JOURNAL CLUB DR RONLY HAREVEN COHN PULMONOLOGY FELLOW

JOURNAL CLUB DR. RONLY HAR-EVEN COHN PULMONOLOGY FELLOW (PGY 6), CROSS CANADA ROUNDS NOV 2017 DIVISION OF PEDIATRIC PULMONOLOGY STOLLERY CHILDREN’S HOSPITAL, EDMONTON, ALBERTA, CANADA


OBJECTIVES 1. Learn about CLM, CPAM, PS- presentation, complications and management. 2. Review the study-” The natural history of prenatally diagnosed congenital cystic lung lesions: long term follow-up of 119 cases”. 3. Appraisal of the above study • JAMA critical review. • Review of the editorial. 4. Future directions. 5. Discussion.

THE IMPORTANCE OF THIS STUDY • Continue debate regarding postnatal management of asymptomatic CPAM/ PS. • One of the largest case series of CPAM and PS following prenatal diagnosis. • The conservative treatment has the longest median length of follow up that has been published so far.

PICO EVALUATION • Patients-Children born between 1996 -2009 diagnosed with CPAM/PS on prenatal US, confirmed by postnatal CT imaging, referred to GOSH and followed up ≥ 5 y. • Intervention- Children were conservatively treated. • Comparator- Surgical treatment electively/urgently. • Outcome- Malignant transformation, respiratory infections and pneumothorax.

CLM: CONGENITAL LUNG MALFORMATION • A heterogenous group of rare disorders that include: Ø CPAM-Congenital pulmonary airways malformations Ø PS-pulmonary sequestrations Ø Bronchogenic cyst, Ø Bronchial atresia, Ø Congenital lobar emphysema, Ø Hybrid form of these lesions.

PRENATALLY DETECTION OF CLM INCREASED WITH TECHNOLOGY IMPROVEMENT Prevalence of prenatal detection of CLM- 4. 15 per 10, 000 births. Smaller asymptomatic lesions can be identified. The first CLM that was diagnosed antenatally was CPAM in 1975 in Australia.

CPAM: CONGENITAL PULMONARY AIRWAYS MALFORMATION • Nonfunctioning cystic lesion. • Prevalence at birth 1: 8, 300 -1: 35, 000 • >80% diagnosed antenatally. • Most common congenital lung lesion 25 -30% of all. • M>F • Mostly Sporadically

CPAM: CONGENITAL PULMONARY AIRWAYS MALFORMATION Lesions are hamartomatous: – contain cystic and adenomatous elements. – connected to the tracheobronchial tree – have blood supply from the pulmonary circulation. – usually unilateral and limited to one lobe

QUESTION CPAM are classified according to their: a. Location of the lesion in the airway b. Radiologic features c. Pathological characteristics d. None of the above e. a+c
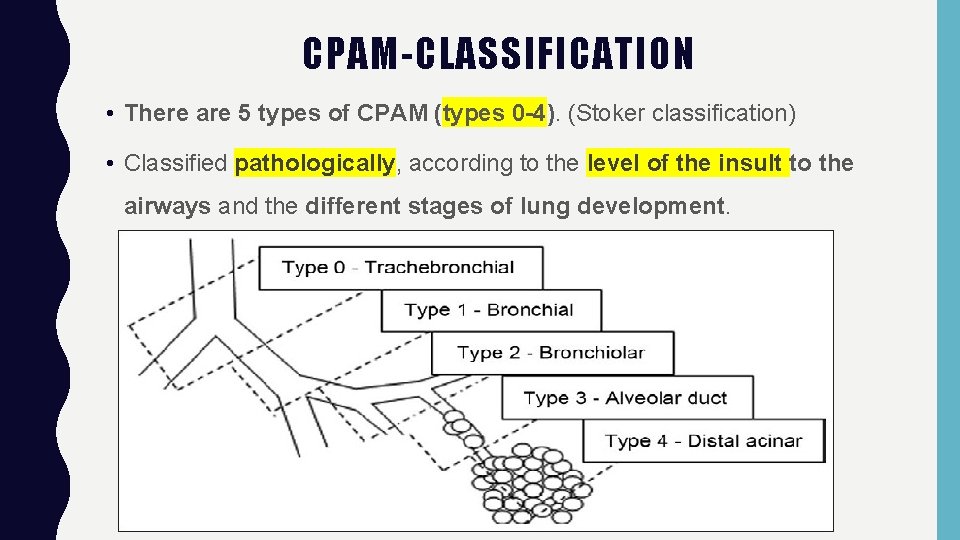
CPAM-CLASSIFICATION • There are 5 types of CPAM (types 0 -4). (Stoker classification) • Classified pathologically, according to the level of the insult to the airways and the different stages of lung development.

CPAM- 5 TYPES PATHOLOGICALLY Types Percentage Location in the airways Lesion description Prognosis Comments Type 0 1 -3% From the Solid, lungs are trachea/bronchi and small and firm involve the whole lung Fatalincompatibl e with life. Type 1 60 -70% From distal bronchi or proximal bronchioles One Large cyst 2 -10 cm, presentation may be late Most common type –good prognosis Associated with malignancy BAC (rare) Type 2 15 -20% Bronchiolar origin Multiple small cysts 0. 5 -2 cm Neonatal period Poor prognosis Associated with other anomalies Type 3 5 -10% Alveolar origin Small cystic area with solid tissue. Solid appearance Respiratory distress in neonatal period/deat h Severe Type 4 10 -15% Acinar origin Thin walled Large Good fluid filled/ air prognosis filled cysts up to Associated with pneumothorax
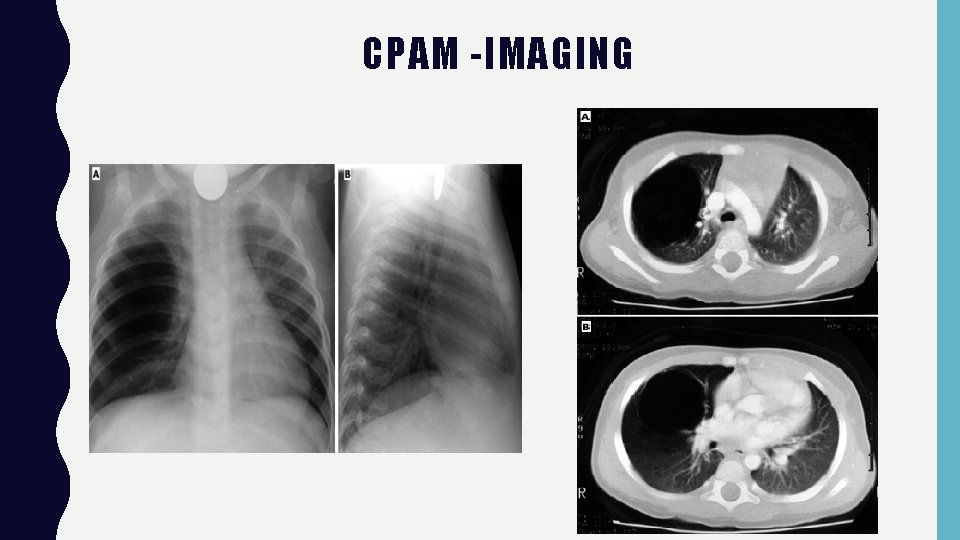
CPAM -IMAGING
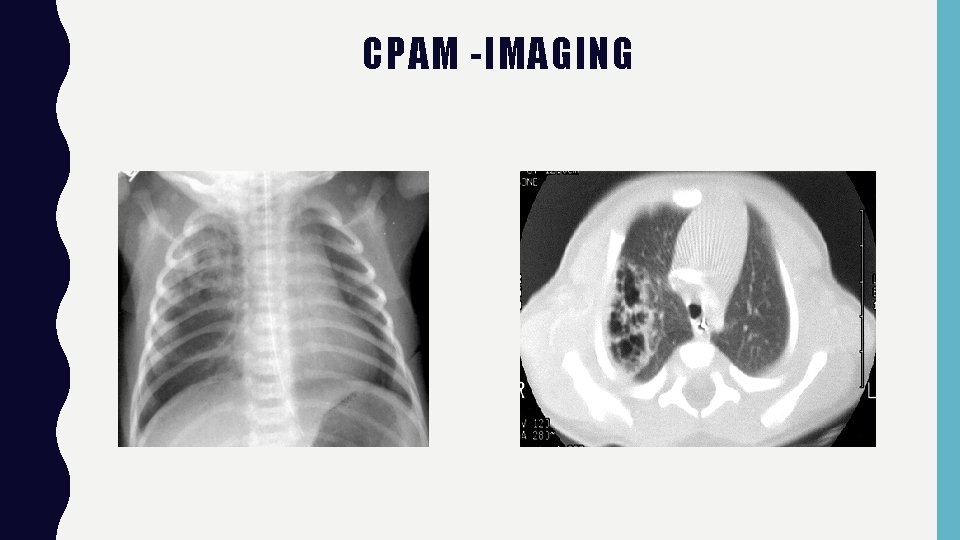
CPAM -IMAGING
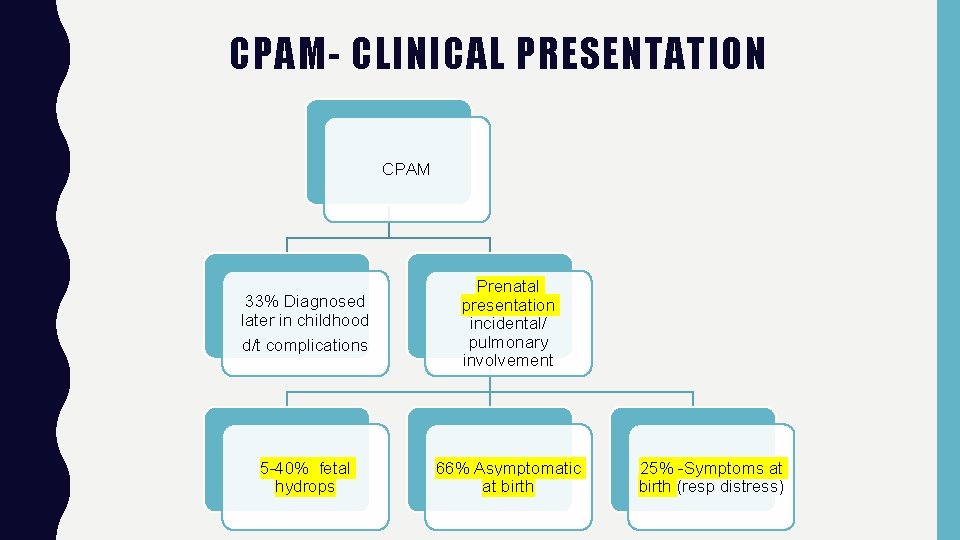
CPAM- CLINICAL PRESENTATION CPAM 33% Diagnosed later in childhood d/t complications Prenatal presentation incidental/ pulmonary involvement 5 -40% fetal hydrops 66% Asymptomatic at birth 25% -Symptoms at birth (resp distress)

CPAM DIAGNOSIS-RECOMMENDATIONS • Family history of cancers/ cystic lesions (type 4 -genetic predisposition). • Prenatal US+/- prenatal MRI. • CXR (all patients) – single lesions of large air filled cysts (type 1, 4), – small numerous air filled cysts (type 2), – large solid mass (type 3) Plain CXR often fail to detect CPAM in asymptomatic patients.

CPAM DIAGNOSIS-RECOMMENDATIONS • Chest CT /chest MRI-for symptomatic patients-preoperative or for asymptomatic patients at 6 m of age or earlier if in high risk for complications. • High risk for complications: large lesions, bilateral/multifocal cysts, family history of PPB, pneumothorax. • Definitive diagnosis- by histological examination. • Reference: Antenatal and postnatal management of congenital cystic adenomatoid malformation”. Kotecha S, et al. Paediatric respiratory reviews 2012; 13: 162 -171.

QUESTION What are the complications of CPAM ? a. infections b. pneumothorax c. malignancy d. all of the above e. none of the above
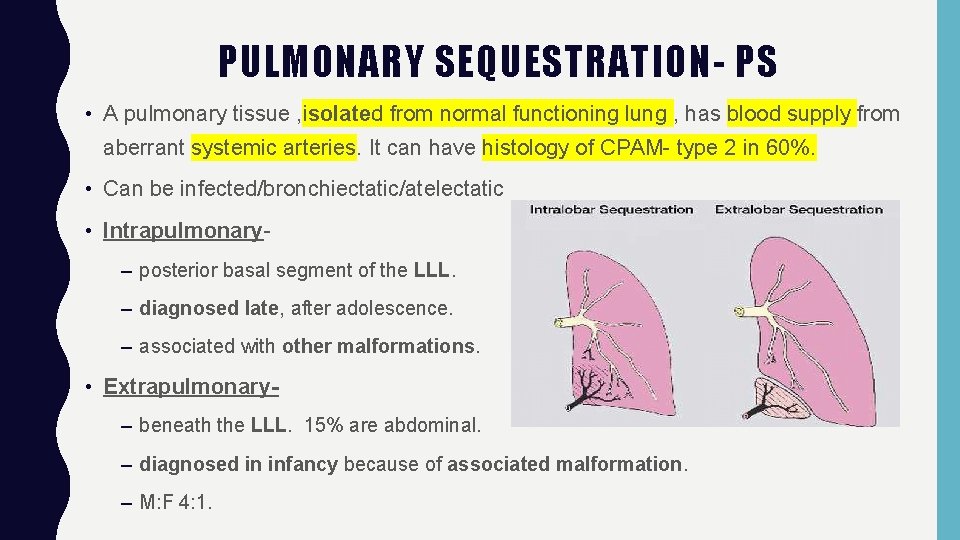
PULMONARY SEQUESTRATION- PS • A pulmonary tissue , isolated from normal functioning lung , has blood supply from aberrant systemic arteries. It can have histology of CPAM- type 2 in 60%. • Can be infected/bronchiectatic/atelectatic • Intrapulmonary– posterior basal segment of the LLL. – diagnosed late, after adolescence. – associated with other malformations. • Extrapulmonary– beneath the LLL. 15% are abdominal. – diagnosed in infancy because of associated malformation. – M: F 4: 1.
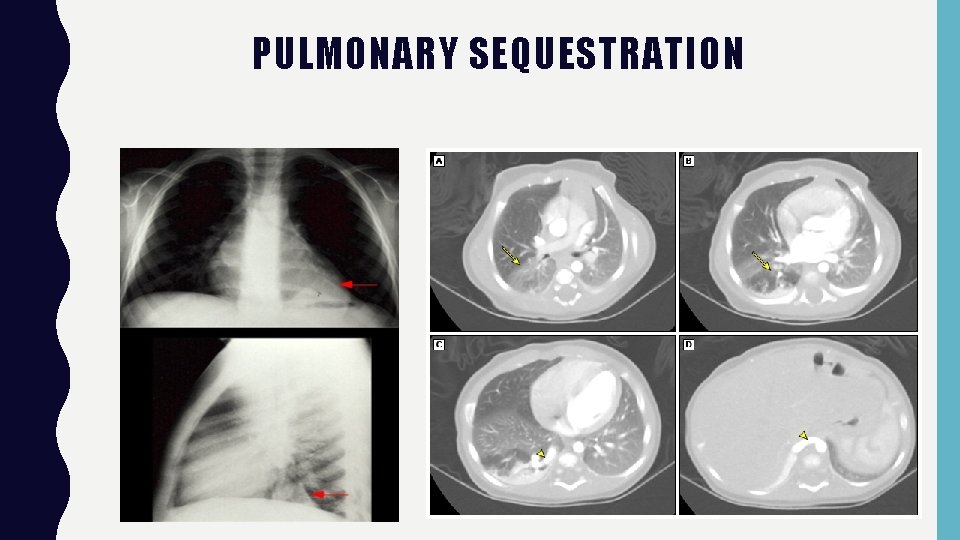
PULMONARY SEQUESTRATION
QUESTION What is the current management suggested for treating CPAM/PS? a. Surgery for all lesions b. Conservative treatment and observation for all lesions c. Surgery for symptomatic lesions and watch and wait for asymptomatic lesions. d. None of the above e. I cannot answer the question, further studies are needed.

MANAGEMENT OF CPAM/PS • Symptomatic lesions in the neonatal period- surgical resection. – The surgery is curative. – Lobectomy preferred over segmentectomy and VATS preferred over open surgery. • Controversy about how to manage asymptomatic lesions. • Uncertainty about – The risk of recurrent infections – The risk of malignancy If an elective surgery is done- it is done before the age of 1 y.

MANAGEMENT OF ASYMPTOMATIC CPAM/PS -CONTROVERSY SURGICAL TREATMENT VERSUS CONSERVATIVE TREATMENT

SURGICAL VERSUS CONSERVATIVE TREATMENT ARGUMENTS FOR CONSERVATIVE TREATMENT • In the past, lesions had been rarely diagnosed as pathogenic • Low risk of complications (low frequency of infections, extremely low risk of malignancy). • Can resolve spontaneously • No exposure to unnecessary surgical and anesthetic risks. ARGUMENTS FOR SURGICAL TREATMENT • Prevention of complications such as malignant transformation, recurrent infections and pneumothorax. • Better compensatory lung growth in early age. • Fewer postoperative complications for elective surgery. • No exposure to repeated CT and long term follow up. • Psychological effect- “to do something”


STUDY QUESTION • Whether a conservative management is a reasonable option to treat asymptomatic patients with CPAM and PS?

METHODS • Descriptive, cohort prospective study from database of fetus with CLM on prenatal US (CPAM/PS) that were confirmed by Chest CT. • Data collected: – Demographic details, – Postnatal CT imaging results, – Date of last clinic attended – Follow up status: continued/lost to follow up/discharged from follow up. – Surgical treatment if done: indications/age at surgery/histologic

INCLUSION/EXCLUSION CRITERIA • Inclusion criteria: 1. CLM on prenatal US and Radiological confirmation of CPAM/PS on postnatal CT imaging. 2. Follow up for ≥ 5 y (5 -18 y, median 9. 9 y). • Exclusion criteria: not reported
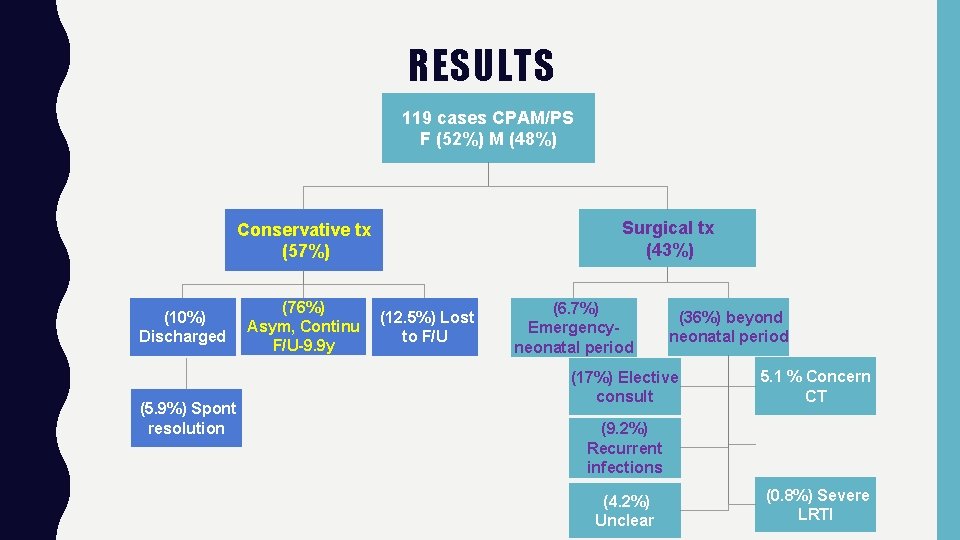
RESULTS 119 cases CPAM/PS F (52%) M (48%) Surgical tx (43%) Conservative tx (57%) (10%) Discharged (5. 9%) Spont resolution (76%) Asym, Continu F/U-9. 9 y (12. 5%) Lost to F/U (6. 7%) Emergencyneonatal period (36%) beyond neonatal period (17%) Elective consult 5. 1 % Concern CT (9. 2%) Recurrent infections (4. 2%) Unclear (0. 8%) Severe LRTI

RESULTS • The only complication - recurrent infections. • Resection due to respiratory infections decreased >2 y of age. • Resection due to respiratory infections disappear >5 y of age. • No features of malignancy. • 4 patients had spontaneous resolution. • In (53%) of the resected lesions, no evidence of inflammation. • Post operatively- no death recorded following elective surgery.
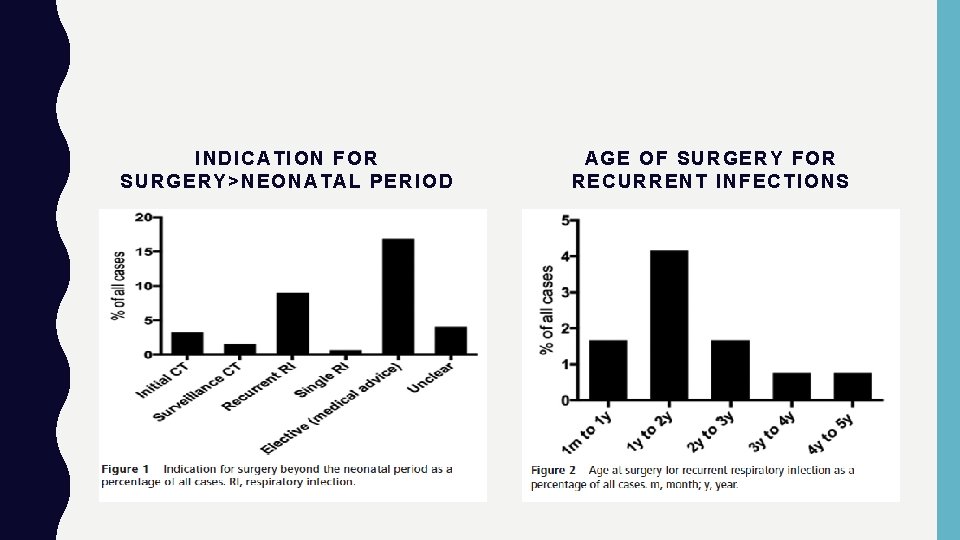
INDICATION FOR SURGERY>NEONATAL PERIOD AGE OF SURGERY FOR RECURRENT INFECTIONS
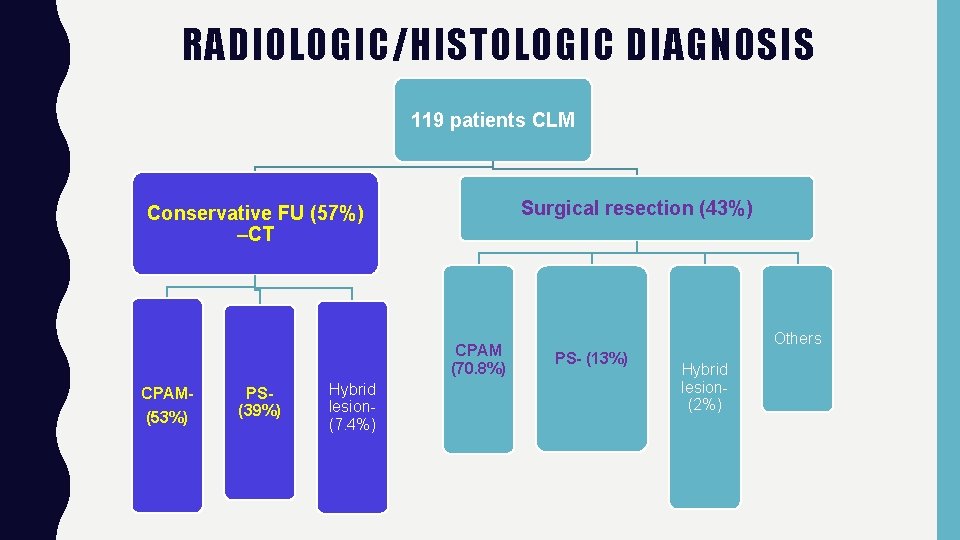
RADIOLOGIC/HISTOLOGIC DIAGNOSIS 119 patients CLM Surgical resection (43%) Conservative FU (57%) –CT CPAM (70. 8%) CPAM(53%) PS(39%) Hybrid lesion(7. 4%) Others PS- (13%) Hybrid lesion(2%)

DISCUSSION Arguments for surgical approach in all asymptomatic patients 1. Prevent malignancy 2. Prevent complications (infection/pneumothorax) 3. Early resection allow compensatory lung regrowth. 4. Elective resection decrease postoperative complications.
COMPLICATIONS –THE RISK OF MALIGNANCYBACKGROUND 2 types of malignancy associated with cystic lung lesions: – PPB-Pleuropulmonary blastoma – BAC-bronchioloalveolar carcinoma

PPB- PLEUROPULMONARY BLASTOMA • Rare lung tumor of childhood <500 cases worldwide. • Incidence 1: 300, 000. • 3 types- type 1 is cystic, type 2 -cystic+solid, type 3 -solid. • Appeared before 6. 8 y but after birth. Only few identified antenatally. • 30% are cystic form, resemble nonmalignant CPAM cysts. • Distinct lesion- Not a malignant transformation of CPAM/PS,

PPB- PLEUROPULMONARY BLASTOMA • Risk factors that support the diagnosis of PPB – Positive family history of associated tumors or cystic lesions. – Genetic diagnosis- mutation in DICER 1 gene in 66%. – Bilateral and multifocal cysts. – Pneumothorax, pleural effusions. – Type 4 CPAM histologically overlap with grade 1 PPB (cystic lesion) PPB-Can develop following prior resection of cystic lesion.
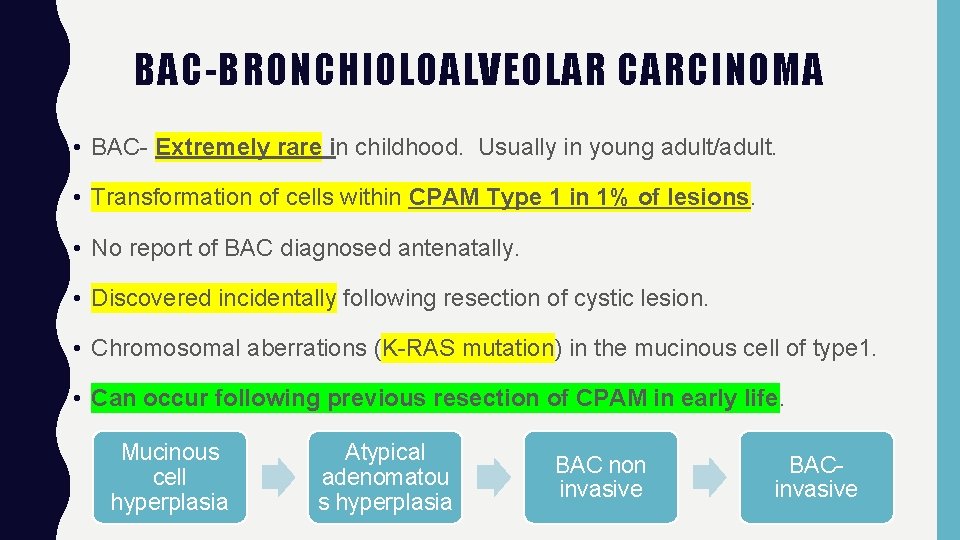
BAC-BRONCHIOLOALVEOLAR CARCINOMA • BAC- Extremely rare in childhood. Usually in young adult/adult. • Transformation of cells within CPAM Type 1 in 1% of lesions. • No report of BAC diagnosed antenatally. • Discovered incidentally following resection of cystic lesion. • Chromosomal aberrations (K-RAS mutation) in the mucinous cell of type 1. • Can occur following previous resection of CPAM in early life. Mucinous cell hyperplasia Atypical adenomatou s hyperplasia BAC non invasive BACinvasive

RISK OF MALIGNANCY- IN THIS ARTICLE • The results were similar to previous studies. • No evidence of malignancy was found in the resected lesions. • No concerns about malignancy in the asymptomatic patients. • Support the assumption that the risk of malignant transformation in early life is rare. • A risk of approximately 1% of malignancy in adolescence and adults cannot be ruled out.

COMPLICATIONS-RECURRENT PULMONARY INFECTIONS -BACKGROUND Recurrent infections is the most common complication. Can be the presenting sign or can occur later in the first few years of life. Frequency –varies: In some case series: 10 -30% of infants developed infection within 1 y versus other that showed only 3% risk of infection in the first 5 y.
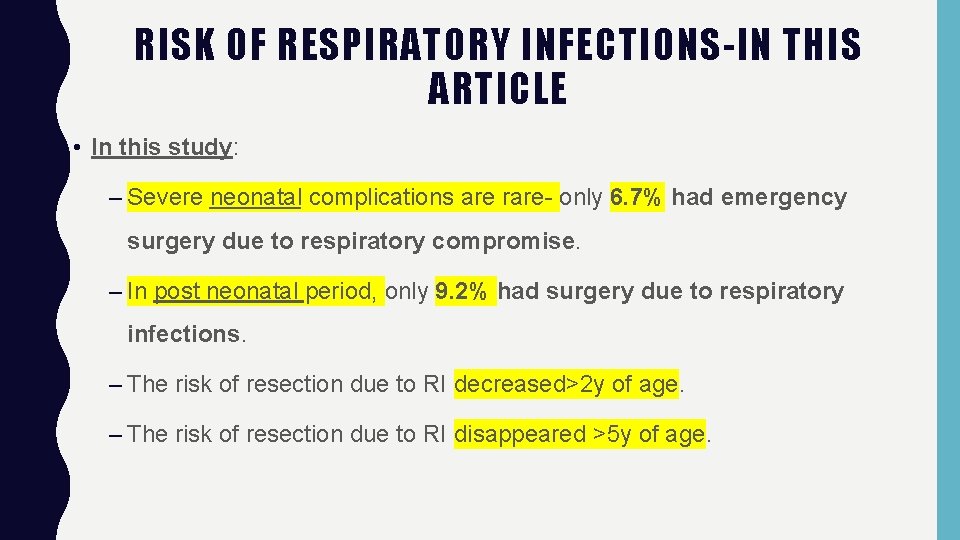
RISK OF RESPIRATORY INFECTIONS-IN THIS ARTICLE • In this study: – Severe neonatal complications are rare- only 6. 7% had emergency surgery due to respiratory compromise. – In post neonatal period, only 9. 2% had surgery due to respiratory infections. – The risk of resection due to RI decreased>2 y of age. – The risk of resection due to RI disappeared >5 y of age.

RISK OF COMPLICATIONS- PNEUMOTHORAX IN THIS ARTICLE • There were no complications of pneumothorax.

POTENTIAL FOR COMPENSATORY LUNG GROWTH FOLLOWING EARLY RESECTION Arguments against : 1. New alveolar growth occur into adolescence and not only in early life. 2. The majority of the lesions are small and reduce in size as the lung grows. 3. 4 lesions disappeared on CT during postnatal period. 4. Other studies -minimal change in lung function after resection in

POSTOPERATIVE COMPLICATIONS • Not recorded in this study. • However, a large meta analysis of 9 case series showed an increased risk of postoperative complication (X 2) in symptomatic patients compared to asymptomatic and support elective surgery.
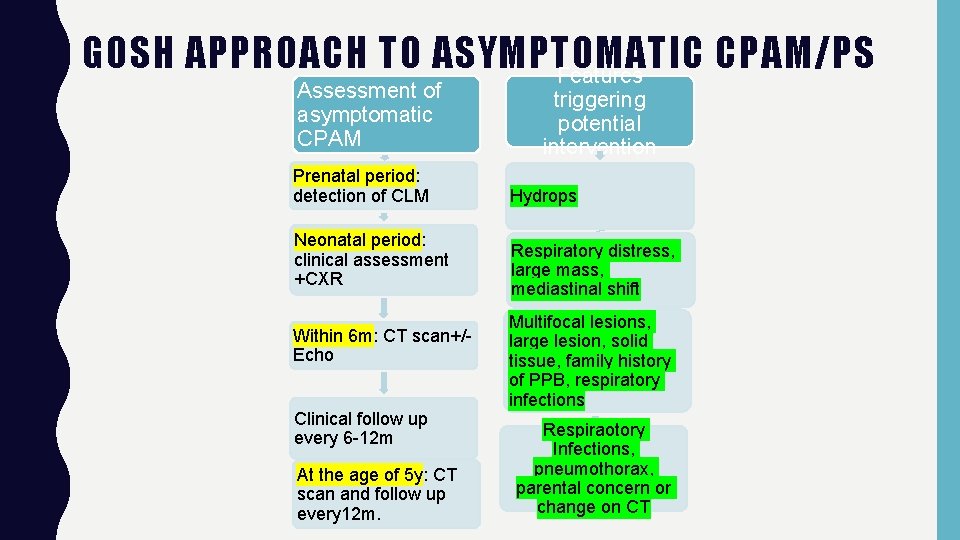
GOSH APPROACH TO ASYMPTOMATIC CPAM/PS Features Assessment of asymptomatic CPAM Prenatal period: detection of CLM Neonatal period: clinical assessment +CXR Within 6 m: CT scan+/Echo Clinical follow up every 6 -12 m At the age of 5 y: CT scan and follow up every 12 m. triggering potential intervention Hydrops Respiratory distress, large mass, mediastinal shift Multifocal lesions, large lesion, solid tissue, family history of PPB, respiratory infections Respiraotory Infections, pneumothorax, parental concern or change on CT

CONCLUSION OF THE STUDY • Conservative approach in asymptomatic CPAM/PS is possible. • Explanation to parents and antenatal counselling is recommended, • The need for surgery due to RI decreases >2 y of age. • Spontaneous resolution can occur in some cases. • Malignancy is rare during pediatric period. • A long term follow up is needed.

APPRAISAL OF THE STUDY- JAMA Are the results valid? • Was there a representative and well defined sample of patients at a similar point in the course of the disease? No. The inclusion criteria was representative for the target population (CLM pt), however along the disease there were selection bias. Many families chose to have an elective resection due to medical advice. • Was follow up sufficiently long and complete? No. The follow up was not long enough regarding the risk for malignancy and wasn’t complete because 12. 5% of the asymptomatic patients were lost for follow up. • Were objective and unbiased outcome criteria used? Yes. Outcomes were defined at the begining of the study, however not enough time was given to assess the risk for malignancy.

APPRAISAL OF THE STUDY • What are the results? – How likely are the outcomes over times? We don’t know. Follow up was not sufficient long enough for the outcome regarding for the risk of malignancy. – How precise are the estimates of likelihood (confidence intervals)? We don’t know. No statistical analysis was done.

APPRAISAL OF THE STUDY • Will the results help me care my patients? – Can the results be applied to my patients? No. No information was reported about the patients’ demographics , co morbidities, and their imaging results. – Will the results lead directly to selecting therapy? No. The sample size was not large enough (only 119 pt , out of which only 68 patients were conservatively followed up) and the validity of the results is questionable. – Are the results useful for reassuring the patients? No. The study cannot rule out the possibility of malignancy which is most concerning for the families.

EDITORIAL ABOUT THE STUDY

SUPPORT THE ARTICLE 1. The results of the study are similar to previous studies. 2. The study provides a median of about 10 y of follow up for 68 pt managed conservatively. 3. Severe neonatal complications are rare-6. 7%. 4. Infection is not the predominant indication for surgery post neonatally. 5. Spontaneous resolution of CPAM is rare-5. 9%.

ARTICLE WEAKNESSES-NO PHENOTYPIC DESCRIPTION • CPAM are highly heterogenous in size and appearance. • Prenatal and postnatal imagining of CPAM provides phenotypic information. • The impact of phenotype on complication rate is unknown. • In this study there was no phenotypic description , only pathological diagnosis that is poorly predicted by imaging. • More studies are needed in that matter.

ARTICLE WEAKNESSES- RISK FOR MALIGNANT TRANSFORMATION • The study neither has sufficient power nor a long enough duration of follow up to rule out the possibility of malignancy. • BAC can develop from K-RAS mutation in mucinous cells of CPAM. • More information is needed about the rate of K-RAS mutation in CPAM and the associated phenotype in CPAM. • Although rare, parents won’t like to take this risk.

ARTICLE WEAKNESSES- PARENTS’ APPROACH • The prenatal diagnosis of a CPAM is stressful for the parents. • Parents can act in a disproportional way- prohibit activities. • Some parents will be uncomfortable with an indeterminate long term prognosis for their child’s malformation. • Some parents will prefer to have surgical excision of the CPAM which cure the condition completely.
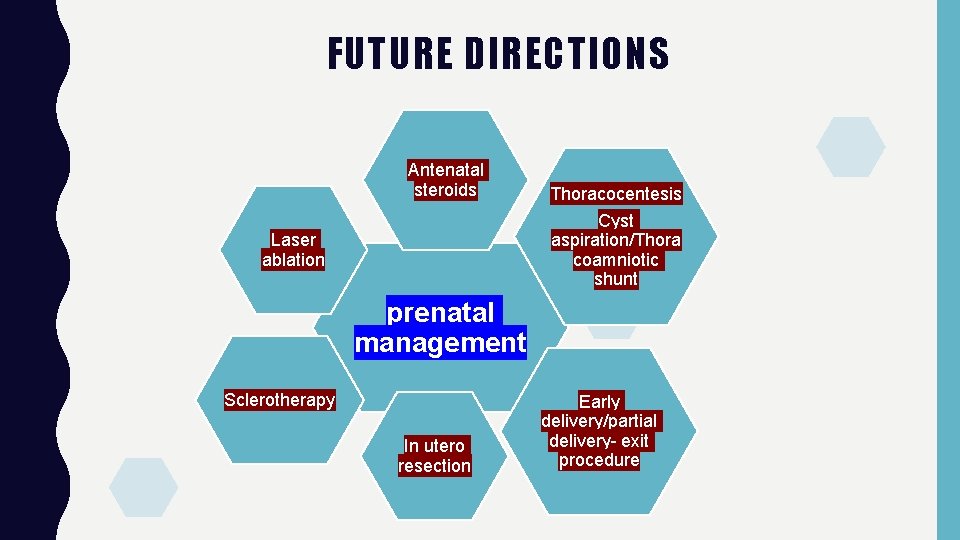
FUTURE DIRECTIONS Antenatal steroids Thoracocentesis Cyst aspiration/Thora coamniotic shunt Laser ablation prenatal management Sclerotherapy In utero resection Early delivery/partial delivery- exit procedure
FUTURE DIRECTIONS Conservative tx Research Long F/U

TAKE HOME MESSAGE • As technology develops our ability to identify CLM antenatally improves. • CPAM is a rare congenital pulmonary airways malformation • CPAM- clinical presentation can be asymptomatic. • There is controversy about how to manage asymptomatic patients. • Conservative approach is possible. • A long term follow up and further studies are needed.

DISCUSSION • What is the current management of CPAM/PS in your hospital?

QUESTIONS?

REFERENCES 1. Cook J, et al. The natural history of prenatally diagnosed congenital cystic lung lesions: long term follow-up of 119 cases. Archives of disease in childhood 2017; 102: 798 -803. 2. Delacourt C, et al. Postnatal management of asymptomatic congenital pulmonary malformation: moving towards evidence-based decisions. Archives of disease in childhood 2017 editorial letter: 102: 789 -790. 3. Kapralik J, et al. Surgical versus conservative management of congenital pulmonary airway malformation in children: A systemic review and meta-analysis. Journal of pediatric surgery 2016; 51: 508 -512. 4. Stanton M, et al. Systemic review and meta-analysis of the postnatal management of congenital cystic lung lesions. Journal of pediatric surgery 2009; 44: 1027 -1033. 5. Kotecha S, et al. Antenatal and postnatal management of congenital cystic adenomatoid malformation. Paediatric respiratory reviews 2012; 13: 162 -171. 6. Oermann CM, Congenital pulmonary airway (cystic adenomatoid) malformation. Uptodate 2017: 1 -22. 7. Stanton M. The argument for a non operative approach to asymptomatic lung lesions. Seminars in Pediatric Surgery 2015; 24 : 183 -186. 8. Singh R. The argument for operative approach to asymptomatic lung lesions. Seminar in Pediatric Surgery 2015; 24: 187 -195. 9. Kendig and Chernick’s. Congenital cystic lesions. Disorders of the Respiratory Tract in Children. Eighth Edition 2012: 337 -343. 10. Hull Jeremy, et al. Congenital lung anomalies. Pediatric Respiratory Medicine, Oxford specialist handbook in pediatrics, second edition 2015; 469 -478

THANK YOU FOR LISTENING
- Slides: 60