Journal Club April 2018 2 article review Introduction

Journal Club April 2018 2 article review



§ Introduction § (1, 3)-β-D glucan (BG) § Pan fungal diagnostic marker § Outcome & prognosis tool § Incorporating BDG into new management strategies for IFI § Aspergillus galactomannan § As early diagnose invasive aspergillosis § As monitoring treatment of IA § Research study review § Discussion § Conclusion

§ Invasive fungal infections (IFI) present a great challenge in modern medicine dt the expansion of the population of immunosuppressed pts worldwide & has greatly increased, with high morbidity and mortality. § Many pt with a clinical suspicion of the presence of an IFI are treated empirically with antifungal therapy, which may involve the unnecessary use of potentially toxic and costly drugs. § The problem is that the diagnosis of IFI is difficult, with non-specific and subtle clinical manifestations & lack of sensitivity of conventional culture methods. § These recent development of serological markers represents a clear advance in the field. § (1, 3)-β-D glucan (BG) § Aspergillus galactomannan. § Key elements for improving the outcome of IFI is the early initiation of appropriate antifungal, which can be accomplished by the early diagnosis such infections

§ Invasive aspergillosis (IA) represents a major threat in people with depressed immune system esp in pts with: - hematologic malignancies - hematopoietic stem cell transplantation (HSCT) - solid tumors - congenital immunodeficiency - chemotherapy-induced neutropenia - solid-organ transplant recipients, - auto-immune disorders - chronic pulmonary diseases. § Non culture-based methods, such as detection of DNA by PCR or measurements of fungal biomarkers in blood or respiratory samples, are important adjunctive tools. § GM and BDG assays have been included as microbiological criteria in the definitions of IFI by the latest version of EORTC and MSG. § The role and interpretation of these tests for the diagnosis of IA in various populations and clinical settings is still debated because of their limited sensitivity and specificity. § Objective of this review is to provide an overview of the current evidence of the performance of these tests and to discuss their role and interpretation in diverse pts populations.
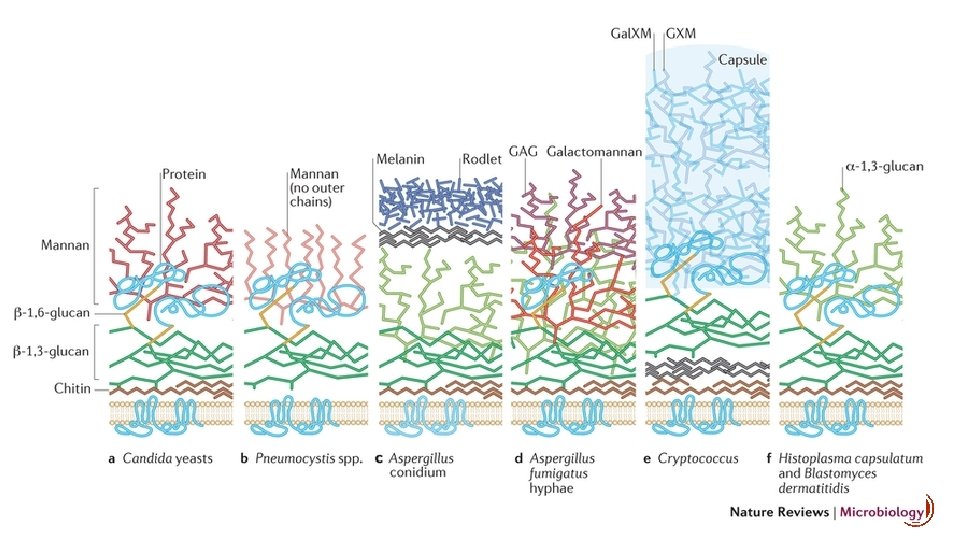


§ (1, 3)-β-DG is an important structural component of the majority of fungal cell walls. § High concentrations of BG in their cell walls Candida, Saccharomyces, Trichosporon, Sporothrix, Penicillium, Fusarium, and Aspergillus. § Others fungal disease that have lower concentration BG: § Scedosporium § Cryptoccocus neoformans § agents of mucormycosis : Mucor spp & Rhizopus spp § agents of endemic mycoses, such as Histoplasma, Coccidioides, and Blastomyces. § It is widely considered that BG results represent active infection and are not affected by; § fungal colonization. § Antifungal therapy
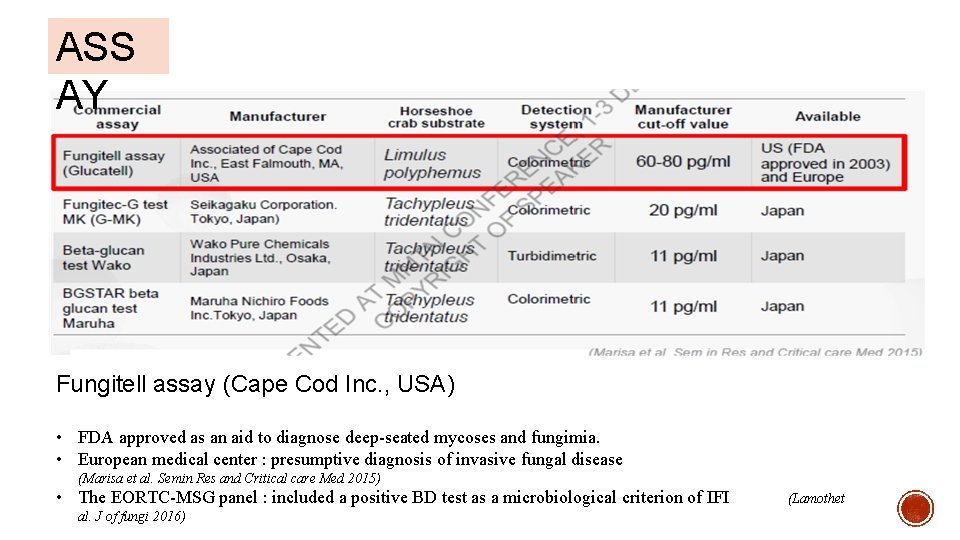
ASS AY Fungitell assay (Cape Cod Inc. , USA) • FDA approved as an aid to diagnose deep-seated mycoses and fungimia. • European medical center : presumptive diagnosis of invasive fungal disease (Marisa et al. Semin Res and Critical care Med 2015) • The EORTC-MSG panel : included a positive BD test as a microbiological criterion of IFI al. J of fungi 2016) (Lamothet

§ This assay is a chromogenic, quantitative enzyme immunoassay (EIA) designed to detect BDG by using purified, lysed horseshoe crab (Tachypleus in Asia and Limulus in North America) amebocytes § The majority of assays are constructed as enzymatic/ colorimetric methods based on the fact that BG triggers the coagulation cascade of the amebocyte cells of the horseshoe crab through the factor G pathway § It is also important to consider that amebocyte lysates from the different crab species have different affinity to BG, resulting in different cut-off values for different assays. § Therefore it is very important to consider the specific kit and region of the world where it is made when interpreting results, as well as the related literature.

MARKER DIAGNOSTIC § Several study had been done in initial validation of BG as diagnostic marker;

§ Unfortunately, BD is not pathogen specific and cannot differentiate fungal species. § • Major limitations : frequent occurrence of false-positive reaction § Non-fungal infection −Pseudomonas spp & −Streptococcus spp. § Non-infectious disease conditions § Hemodialysis with cellulose membranes § IV medication IVIG, Albumin & Antibiotic (Iv amoxicillin-clavulanate) § Use of cellulose filters for intravenous administration § Surgical Gauze

PROGNOSIS ASSESSMENT TOOL § Beyond diagnosis, BG assay also has now been focused on the correlation of BG levels with outcomes and prognosis. § Although evidence is accumulating for the use of BG for these purposes, this approach is still considered experimental and has not been endorsed by any guidelines.

§ Aside from diagnostic and prognostic applications of BG, research is now focusing on incorporating BG monitoring as part of an early or “pre-emptive” tx approach. § There are no data supporting the use of BDG testing in BAL
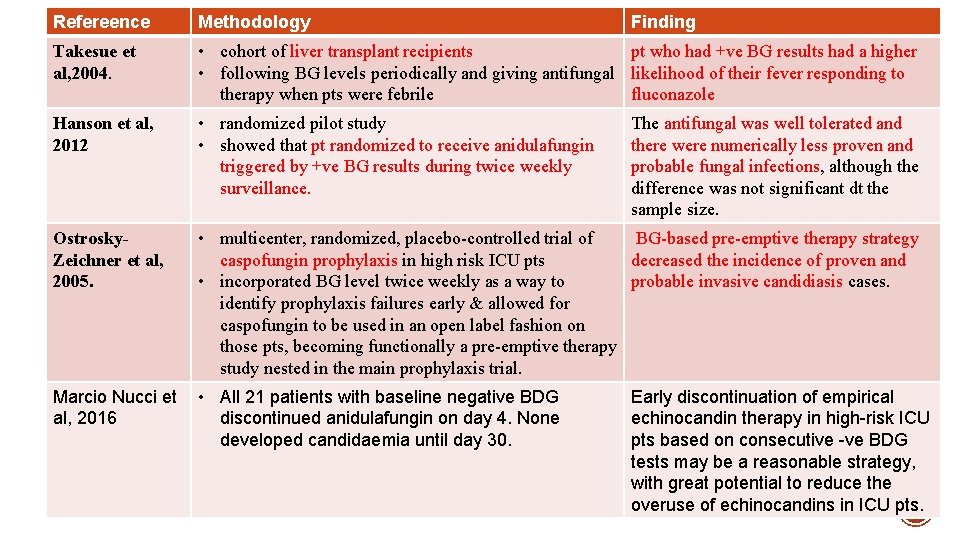
Refereence Methodology Finding Takesue et al, 2004. • cohort of liver transplant recipients pt who had +ve BG results had a higher • following BG levels periodically and giving antifungal likelihood of their fever responding to therapy when pts were febrile fluconazole Hanson et al, 2012 • randomized pilot study • showed that pt randomized to receive anidulafungin triggered by +ve BG results during twice weekly surveillance. Ostrosky. Zeichner et al, 2005. • multicenter, randomized, placebo-controlled trial of BG-based pre-emptive therapy strategy caspofungin prophylaxis in high risk ICU pts decreased the incidence of proven and • incorporated BG level twice weekly as a way to probable invasive candidiasis cases. identify prophylaxis failures early & allowed for caspofungin to be used in an open label fashion on those pts, becoming functionally a pre-emptive therapy study nested in the main prophylaxis trial. Marcio Nucci et al, 2016 • All 21 patients with baseline negative BDG discontinued anidulafungin on day 4. None developed candidaemia until day 30. The antifungal was well tolerated and there were numerically less proven and probable fungal infections, although the difference was not significant dt the sample size. Early discontinuation of empirical echinocandin therapy in high-risk ICU pts based on consecutive -ve BDG tests may be a reasonable strategy, with great potential to reduce the overuse of echinocandins in ICU pts.

§ BDG assay is § less specific in the early stages, § good negative predictive value and so it could be of help in order to reduce the percentage of false positives. § The fact that the BDG assay was positive for all of the cases of IA (probable and proven) confirms that this test could be a useful tool to screen for IA.


§ Major component of fungal cell wall (polysaccharide) especially Aspergillus spp. and also found in § Family of Trichocomaceaeie, § Penicillium spp. § Paecilomyces spp. § Fusarium spp. § Histoplasma capsulatum § Pneumocystis jirovecii

§ The Platelia® Aspergillus EIA is a one stage immune enzymatic sandwich microplate assay which detects circulating GM from body fluids (mainly serum and BAL). § This enzyme immunoassay uses the rat monoclonal antibody EBA-2 directed against Aspergillus galactomannan. § The Ag is first bound in the wells of the microplate coated with the EBA-2 antibody and then revealed by binding to the peroxidase-linked EBA-2 antibody resulting in a colorimetric reaction after addition of the substrate. § The optical density (OD) is measured and expressed as a ratio of a control sample. § The cut-off of positivity was initially set up at an OD of 1. 5 and subsequently lowered at 0. 5 by the manufacturer. § The EORTC-MSG panel included a positive GM value as a microbiological criterion of IFI, without specifying a threshold to define a positive result. § (ECIL) lab working group recommended to consider a single value of > 0. 7 or two consecutive values > 0. 5 in serum as the cut-off that should prompt further diagnostic work-up (e. g. , CT-scan) for presumptive IFI. § Higher cut-off (single value >0. 8 or 1. 0) should be considered for BAL and CSF samples
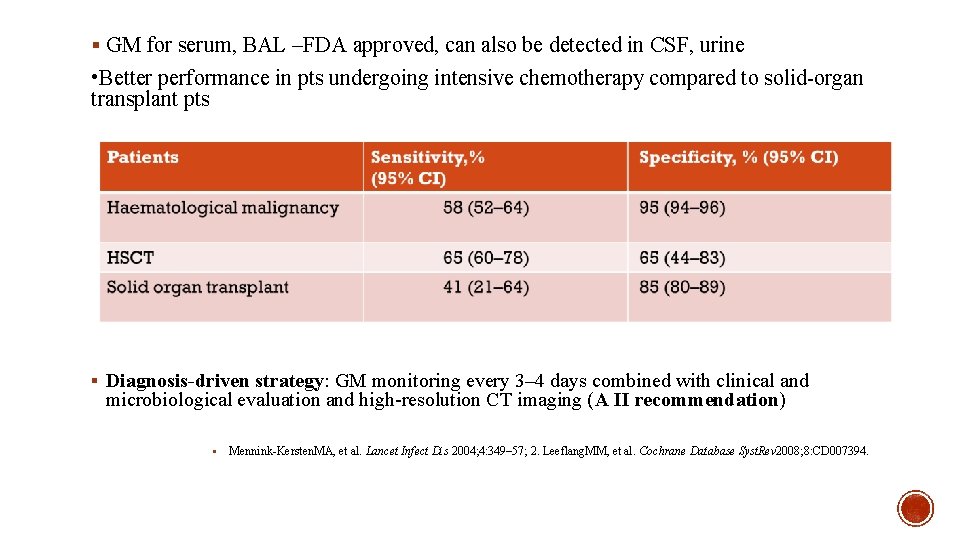
§ GM for serum, BAL –FDA approved, can also be detected in CSF, urine • Better performance in pts undergoing intensive chemotherapy compared to solid-organ transplant pts § Diagnosis-driven strategy: GM monitoring every 3– 4 days combined with clinical and microbiological evaluation and high-resolution CT imaging (A II recommendation) § Mennink-Kersten. MA, et al. Lancet Infect Dis 2004; 4: 349– 57; 2. Leeflang. MM, et al. Cochrane Database Syst. Rev 2008; 8: CD 007394.

EARLY DIAGNOSE § The serum galactomannan index assay (GMI) has been widely used for the dx of IA, with an excellent performance in neutropenic pts with hematological malignancies or in the preengraftment phase post-hematopoietic cell transplantation. § The sensitivity of the test in pts not receiving antifungal therapy was reported as 87. 5% - 96. 8% and specificity was 82. 4%, respectively. § The best use of GMI in this setting is : § screening method to early diagnose IA ; protocol for early dx or screening is to perform the test 3 x per week § to trigger the initiation of antifungal therapy (diagnostic-driven or preemptive antifungal therapy). § The easiest way of using GMI in this context is to rely on its high negative predictive value and rule out IA when the test is repeatedly negative.

§ Cause of false-negative test : § receiving an anti-mold fungal agent § no longer neutropenic pts § Therefore, if pt is neutropenic and is not receiving an anti-mold antifungal agent, repeatedly negative GMI is good at ruling out IA. § If one positive test, additional measures ( eg: chest and sinuses CT) made according to the pre- test probability of IA. § Clinician should rely on a curve rather than on just a single test. § Because animal models have consistently shown a sharp relationship between serum GMI and fungal burden in tissue, the best interpretation for an ascending GMI curve is that the pt is developing IA. § A consequence of the early diagnosis of IA is that the outcome is improved because the disease is being treated with a low fungal burden in the same way that the outcome of neoplastic diseases is better the lower the tumor burden.

TREATMENT MONITOR § Tx response in IA has relied on criteria that take into account improvement in clinical findings and imaging. § The problem is that sx are non-specific and radiological images frequently worsen during the course of treatment of IA. § Specifically, currently accepted criteria define response with a specific percentage of reduction in the volume of infiltrates. However, the reliability and reproducibility of such readings are greatly imprecise. § On the other hand, GMI trends down early in the course of disease, when pts are responding to tx, and its kinetics may be very useful to predict the outcome. § European Organization for Treatment and Research of Cancer/Mycosis Study Group response criteria with criteria based on the kinetics of GMI, success as survival defined as : § repeatedly negative serum GMI for ≥ 2 weeks after the first negative GMI in the absence of new extra- pulmonary IA lesions and failure as a persistently positive serum GMI. § The normalization of GMI within 7 days from the first positive test was strongly a/w the outcome. § Conclusion : These serum biomarker may be useful to monitor therapy as early as 7 days after diagnosis.

§ While GMI is very useful in neutropenic pts, its value in non-neutropenic pts is less certain. § In one study: § sensitivity of GMI was 23. 1% § specificity was 76. 1% § positive predictive value of 1. 6% § negative predictive value of 98. 3%. § In non-neutropenic patients false-negative results are more frequent. § Therefore, the best use of GMI is when results of sequential tests trend up. § In this setting, every attempt to confirm the diagnosis of IA should be advanced.
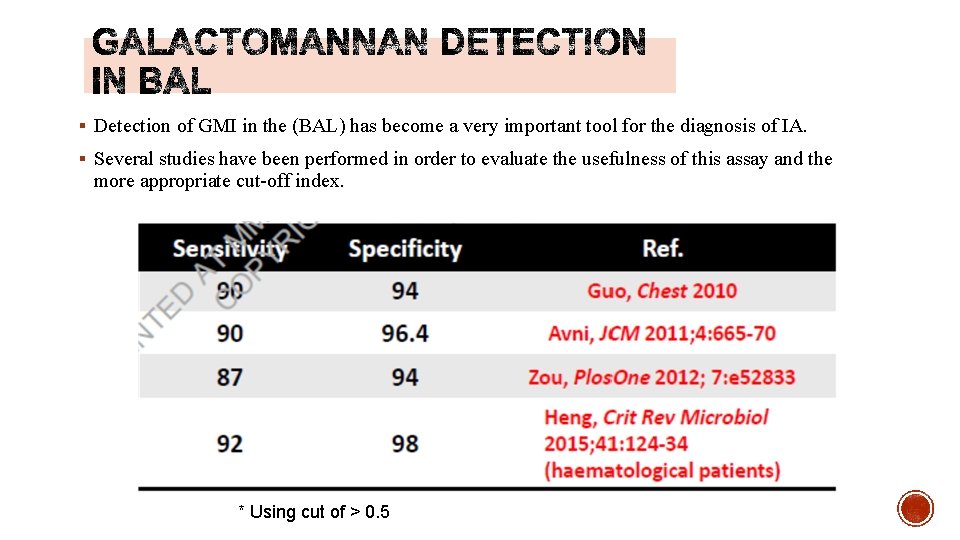
§ Detection of GMI in the (BAL) has become a very important tool for the diagnosis of IA. § Several studies have been performed in order to evaluate the usefulness of this assay and the more appropriate cut-off index. * Cut off > 1. 0 * Using cut of > 0. 5
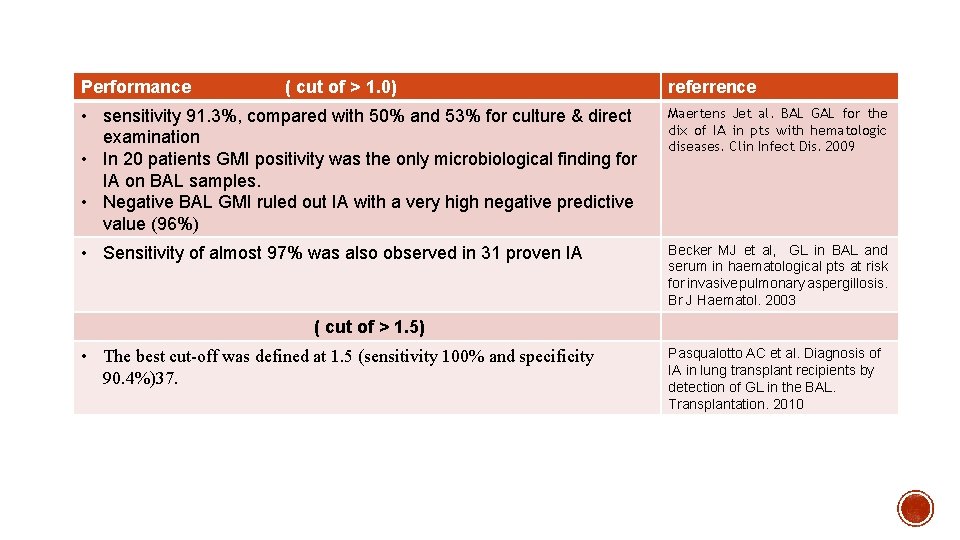
Performance ( cut of > 1. 0) referrence • sensitivity 91. 3%, compared with 50% and 53% for culture & direct examination • In 20 patients GMI positivity was the only microbiological finding for IA on BAL samples. • Negative BAL GMI ruled out IA with a very high negative predictive value (96%) Maertens Jet al. BAL GAL for the dix of IA in pts with hematologic diseases. Clin Infect Dis. 2009 • Sensitivity of almost 97% was also observed in 31 proven IA Becker MJ et al, GL in BAL and serum in haematological pts at risk for invasive pulmonary aspergillosis. Br J Haematol. 2003 ( cut of > 1. 5) • The best cut-off was defined at 1. 5 (sensitivity 100% and specificity 90. 4%)37. Pasqualotto AC et al. Diagnosis of IA in lung transplant recipients by detection of GL in the BAL. Transplantation. 2010

§ The performance of GM in BAL has been assessed in several studies and 2 meta-analyses suggest an overall performance of 85% sensitivity and 90%– 95% specificity. § The requirement of a higher cut-off (OD 1. 0 versus 0. 5) was associated with a substantial gain in specificity (95% vs. 90%, respectively), without loss of sensitivity. § Good diagnostic accuracy of GM in BAL was also reported in solid-organ transplant recipients (in particular lung transplant recipients) with sensitivity and specificity comparable to those observed in hematologic cancer patients. § Combination of GM testing with molecular methods (PCR targeting A. fumigatus or Aspergillus spp. ) has been suggested for improved performance (97% sensitivity and specificity for either GM or PCR positive). § Both the EORTC-MSG and ECIL panels have validated GM testing in CSF for the diagnosis of cerebral aspergillosis, which is supported by a recent study showing a sensitivity of 88% and a specificity of 96% in this setting.

§ The major conclusions from multiple studies, these analyses (GM & BDG) can be summarized as follows: § (1) Among high-risk pts with hematological malignancies & chemotherapy-induced neutropenia or allogeneic HSCT, both tests have a similar performance with a limited sensitivity (60%– 80%) & specificity > 90%. § (2) Requirement of 2 consecutive positive tests results in a ↑ specificity (95%– 99%), with a slight loss of sensitivity. § (3) Insufficient data are available to assess the performance of these tests in solid-organ transplant recipients and other populations of immunocompromised pts at low or moderate risk of IA. § However, sensitivity appears to be markedly decreased in this setting (40% or less), which can be explained by the limited angio-invasion in pts with better immune defenses compared to those with neutropenia. § Concerns about poor specificity have also been raised, in particular for BDG testing in lung transplant recipients in one study. § (4) Data among pediatric populations are also lacking. However, the performance of these tests (especially for GM) seems to be similar to that in adults, but lower specificity has been reported in some studies.

§ The diagnosis of IA relies on a constellation of clinical and paraclinical parameters, most of which are only presumptive and do not constitute per se a proof of infection. § Because of their limited sensitivity and specificity, GM and BDG results should only be interpreted in conjunction with other clinical, radiological and microbiological criteria of IA as defined by the EORTC-MSG consensus. § From this point of view, two important questions should come to mind regarding the utility of GM and BDG testing in clinical practice. § First, which patient should be tested? § Second, when and how the test should be performed (in other words, what should be the approach or strategy of screening)?

§ For a given sensitivity and specificity, the PPV & NPV values, will depend on the prevalence of the disease in a defined population and/or the pre-test probability of the disease in an individual. § If we consider a prevalence of IA of about 10% in a high-risk population of pts with hematologic malignancies & chemotherapy-induced neutropenia & a sensitivity and specificity of GM or BDG around 80% & 90% respectively, the PPV would be about 50% & NPV 97%. § If the pt has concomitant radiological signs consistent with IA (high pre-test probability), a +ve result would definitely indicate an at least probable IA. § However, assuming a similar specificity in a population with a lower IA incidence (e. g. , 2%– 3%), such as solid-organ transplant recipients, the PPV would fall below 20% and a positive result would likely represent a false positive result if clinical suspicion is low or moderate. § Moreover, because the sensitivity is also decreased in this later population (as mentioned above), we cannot rely on a -ve result to exclude the disease if clinical/radiological criteria are present.

§ Based on these considerations, we can conclude that GM and BDG testing has a role for routine IA diagnosis in pts with hematological malignancies during the high risk period (i. e. , neutropenia or post allogeneic HSCT). § However, their value is limited in other settings, such as solid-organ transplant recipients or non-neutropenic pts, in which IA prevalence is lower and sensitivity/specificity of the tests is decreased. § Use of GM or BDG testing in these populations should be determined on the basis of individual assessment of the pre-test probability of IA according to host factors and clinical signs.

WHEN SHOULD BE PERFORMED? § Punctual testing can be performed in case of clinical suspicion of IA (e. g. , ab. N radiological finding consistent with IA) to precise the diagnosis. § A a positive GM or BDG testing would be the first hint of the disease and trigger further diagnostic work- up including CT-scan and bronchoscopy. § Alternatively, monitoring of BDG or GM can be performed in a selected population of high-risk patients (e. g. , 2 or 3 times per week among onco-hematological pts during the neutropenic phase) in the absence of clinical sn or sx for early detection of IA. § While monitoring strategies are applied in many onco-hematology wards, few studies have assessed the real benefit of this pre-emptive approach. § These monitoring strategy may be justified in high-risk patients. § Another important question is the number of positive tests required to define a positive result (i. e. , one single versus two consecutive positive tests). § As false positive results are relatively frequent and any positive result should, at least, be confirmed on a second aliquot of the sample. § Most experts recommend the repetition of the test on a subsequent serum sample, most studies addressing a significant increase of specificity (from 80%– 90% to > 97%) with the requirement of 2 consecutive positive tests in serum for the diagnosis of proven/probable IA.

HOW SHOULD BE PERFORMED? § GM testing can also be performed in samples other than serum (BAL, CSF). § The sensitivity and specificity of the test in BAL are higher than those reported in serum and they recommend testing of GM in BAL in all immunocompromised pts undergoing bronchoscopy for suspicion of pulmonary IA on the basis of CT imaging. § The cut-off is still debated, but an OD result <0. 5 virtually rules out the diagnosis, while a value > 3 has near 100% PPV. § GM is also useful in the follow-up for assessment of therapeutic response. § While radiological signs of improvement are often delayed and not evident before 2 weeks or more, a decline or increase of GM values in serum may be the first indicator of therapeutic success or failure, respectively. § They recommend weekly monitoring of GM values during antifungal therapy of IA until values <0. 5 are achieved
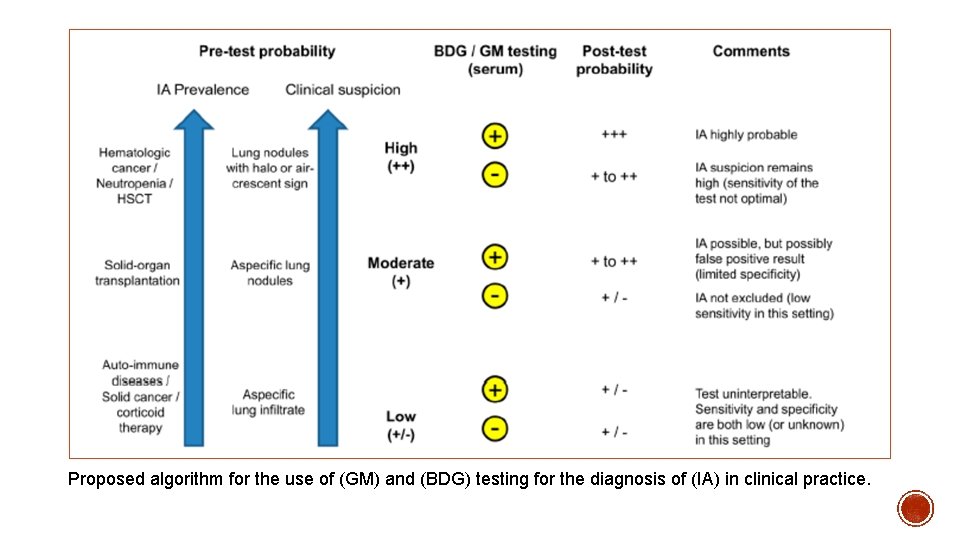
Proposed algorithm for the use of (GM) and (BDG) testing for the diagnosis of (IA) in clinical practice.
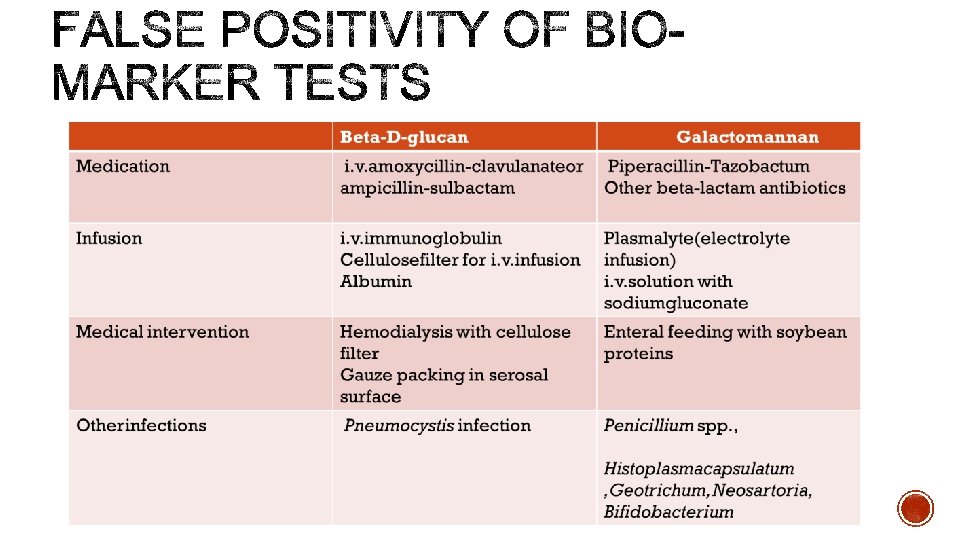

§ Measuring the serum or plasma BDG levels has a high level of accuracy in the discrimination of pt with or without IFIs, mainly dt Candida or Aspergillus. § The use of the BDG assay in combination with the GAL assay could be of great interest to clinicians who can use these assays to exclude or confirm suspected IFIs, particularly in pts with haematological malignancies. § In clinical practice, the proper use of the BDG and GAL test would require a good knowledge of its characteristics, especially with regard to the fungal pathogen-induced diseases that this test does not detect and the factors associated with a false-positive test result.

§ In high risk pts with clinical/radiological signs of possible IA (high pre-test probability), a positive GM or BDG would probably have little impact on therapeutic decisions as these patients would be treated anyway. § However, it may reinforce the diagnosis of IA (from possible to probable), help in differentiating IA from other mycoses, and serve as a baseline for follow-up. § In these high-risk patients, GM or BDG monitoring (once or twice weekly) may also be helpful for the early detection of IA in pre-emptive strategies. § Inversely, in pts with low pre-test probability, GM or BDG results may be difficult to interpret and turn to be more confusing than helpful because of low positive predictive value (high risk of false positive results) and decreased sensitivity in non-hematological populations. § There is a large “in-between” or grey zone, in which GM or BDG testing may be helpful for therapeutic decisions when interpreted in conjunction with host factors and clinical/radiological findings on an individual basis. § Finally, it should reminded that neither GM nor BDG are appropriate to detect mucormycosis, the second most frequent invasive mold infection that affects the same patients’ population as IA and has a similar clinical presentation.

§ In conclusion, GM or BDG testing should be targeted on the right population and right situation to offer the optimal benefit in the approach of IFI diagnosis and management. § Appling these assays on a non-selected pt population could cause misleading results and difficulty in interpreting both positive results and discrepancies between the two tests.

§ Tortoranoet al. J Clin. Microbiol 2012, Huang et al. AIDS 2007, Wheat et al. Clin. Vaccine Immunol 2007

§ TQ
- Slides: 41