Journal Club Association between SARSCo V2 Neutralizing Antibodies

Journal Club Association between SARS-Co. V-2 Neutralizing Antibodies and Commercial Serological Assays M. S. Tang, J. B. Case, C. E. Franks, R. E. Chen, N. W. Anderson, J. P. Henderson, M. S. Diamond, A. M. Gronowski, C. W. Farnsworth December 2020 https: //doi. org/10. 1093/clinchem/hvaa 211 © Copyright 2020 by the American Association for Clinical Chemistry
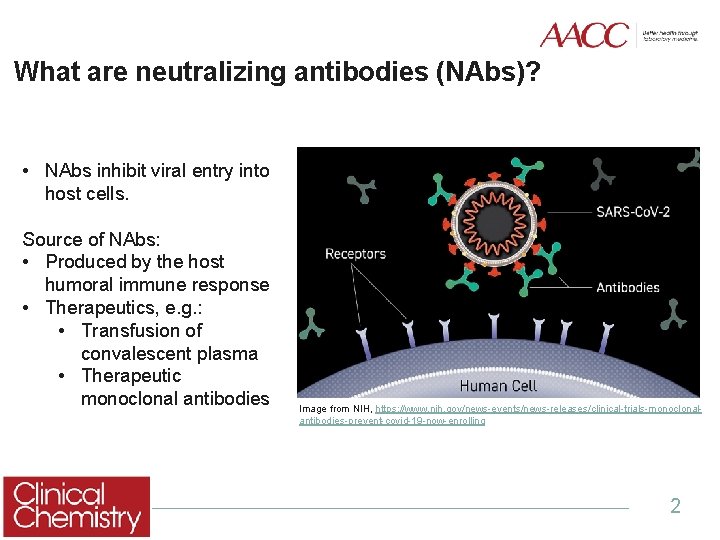
What are neutralizing antibodies (NAbs)? • NAbs inhibit viral entry into host cells. Source of NAbs: • Produced by the host humoral immune response • Therapeutics, e. g. : • Transfusion of convalescent plasma • Therapeutic monoclonal antibodies Image from NIH, https: //www. nih. gov/news-events/news-releases/clinical-trials-monoclonalantibodies-prevent-covid-19 -now-enrolling 2
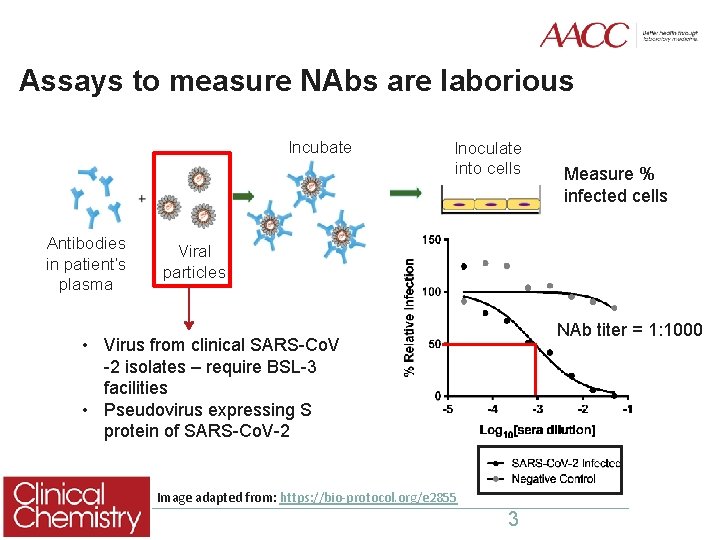
Assays to measure NAbs are laborious Incubate Antibodies in patient’s plasma Inoculate into cells Measure % infected cells Measure Viral particles NAb titer = 1: 1000 • Virus from clinical SARS-Co. V -2 isolates – require BSL-3 facilities • Pseudovirus expressing S protein of SARS-Co. V-2 Image adapted from: https: //bio-protocol. org/e 2855 3

Hypothesis Advantages of commercially available serological assays: • High-throughput • Relatively inexpensive • Use readily available instrumentation Study question: • Can commercial serology assays predict the presence and concentration of neutralizing antibodies? 4

Study design • 71 plasma specimens: • 66 specimens from patients with PCR-confirmed SARSCo. V-2 • 5 presumed negative specimens from 2015 • All specimens were measured on: 1. Abbott Ig. G serological assay 2. Roche total Ig serological assay 3. EUROIMMUN Ig. G serological assay 4. A research use neutralizing assay 1 Q: What statistics should be used to evaluate the correlation between serological and neutralizing assays? 1 Case & Rothlauf, Cell Host & Microbe 2020, 28: 475 -485. 5
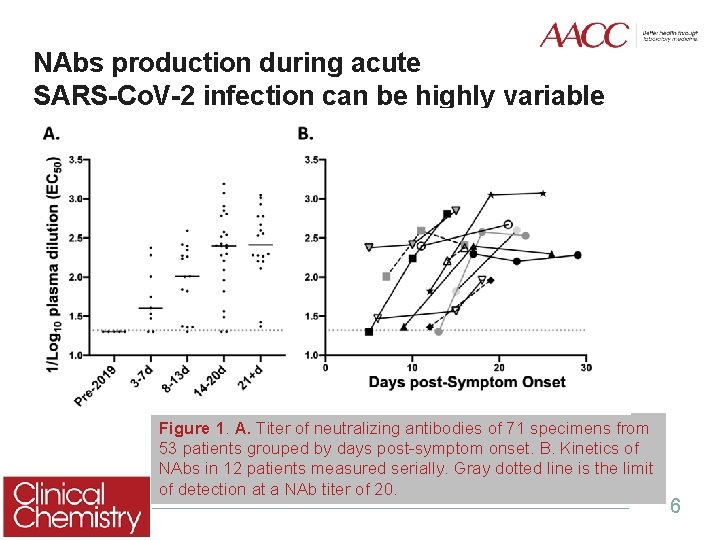
NAbs production during acute SARS-Co. V-2 infection can be highly variable Figure 1. A. Titer of neutralizing antibodies of 71 specimens from 53 patients grouped by days post-symptom onset. B. Kinetics of NAbs in 12 patients measured serially. Gray dotted line is the limit of detection at a NAb titer of 20. 6
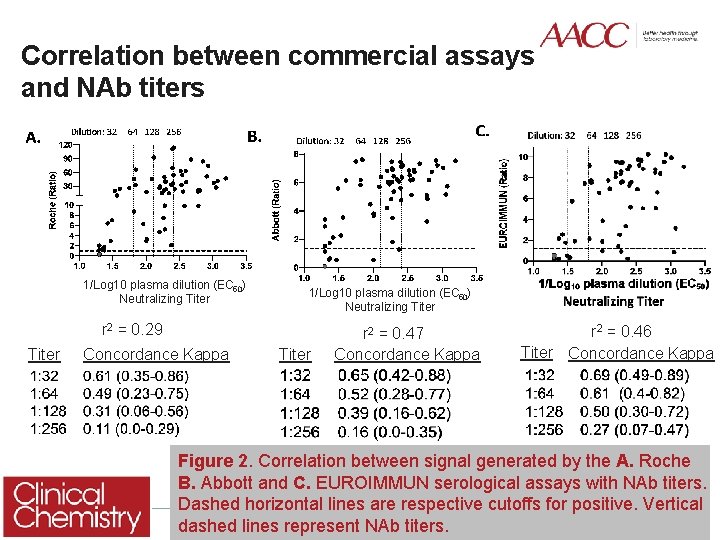
Correlation between commercial assays and NAb titers 1/Log 10 plasma dilution (EC 50) Neutralizing Titer r 2 = 0. 29 Titer Concordance Kappa Titer r 2 = 0. 47 Concordance Kappa r 2 = 0. 46 Titer Concordance Kappa Figure 2. Correlation between signal generated by the A. Roche B. Abbott and C. EUROIMMUN serological assays with NAb titers. Dashed horizontal lines are respective cutoffs for positive. Vertical 7 dashed lines represent NAb titers.
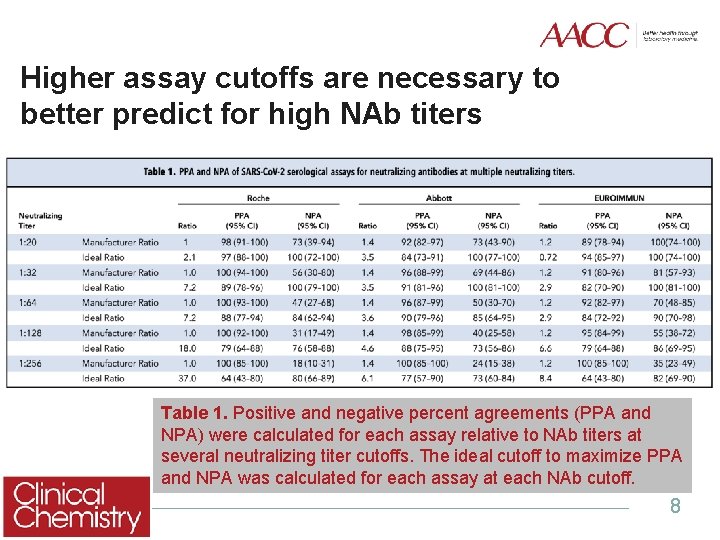
Higher assay cutoffs are necessary to better predict for high NAb titers Table 1. Positive and negative percent agreements (PPA and NPA) were calculated for each assay relative to NAb titers at several neutralizing titer cutoffs. The ideal cutoff to maximize PPA and NPA was calculated for each assay at each NAb cutoff. 8
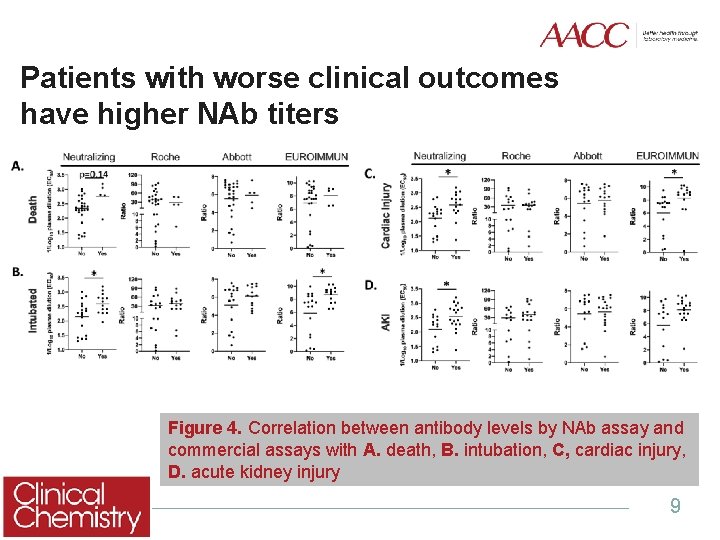
Patients with worse clinical outcomes have higher NAb titers Figure 4. Correlation between antibody levels by NAb assay and commercial assays with A. death, B. intubation, C, cardiac injury, D. acute kidney injury 9

Questions for discussion: • Should results from serological assays be used to predict for presence of NAbs? • Why did patients with worse clinical outcomes have higher NAb titers? 10

Discussion Should results from serological assays be used to predict for presence of NAbs? • Commercial serological and neutralizing assays have poor negative percent agreement, regardless of whether the serological assay measures antibodies against viral spiked or nucleocapsid proteins • Serological assay cutoffs should be increased if goal is to predict for high NAb titers • Commercial serological assays are poor proxies for neutralization 11

Discussion Why did patients with worse clinical outcomes have higher NAb titers? • Mechanisms unclear, but possible explanations are: • Hyperactive immune response in severe disease • Higher antigen burden in severe disease • Similar observations made in other studies 1, 2 • Important to remember that host response to SARS-Co. V -2 also involves cellular immunity 3 1 Klein et al, J Clin Invest. 2020; 130(11): 6141 -6150. et al, Nature Medicine. 2020; 26: 1200– 1204. 3 Grifoni et al, Cell. 2020; 181: 1489 -1501. 2 Long 12

Thank you for participating in this month’s Clinical Chemistry Journal Club. Additional Journal Clubs are available at www. clinchem. org Follow us 13
- Slides: 13