Journal Club ASCEND Study Collaborative Group Bowman L

Journal Club ASCEND Study Collaborative Group, Bowman L, Mafham M, Wallendszus K, Stevens W, Buck G, Barton J, Murphy K, Aung T, Haynes R, Cox J, Murawska A, Young A, Lay M, Chen F, Sammons E, Waters E, Adler A, Bodansky J, Farmer A, Mc. Pherson R, Neil A, Simpson D, Peto R, Baigent C, Collins R, Parish S, Armitage J. Effects of n-3 Fatty Acid Supplements in Diabetes Mellitus. N Engl J Med. 2018 Oct 18; 379(16): 1540 -1550. doi: 10. 1056/NEJMoa 1804989. Bohula EA, Scirica BM, Inzucchi SE, Mc. Guire DK, Keech AC, Smith SR, Kanevsky E, Murphy SA, Leiter LA, Dwyer JP, Corbalan R, Hamm C, Kaplan L, Nicolau JC, Ophuis TO, Ray KK, Ruda M, Spinar J, Patel T, Miao W, Perdomo C, Francis B, Dhadda S, Bonaca MP, Ruff CT, Sabatine MS, Wiviott SD; CAMELLIATIMI 61 Steering Committee Investigators. Effect of lorcaserin on prevention and remission of type 2 diabetes in overweight and obese patients (CAMELLIA-TIMI 61): a randomised, placebo-controlled trial. Lancet. 2018 Nov 24; 392(10161): 2269 -2279. doi: 10. 1016/S 0140 -6736(18)32328 -6. Epub 2018 Oct 4. 2018年 12月6日 8: 30 -8: 55 4階 医局 埼玉医科大学 総合医療センター 内分泌・糖尿病内科 Department of Endocrinology and Diabetes, Saitama Medical Center, Saitama Medical University 松田 昌文 Matsuda, Masafumi
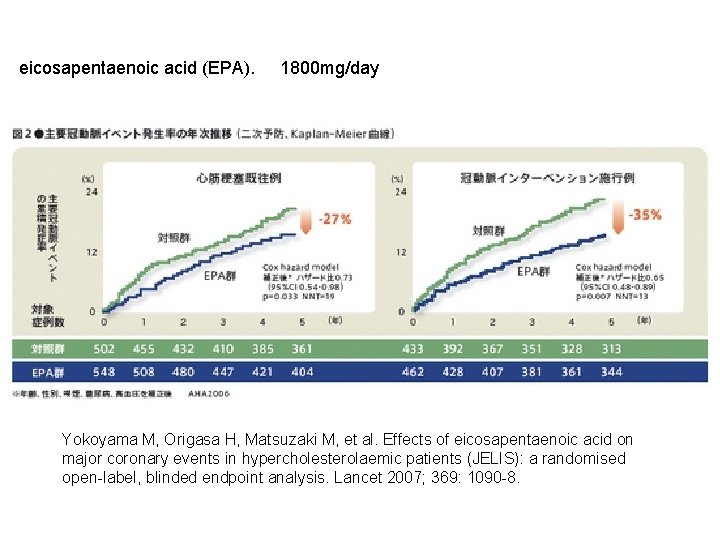
eicosapentaenoic acid (EPA). 1800 mg/day Yokoyama M, Origasa H, Matsuzaki M, et al. Effects of eicosapentaenoic acid on major coronary events in hypercholesterolaemic patients (JELIS): a randomised open-label, blinded endpoint analysis. Lancet 2007; 369: 1090 -8.
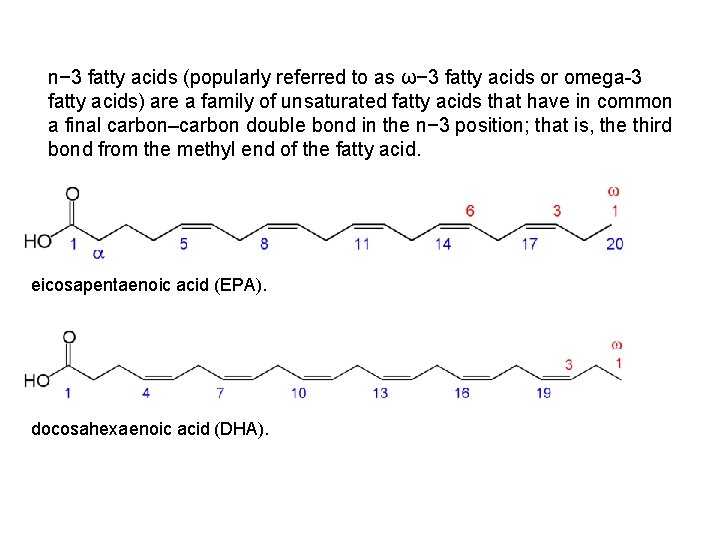
n− 3 fatty acids (popularly referred to as ω− 3 fatty acids or omega-3 fatty acids) are a family of unsaturated fatty acids that have in common a final carbon–carbon double bond in the n− 3 position; that is, the third bond from the methyl end of the fatty acid. eicosapentaenoic acid (EPA). docosahexaenoic acid (DHA).

2010年 9月2日抄読会 the Division of Human Nutrition, Wageningen University, Wageningen (D. K. , J. M. G. ); and the Department of Psychiatry, Leiden University Medical Center, Leiden (E. J. G. ) in the Netherlands. 10. 1056/nejmoa 1003603 nejm. org
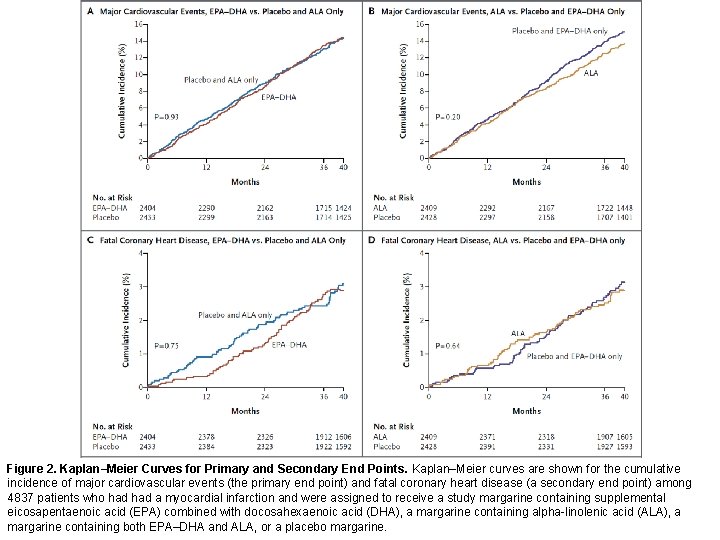
Figure 2. Kaplan–Meier Curves for Primary and Secondary End Points. Kaplan–Meier curves are shown for the cumulative incidence of major cardiovascular events (the primary end point) and fatal coronary heart disease (a secondary end point) among 4837 patients who had a myocardial infarction and were assigned to receive a study margarine containing supplemental eicosapentaenoic acid (EPA) combined with docosahexaenoic acid (DHA), a margarine containing alpha-linolenic acid (ALA), a margarine containing both EPA–DHA and ALA, or a placebo margarine.

Coordinating Office (Clinical Trial Service Unit, Nuffield Department of Population Health, University of Oxford): N Engl J Med. 2018 Oct 18; 379(16): 1540 -1550. doi: 10. 1056/NEJMoa 1804989.

Background Increased intake of n− 3 fatty acids has been associated with a reduced risk of cardiovascular disease in observational studies, but this finding has not been confirmed in randomized trials. It remains unclear whether n− 3 (also called omega 3) fatty acid supplementation has cardiovascular benefit in patients with diabetes mellitus.

Methods We randomly assigned 15, 480 patients with diabetes but without evidence of atherosclerotic cardiovascular disease to receive 1 -g capsules containing either n− 3 fatty acids (fatty acid group) or matching placebo (olive oil) daily. The primary outcome was a first serious vascular event (i. e. , nonfatal myocardial infarction or stroke, transient ischemic attack, or vascular death, excluding confirmed intracranial hemorrhage). The secondary outcome was a first serious vascular event or any arterial revascularization.
![Protocol version 6 November 20, 2003 Protocol [V 10. 2_2018 -07 -23] Protocol version 6 November 20, 2003 Protocol [V 10. 2_2018 -07 -23]](http://slidetodoc.com/presentation_image/0f94ebf6874b5aa06e48772c21facbe0/image-10.jpg)
Protocol version 6 November 20, 2003 Protocol [V 10. 2_2018 -07 -23]

Figure 1. Enrollment and Completeness of Data. 1 -g capsules containing either 840 mg of marine n− 3 fatty acids (460 mg of eicosapentaenoic acid [EPA] and 380 mg of docosahexaenoic acid [DHA]) (fatty acid group) or a matching placebo capsule (olive oil)

* Plus–minus values are means ±SD. There were no significant differences between the assigned groups. Percentages may not total 100 because of rounding. IQR denotes interquartile range. † Race and ethnic group were reported by the patients. Other groups were Indian, Pakistani, or Bangladeshi (1%), African or Caribbean (1%), and other or unknown (1%). ‡ The body-mass index (the weight in kilograms divided by the square of the height in meters) was based on values for height and weight reported by the patients. § The presence of type 2 diabetes was based on a broad clinical definition that included the age at the time of diagnosis, use of insulin within 1 year after diagnosis, and body-mass index. ¶ Blood pressure was measured at the time that a blood sample was obtained, generally before randomization. ‖ The vascular risk score was the predicted 5 -year risk of a serious vascular event (including transient ischemic attack) without the receipt of aspirin or n− 3 fatty acids. The risk was categorized as low (<5%), moderate (5% to <10%), and high (≥ 10%). Details about how this score was calculated are provided in Supplementary Appendix 1.

Figure 2. First Serious Vascular Events during Follow-up. Panel A shows a Kaplan–Meier plot of the first serious vascular event (a composite of nonfatal myocardial infarction or ischemic stroke, transient ischemic attack, or vascular death excluding confirmed intracranial hemorrhage) during follow-up. The numbers of patients at risk at the start of each year of follow-up are shown, along with the cumulative number (±SE) of patients in whom a serious vascular event was avoided per 1000 patients in the fatty acid group, as compared with the placebo group. The inset graph shows the same data on an expanded y axis. Panel B shows rate ratios for the first serious vascular event among the patients in the fatty acid group, as compared with the placebo group, according to follow-up period. The numbers at risk declined with each period of follow-up because of data censoring, so the percentages are the number of events as a proportion of the number of patients at risk at the start of the period. For each period of follow-up, rate ratios are plotted as squares, with the size of each square proportional to the amount of statistical information that was available. The horizontal lines represent 95% confidence intervals, which have not been
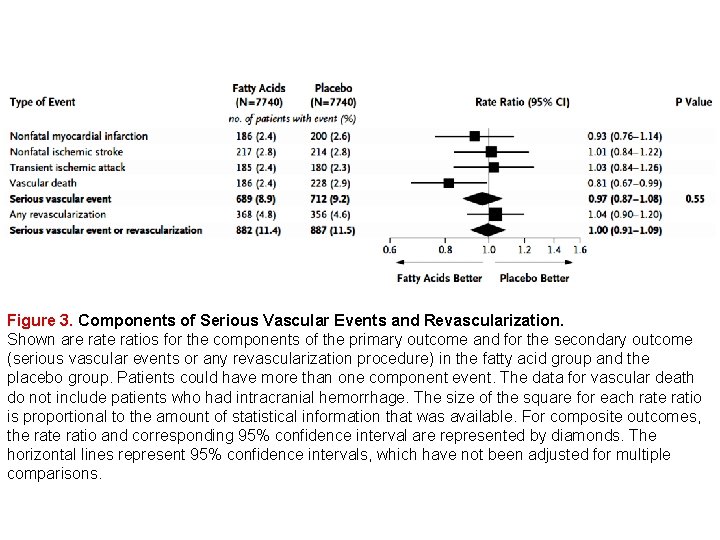
Figure 3. Components of Serious Vascular Events and Revascularization. Shown are ratios for the components of the primary outcome and for the secondary outcome (serious vascular events or any revascularization procedure) in the fatty acid group and the placebo group. Patients could have more than one component event. The data for vascular death do not include patients who had intracranial hemorrhage. The size of the square for each rate ratio is proportional to the amount of statistical information that was available. For composite outcomes, the ratio and corresponding 95% confidence interval are represented by diamonds. The horizontal lines represent 95% confidence intervals, which have not been adjusted for multiple comparisons.
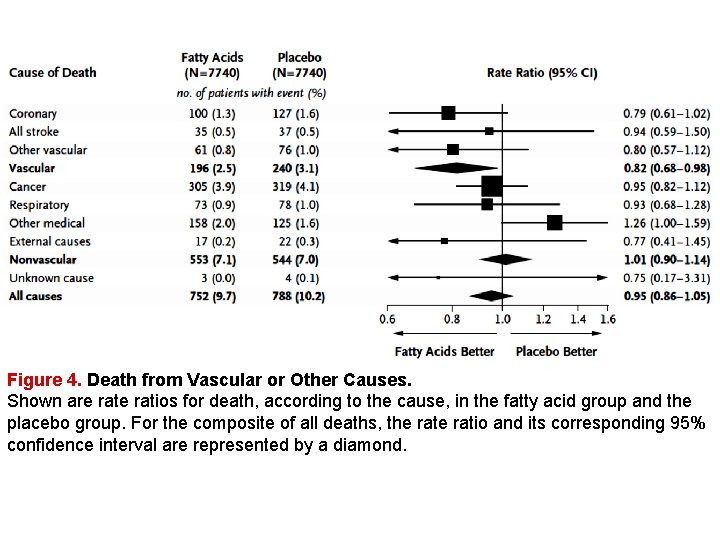
Figure 4. Death from Vascular or Other Causes. Shown are ratios for death, according to the cause, in the fatty acid group and the placebo group. For the composite of all deaths, the ratio and its corresponding 95% confidence interval are represented by a diamond.

Results During a mean follow-up of 7. 4 years (adherence rate, 76%), a serious vascular event occurred in 689 patients (8. 9%) in the fatty acid group and in 712 (9. 2%) in the placebo group (rate ratio, 0. 97; 95% confidence interval [CI], 0. 87 to 1. 08; P=0. 55). The composite outcome of a serious vascular event or revascularization occurred in 882 patients (11. 4%) and 887 patients (11. 5%), respectively (rate ratio, 1. 00; 95% CI, 0. 91 to 1. 09). Death from any cause occurred in 752 patients (9. 7%) in the fatty acid group and in 788 (10. 2%) in the placebo group (rate ratio, 0. 95; 95% CI, 0. 86 to 1. 05). There were no significant between-group differences in the rates of nonfatal serious adverse events.

Conclusions Among patients with diabetes without evidence of cardiovascular disease, there was no significant difference in the risk of serious vascular events between those who were assigned to receive n− 3 fatty acid supplementation and those who were assigned to receive placebo. (Funded by the British Heart Foundation and others; Current Controlled Trials number, ISRCTN 60635500; Clinical. Trials. gov number, NCT 00135226. )

To be reviewed? The ASCEND Study Collaborative Group. Effects of aspirin for primary prevention in persons with diabetes mellitus. N Engl J Med 2018; 379: 1529 -1539



https: //www. eisai. co. jp/news/2018/pdf/news 201862 pdf. pdf




2012年 5月17日抄読会 1 Weight Management Center, Department of Psychiatry and Behavioral Sciences, Medical University of South Carolina, Charleston, South Carolina, USA; 2 Translational Research Institute for Metabolism and Diabetes, Florida Hospital and the Sanford-Burnham Medical Research Institute, Orlando, Florida, USA; 3 Med. Star Health Research Institute at Washington Hospital Center and Georgetown University, Washington, DC, USA; 4 Arena Pharmaceuticals, San Diego, California, USA. obesity doi: 10. 1038/oby. 2012. 66
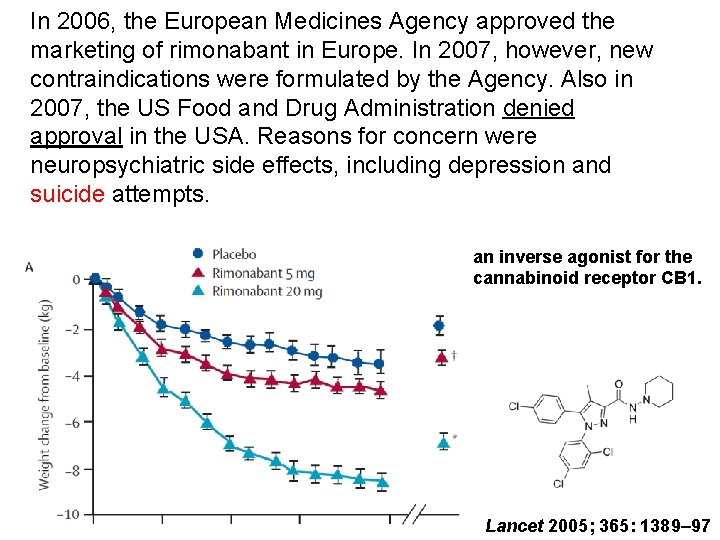
In 2006, the European Medicines Agency approved the marketing of rimonabant in Europe. In 2007, however, new contraindications were formulated by the Agency. Also in 2007, the US Food and Drug Administration denied approval in the USA. Reasons for concern were neuropsychiatric side effects, including depression and suicide attempts. an inverse agonist for the cannabinoid receptor CB 1. Lancet 2005; 365: 1389– 97
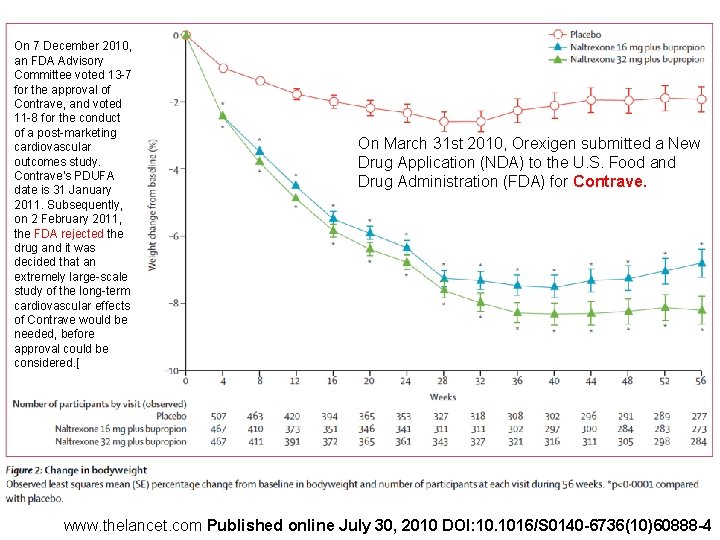
On 7 December 2010, an FDA Advisory Committee voted 13 -7 for the approval of Contrave, and voted 11 -8 for the conduct of a post-marketing cardiovascular outcomes study. Contrave's PDUFA date is 31 January 2011. Subsequently, on 2 February 2011, the FDA rejected the drug and it was decided that an extremely large-scale study of the long-term cardiovascular effects of Contrave would be needed, before approval could be considered. [ On March 31 st 2010, Orexigen submitted a New Drug Application (NDA) to the U. S. Food and Drug Administration (FDA) for Contrave. www. thelancet. com Published online July 30, 2010 DOI: 10. 1016/S 0140 -6736(10)60888 -4
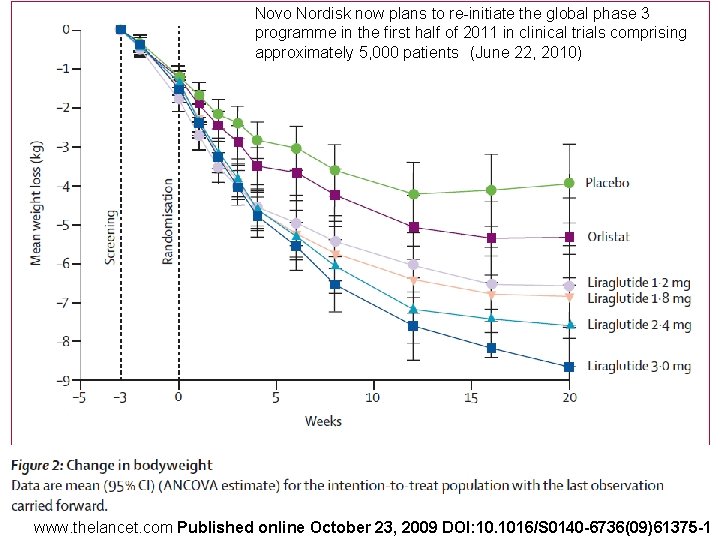
Novo Nordisk now plans to re-initiate the global phase 3 programme in the first half of 2011 in clinical trials comprising approximately 5, 000 patients (June 22, 2010) www. thelancet. com Published online October 23, 2009 DOI: 10. 1016/S 0140 -6736(09)61375 -1

Phentermine/topiramate (Qnexa) Phentermine is an appetite suppressant and stimulant of the amphetamine and phenethylamine class. Topiramate is an anticonvulsant that has weight loss side effects. On December 28, 2009 a new drug application (NDA) was submitted to the FDA for approval and on March 1, 2010, Vivus announced that the agency accepted the NDA. FDA approval was declined in October 2010 due to concerns about dangerous side effects, including suicidal thoughts, heart palpitations, memory lapses and birth defects. In January 2011, the Food and Drug Administration expressed concerns about the potential for Qnexa to cause birth defects and asked Vivus to examine this possibility before the drug can be approved. On February 22, 2012, FDA advisors voted 20: 2 to recommend that the FDA adopt phentermine/topiramate as an obesity treatment. Final approval is expected later in 2012, with recommendations for post-market monitoring for cardiovascular risk and an indication against use by pregnant women.

http: //en. wikipedia. org/wiki/5 -HT_receptor Serotonin or 5 -hydroxytryptamine (5 -HT) is a monoamine neurotransmitter. The serotonin receptors modulate the release of many neurotransmitters, including glutamate, GABA, dopamine, epinephrine / norepinephrine, and acetylcholine, as well as many hormones, including oxytocin, prolactin, vasopressin, cortisol, corticotropin, and substance P, among others. The serotonin receptors influence various biological and neurological processes such as aggression, anxiety, appetite, cognition, learning, memory, mood, nausea, sleep, and thermoregulation. The serotonin receptors are the target of a variety of pharmaceutical and illicit drugs, including many antidepressants, antipsychotics, anorectics, antiemetics, gastroprokinetic agents, antimigraine agents, hallucinogens, and entactogens.
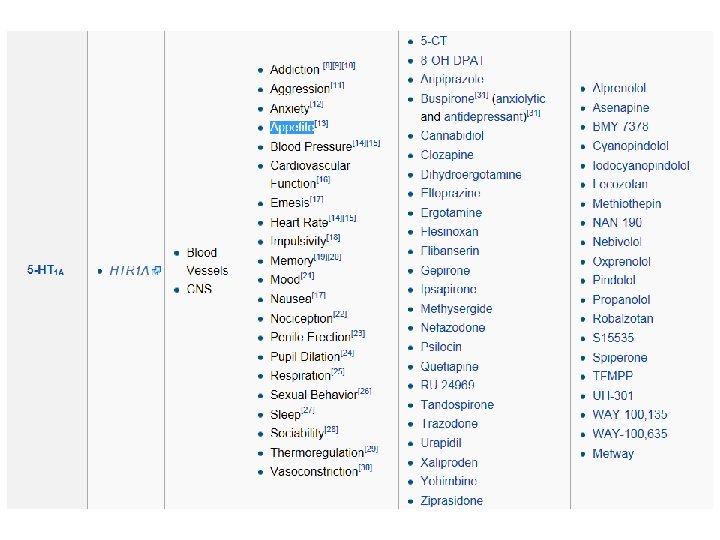
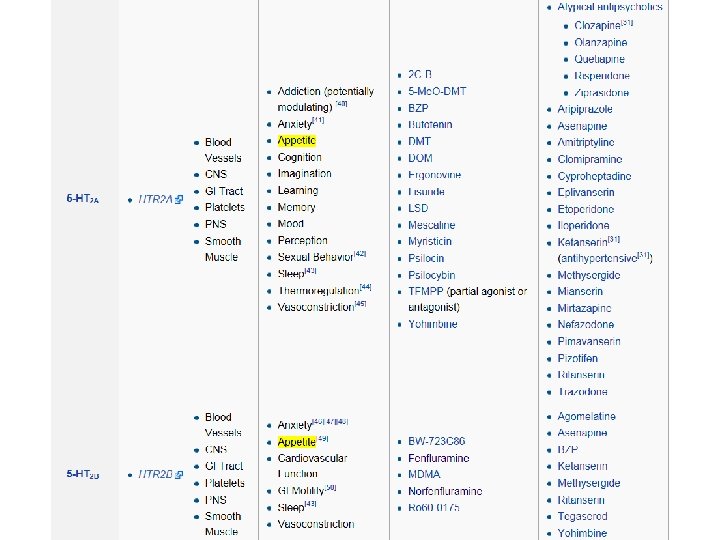
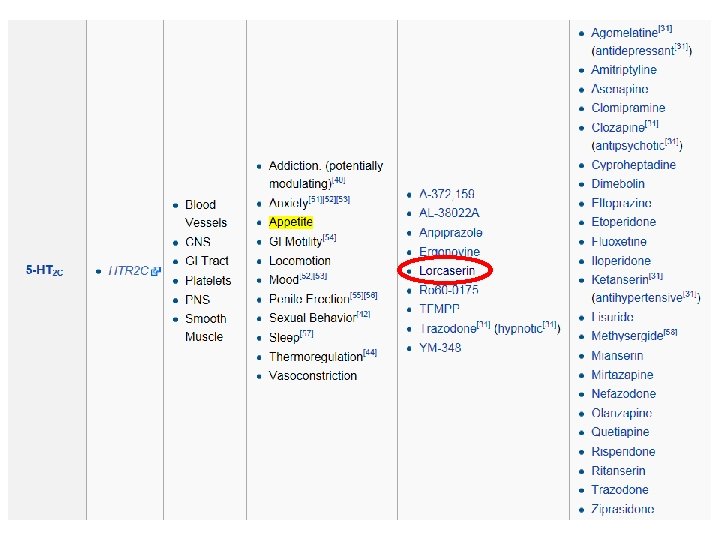

Note that there is no 5 -HT 1 C receptor since, after the receptor was cloned and further characterized, it was found to have more in common with the 5 -HT 2 family of receptors and was redesignated as the 5 -HT 2 C receptor. Note that there is also no 5 -HT 5 B receptor, as it exists only in mice and rats and not in humans or monkeys. Very nonselective agonists of 5 -HT receptor subtypes include ergotamine (an antimigraine), which activates 5 -HT 1 A, 5 -HT 1 D, 5 -HT 1 B, D 2 and norepinephrine receptors. LSD (a psychedelic) is a 5 -HT 1 A, 5 -HT 2 C, 5 -HT 5 A, 5 -HT 5, 5 -HT 6 agonist.
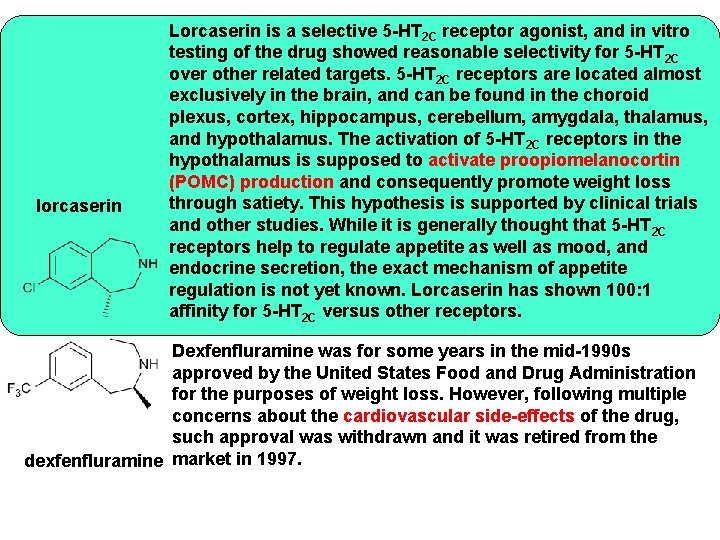
lorcaserin Lorcaserin is a selective 5 -HT 2 C receptor agonist, and in vitro testing of the drug showed reasonable selectivity for 5 -HT 2 C over other related targets. 5 -HT 2 C receptors are located almost exclusively in the brain, and can be found in the choroid plexus, cortex, hippocampus, cerebellum, amygdala, thalamus, and hypothalamus. The activation of 5 -HT 2 C receptors in the hypothalamus is supposed to activate proopiomelanocortin (POMC) production and consequently promote weight loss through satiety. This hypothesis is supported by clinical trials and other studies. While it is generally thought that 5 -HT 2 C receptors help to regulate appetite as well as mood, and endocrine secretion, the exact mechanism of appetite regulation is not yet known. Lorcaserin has shown 100: 1 affinity for 5 -HT 2 C versus other receptors. Dexfenfluramine was for some years in the mid-1990 s approved by the United States Food and Drug Administration for the purposes of weight loss. However, following multiple concerns about the cardiovascular side-effects of the drug, such approval was withdrawn and it was retired from the dexfenfluramine market in 1997.

Dexfenfluramine is believed to be solely responsible for the appetite suppressant properties of fenfluramine, of which it has been demonstrated to mediate predominantly via activation of postsynaptic 5 -HT 1 B and 5 -HT 2 C receptors through a combination of indirect serotonin releasing agent and direct serotonin receptor agonist activities (the latter of which are mediated fully by its active metabolite dexnorfenfluramine). Levonorfenfluramine, an active metabolite of levofenfluramine, is also a fairly potent serotonin releasing agent (with a potency of approximately 1/2 that of norfenfluramine and 1/6 th that of dexfenfluramine) and, similarly to dexnorfenfluramine, 5 -HT 2 B and 5 -HT 2 C receptor agonist, as well as a somewhat less potent norepinephrine reuptake inhibitor (about 1/2 that of its efficacy as a serotonin releaser). Norfenfluramine, or 3 -trifluoromethylamphetamine, is a drug of the amphetamine family which behaves as a serotonin and norepinephrine releasing agent and potent 5 -HT 2 A, 5 -HT 2 B, and 5 HT 2 C agonist.

fenfluramine Fenfluramine was introduced on the U. S. market in 1973. It is the racemic mixture of two enantiomers, dextrofenfluramine and levofenfluramine. It increases the level of the neurotransmitter serotonin, a chemical that regulates mood, appetite and other functions. Fenfluramine causes the release of serotonin by disrupting vesicular storage of the neurotransmitter, and reversing serotonin transporter function. The result is a feeling of fullness and loss of appetite. The drug was withdrawn from the U. S. market in 1997 after reports of heart valve disease, and pulmonary hypertension, including a condition known as cardiac fibrosis. Since fenfluramine and its active metabolite norfenfluramine stimulate serotonin receptors, this may have led to the valvular abnormalities found in patients using fenfluramine. In particular norfenfluramine is a potent agonist of 5 -HT 2 B receptors, which are plentiful in human cardiac valves. Both fenfluramine and benfluorex form norfenfluramine as a metabolite. This side effect led to the withdrawal of fenfluramine as an anorectic drug worldwide, and later to the withdrawal of benfluorex in Europe. benfluorex

Lorcaserin (APD-356, trade name Lorqess) is a weight-loss drug developed by Arena Pharmaceuticals. It has serotonergic properties and acts as an anorectic. On 22 December 2009 a New Drug Application (NDA) was submitted to the Food and Drug Administration (FDA) in the United States. On 16 September 2010, an FDA advisory panel voted to recommend against approval of the drug based on concerns over both safety and efficacy. In October 2010, the FDA stated that it could not approve the application for lorcaserin in its present form. On 10 May 2012, after a new round of studies submitted by Arena, an FDA panel voted to recommend lorcaserin with certain restrictions and patient monitoring. The restrictions include patients with a BMI of over 30, or with a BMI over 27 and a comorbidity like high blood pressure or type 2 diabetes.


TIMI Study Group, Division of Cardiovascular Medicine, Brigham and Women’s Hospital, Boston, MA, USA (E A Bohula MD, B M Scirica MD, E Kanevsky MS, S A Murphy MPH, M P Bonaca MD, C T Ruff MD, Prof M S Sabatine MD, S D Wiviott MD); Section of Endocrinology, Yale School of Medicine, New Haven, CT, USA (Prof S E Inzucchi MD); Division of Cardiology, University of Texas Southwestern Medical Center, Dallas, TX, USA (Prof D K Mc. Guire MD); NHMRC Clinical Trials Centre, University of Sydney, NSW, Australia (Prof A C Keech MD); Translational Research Institute for Metabolism and Diabetes, Florida Hospital, Orlando, FL, USA (S R Smith MD); Li Ka Shing Knowledge Institute, St Michael’s Hospital, University of Toronto, ON, Canada (Prof L A Leiter MD); Division of Nephrology/ Hypertension, Vanderbilt University Medical Center, Nashville, TN, USA (Prof J P Dwyer MD); Department of Cardiovascular Diseases, Catholic University School of Medicine, Santiago, Chile (R Corbalan MD); The Kerckhoff Heart Center, Bad Nauheim, Germany (Prof C Hamm MD); Obesity, Metabolism and Nutrition Institute, Massachusetts General Hospital, Boston, MA, USA (L Kaplan MD); Heart Institute, University of São Paulo, Brazil (Prof J C Nicolau MD); CWZ Hospital, Nijmegen, Netherlands (T O Ophuis MD); Imperial Centre for Cardiovascular Disease Prevention, School of Public Health, Imperial College London, UK (Prof K K Ray FRCP); Russian Cardiologic Research and Production Complex of Rosmedtechnology, Moscow, Russia (Prof M Ruda MD); Internal Cardiology Department, University Hospital Brno, Czech Republic (J Spinar MD); and Eisai, Woodcliff Lake, NJ, USA (T Patel Ph. D, W Miao MD, C Perdomo MS, B Francis MD, S Dhadda Ph. D) Lancet. 2018 Nov 24; 392(10161): 2269 -2279. doi: 10. 1016/S 0140 -6736(18)32328 -6.

Background There is a direct relationship between bodyweight and risk of diabetes. Lorcaserin, a selective serotonin 2 C receptor agonist that suppresses appetite, has been shown to facilitate sustained weight loss in obese or overweight patients. We aimed to evaluate the long-term effects of lorcaserin on diabetes prevention and remission.

Methods In this randomised, double-blind, placebo-controlled trial done in eight countries, we recruited overweight or obese patients (body-mass index ≥ 27 kg/m 2) with or at high risk for atherosclerotic vascular disease. Eligible patients were aged 40 years or older; patients at high risk for atherosclerotic vascular disease had to be aged 50 years or older with diabetes and at least one other risk factor. Patients were randomly assigned to receive either lorcaserin (10 mg twice daily) or matching placebo. Additionally, all patients had access to a standardised weight management programme based on lifestyle modification. The prespecified primary metabolic efficacy endpoint of time to incident diabetes was assessed in patients with prediabetes at baseline. The prespecified secondary outcomes for efficacy were incident diabetes in all patients without diabetes, achievement of normoglycaemia in patients with prediabetes, and change in glycated haemoglobin (Hb. A 1 c) in patients with diabetes. Hypoglycaemia was a prespecified safety outcome. Analysis was by intention to treat, using Cox proportional hazard models for time-to-event analyses. This trial is registered with Clinical. Trials. gov, number NCT 02019264.
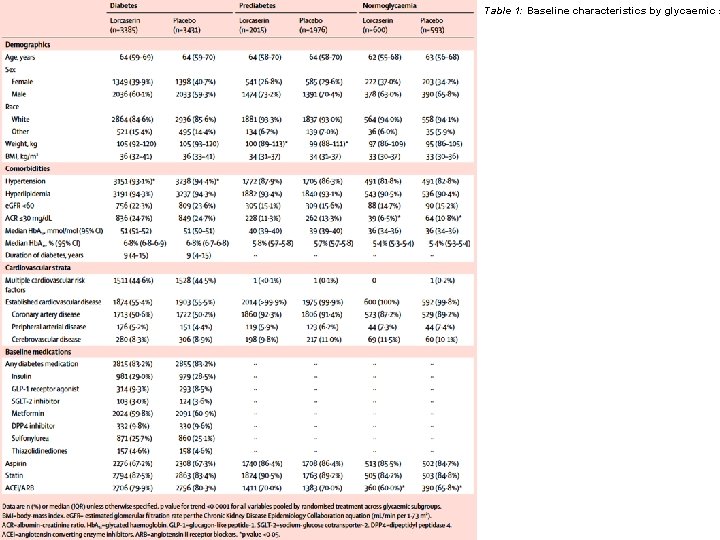
Table 1: Baseline characteristics by glycaemic s
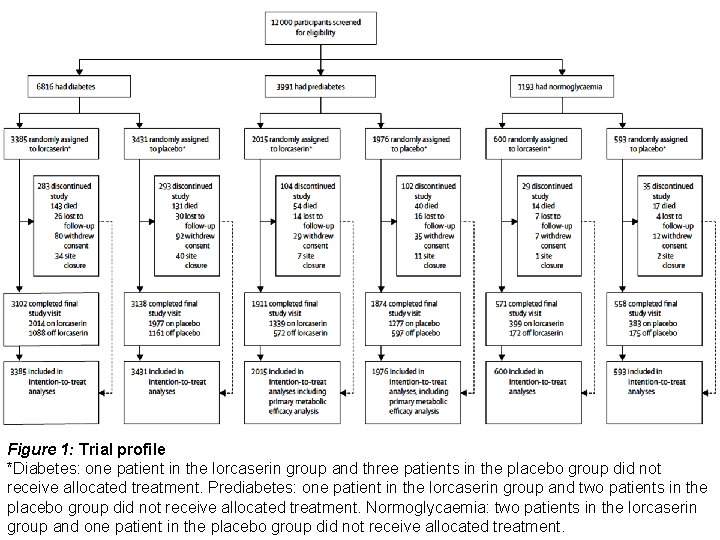
Figure 1: Trial profile *Diabetes: one patient in the lorcaserin group and three patients in the placebo group did not receive allocated treatment. Prediabetes: one patient in the lorcaserin group and two patients in the placebo group did not receive allocated treatment. Normoglycaemia: two patients in the lorcaserin group and one patient in the placebo group did not receive allocated treatment.
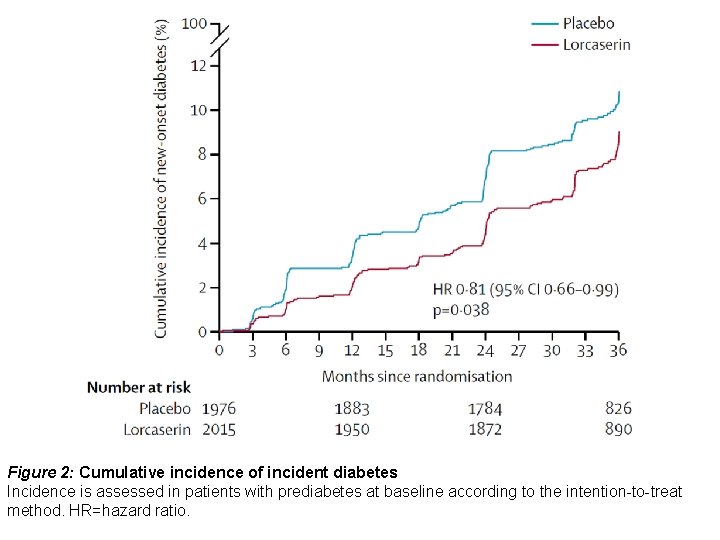
Figure 2: Cumulative incidence of incident diabetes Incidence is assessed in patients with prediabetes at baseline according to the intention-to-treat method. HR=hazard ratio.
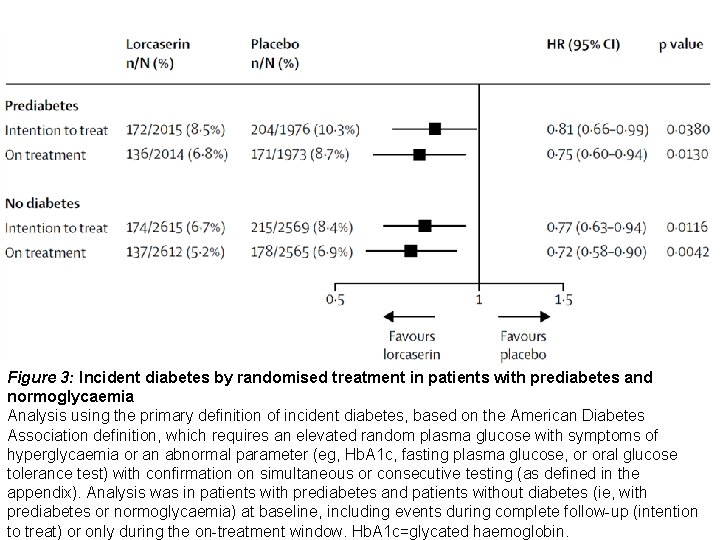
Figure 3: Incident diabetes by randomised treatment in patients with prediabetes and normoglycaemia Analysis using the primary definition of incident diabetes, based on the American Diabetes Association definition, which requires an elevated random plasma glucose with symptoms of hyperglycaemia or an abnormal parameter (eg, Hb. A 1 c, fasting plasma glucose, or oral glucose tolerance test) with confirmation on simultaneous or consecutive testing (as defined in the appendix). Analysis was in patients with prediabetes and patients without diabetes (ie, with prediabetes or normoglycaemia) at baseline, including events during complete follow-up (intention to treat) or only during the on-treatment window. Hb. A 1 c=glycated haemoglobin.

DBW DHb. A 1 c DFBG 1. 0 mmol/L =18 mg/d. L Figure 4: Change in weight, Hb. A 1 c, and fasting plasma glucose by baseline glycaemic subgroup All analyses were done in the intention-to-treat population. Change from baseline shown as leastsquared mean (95% CI) based on linear mixed effect model with repeated measures including model terms with treatment, visit by treatment interaction, baseline value, and randomisation

Figure 5: Remission of diabetes or prediabetes Analyses are by intention to treat. Remission of hyperglycaemia is defined as Hb. A 1 c of <48 mmol/mol (6· 5%) and fasting plasma glucose <126 mg/d. L (<7· 0 mmol/L) in the absence of anti-hyperglycaemia medication in patients with diabetes at baseline. Achievement of normoglycaemia is defined as Hb. A 1 c of ≤ 38 mmol/mol (5· 6%) and fasting plasma glucose <100 mg/d. L (<5· 5 mmol/L) in the absence of anti-hyperglycaemia medication in patients with diabetes or prediabetes at baseline. Both criteria (Hb. A 1 c and fasting plasma glucose) are required only if both tests are available. “Persistent” requires criteria to be achieved, confirmed, and maintained through the duration of the study. “Sustained” requires criteria to be achieved, confirmed, and maintained for two consecutive measurements separated by 30 days or more. “Any” is defined by achievement of at least one criteria at one or more timepoints during the study. Hb. A 1 c=glycated haemoglobin.
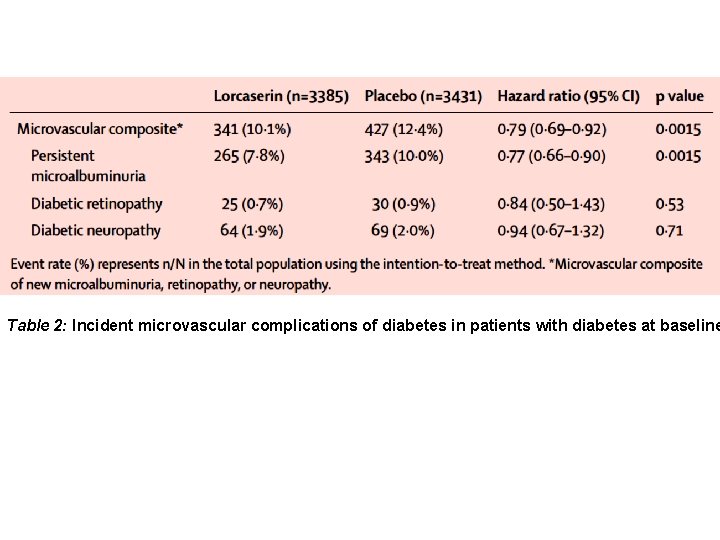
Table 2: Incident microvascular complications of diabetes in patients with diabetes at baseline


Findings Between Feb 7, 2014, and Nov 20, 2015, 12 000 patients were randomly assigned to lorcaserin or placebo (6000 patients in each group) and followed up for a median of 3· 3 years (IQR 3· 0– 3· 5). At baseline, 6816 patients (56· 8%) had diabetes, 3991 (33· 3%) prediabetes, and 1193 (9· 9%) normoglycaemia. At 1 year, patients treated with lorcaserin had a net weight loss beyond placebo of 2· 6 kg (95% CI 2· 3– 2· 9) for those with diabetes, 2· 8 kg (2· 5– 3· 2) for those with prediabetes, and 3· 3 kg (2· 6– 4· 0) for those with normoglycaemia (p<0· 0001 for all analyses). Lorcaserin reduced the risk of incident diabetes by 19% in patients with prediabetes (172 [8· 5%] of 2015 vs 204 [10· 3%] of 1976; hazard ratio 0· 81, 95% CI 0· 66– 0· 99; p=0· 038) and by 23% in patients without diabetes (174 [6· 7%] of 2615 vs 215 [8· 4%] of 2569; 0· 77, 0· 63– 0· 94; p=0· 012). Lorcaserin resulted in a non-significant increase in the rate of achievement of normoglycaemia in patients with prediabetes (185 [9· 2%] vs 151 [7· 6%]; 1· 20, 0· 97– 1· 49; p=0· 093). In patients with diabetes, lorcaserin resulted in a reduction of 0· 33% (95% CI 0· 29– 0· 38; p<0· 0001) in Hb. A 1 c compared with placebo at 1 year from a mean baseline of 53 mmol/mol (7· 0%). In patients with diabetes at baseline, severe hypoglycaemia with serious complications was rare, but more common with lorcaserin (12 [0· 4%] vs four [0· 1%] events; p=0· 054).

Interpretation Lorcaserin decreases risk for incident diabetes, induces remission of hyperglycaemia, and reduces the risk of microvascular complications in obese and overweight patients, supporting the role of lorcaserin as an adjunct to lifestyle modification for chronic management of weight and metabolic health. Funding Eisai.


- Slides: 55