randomly assigned to be screened with adenosine-stress radionuclide myocardial perfusion imaging (MPI) or not to be screened. Participants were recruited from diabetes clinics and practices and prospectively followed up from August 2000 to September 2007. Main Outcome Measure is Cardiac death or nonfatal myocardial infarction (MI).
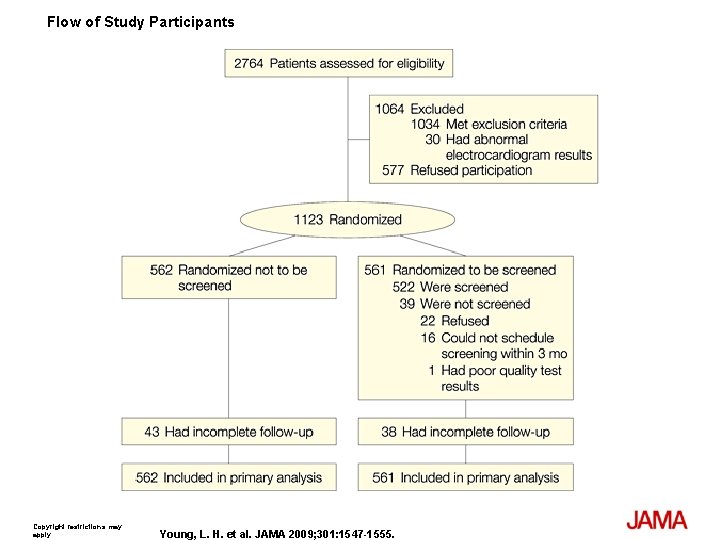
Flow of Study Participants Copyright restrictions may apply. Young, L. H. et al. JAMA 2009; 301: 1547 -1555.
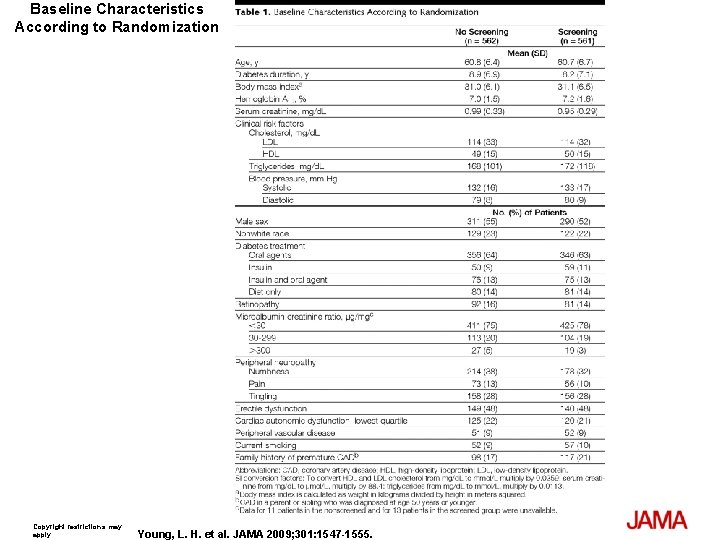
Baseline Characteristics According to Randomization Copyright restrictions may apply. Young, L. H. et al. JAMA 2009; 301: 1547 -1555.
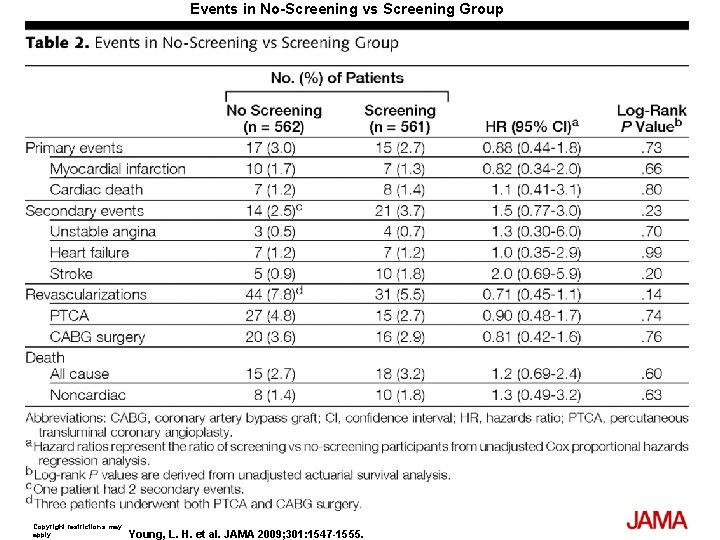
Events in No-Screening vs Screening Group Copyright restrictions may apply. Young, L. H. et al. JAMA 2009; 301: 1547 -1555.
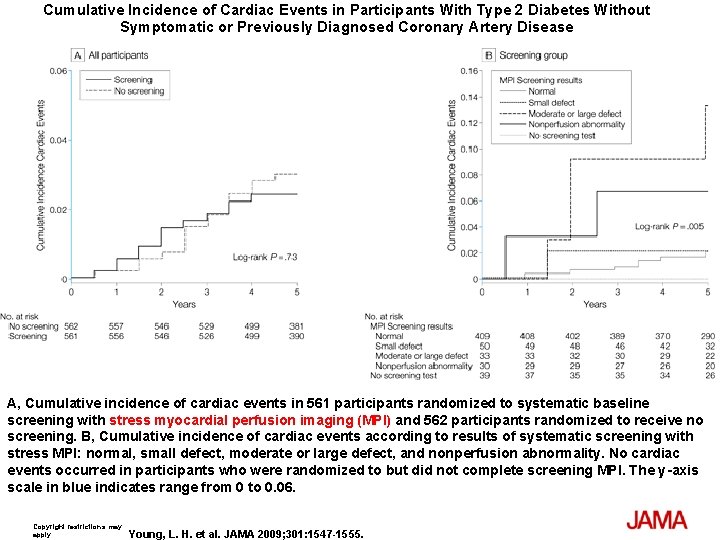
Cumulative Incidence of Cardiac Events in Participants With Type 2 Diabetes Without Symptomatic or Previously Diagnosed Coronary Artery Disease A, Cumulative incidence of cardiac events in 561 participants randomized to systematic baseline screening with stress myocardial perfusion imaging (MPI) and 562 participants randomized to receive no screening. B, Cumulative incidence of cardiac events according to results of systematic screening with stress MPI: normal, small defect, moderate or large defect, and nonperfusion abnormality. No cardiac events occurred in participants who were randomized to but did not complete screening MPI. The y-axis scale in blue indicates range from 0 to 0. 06. Copyright restrictions may apply. Young, L. H. et al. JAMA 2009; 301: 1547 -1555.
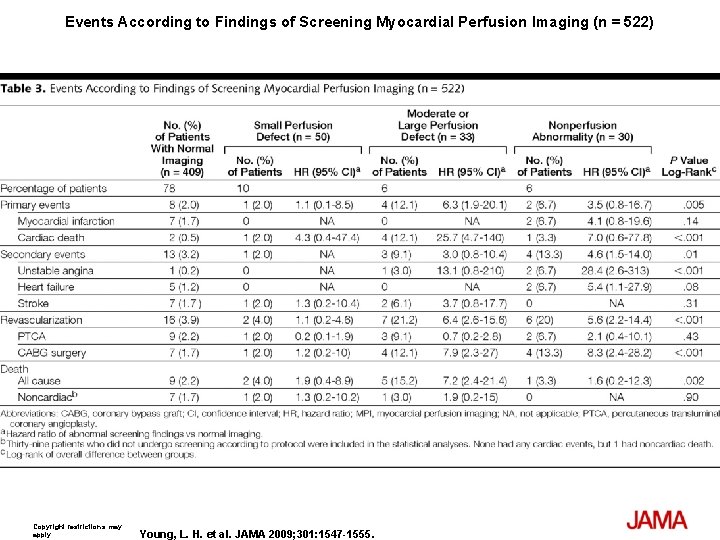
Events According to Findings of Screening Myocardial Perfusion Imaging (n = 522) Copyright restrictions may apply. Young, L. H. et al. JAMA 2009; 301: 1547 -1555.
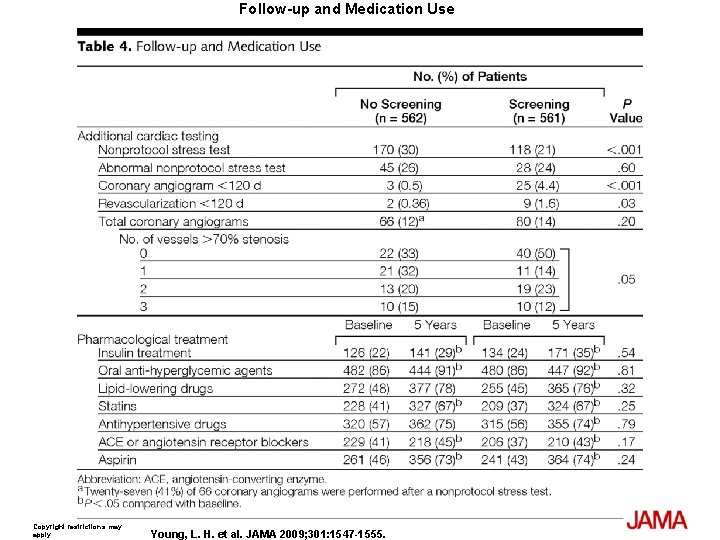
Follow-up and Medication Use Copyright restrictions may apply. Young, L. H. et al. JAMA 2009; 301: 1547 -1555.
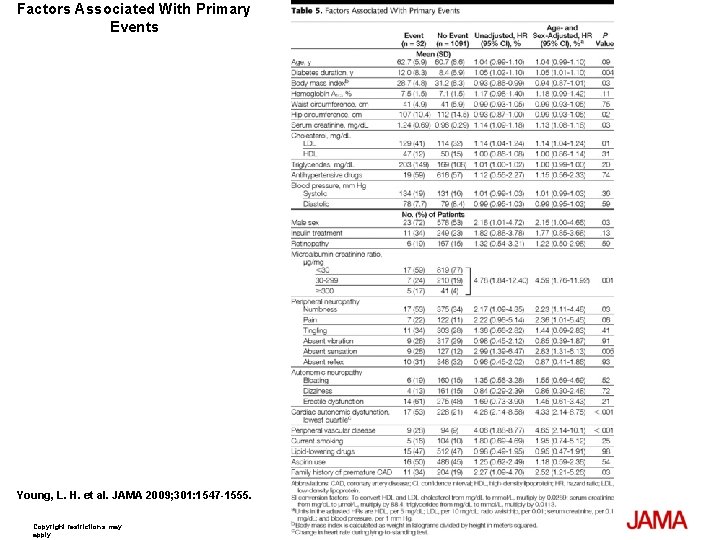
Factors Associated With Primary Events Young, L. H. et al. JAMA 2009; 301: 1547 -1555. Copyright restrictions may apply.
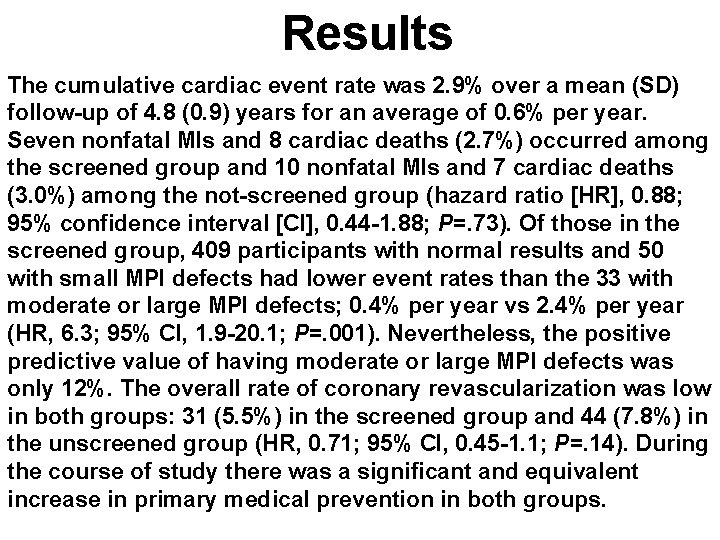
Results The cumulative cardiac event rate was 2. 9% over a mean (SD) follow-up of 4. 8 (0. 9) years for an average of 0. 6% per year. Seven nonfatal MIs and 8 cardiac deaths (2. 7%) occurred among the screened group and 10 nonfatal MIs and 7 cardiac deaths (3. 0%) among the not-screened group (hazard ratio [HR], 0. 88; 95% confidence interval [CI], 0. 44 -1. 88; P=. 73). Of those in the screened group, 409 participants with normal results and 50 with small MPI defects had lower event rates than the 33 with moderate or large MPI defects; 0. 4% per year vs 2. 4% per year (HR, 6. 3; 95% CI, 1. 9 -20. 1; P=. 001). Nevertheless, the positive predictive value of having moderate or large MPI defects was only 12%. The overall rate of coronary revascularization was low in both groups: 31 (5. 5%) in the screened group and 44 (7. 8%) in the unscreened group (HR, 0. 71; 95% CI, 0. 45 -1. 1; P=. 14). During the course of study there was a significant and equivalent increase in primary medical prevention in both groups.

Limitation The cardiac event rates were significantly lower than originally anticipated at the time of the design of the study and therefore the DIAD study does not have the power to exclude a small difference between the screened and unscreened participants. Based on the observed cardiac event rate, we would estimate that the study only had 14% power to detect a 20% difference between the 2 groups. A 3 - to 4 -fold larger study would be required to exclude such a difference, and it is not clear that a reduction in cardiac events from 0. 6% to 0. 5% per year even if proved would justify cardiac screening. Another potential limitation to consider is that nonprotocol stress tests were done during follow-up when clinically indicated in both groups. In addition, screening led to only a modest reduction in subsequent diagnostic testing. Testing was typically performed to evaluate potential cardiac symptoms but may have also been undertaken for risk stratification in some participants. In the no-screening group, such testing represents a crossover to a physician-directed screening strategy and theoretically might have counter-balanced a benefit of protocolmandated systematic screening. However, because the DIAD study did not prohibit physician directed cardiac evaluation, the results are more applicable to current day medicine in which patients are often evaluated for symptoms or preoperative risk stratification or when considered particularly high risk by their physicians.

Conclusion In this contemporary study population of patients with diabetes, the cardiac event rates were low and were not significantly reduced by MPI screening for myocardial ischemia over 4. 8 years. Trial Registration clinicaltrials. gov Identifier: NCT 00769275

- Slides: 43