Johann Wolfgang Dobereiner 1829 Broke elements into Triads
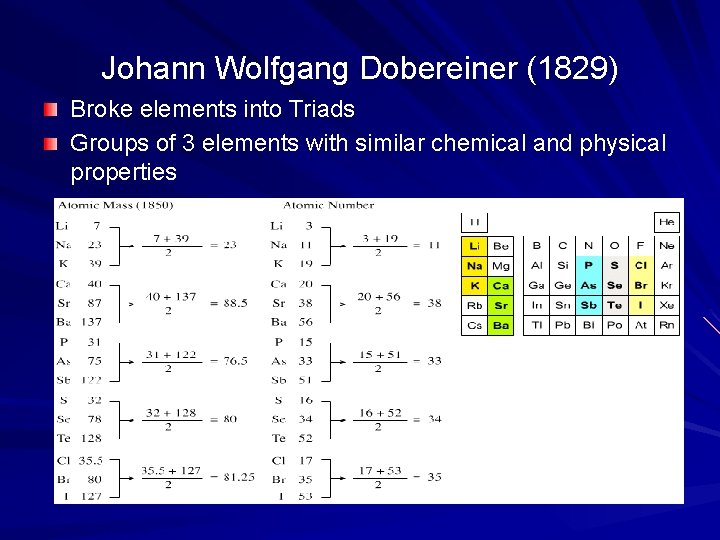
Johann Wolfgang Dobereiner (1829) Broke elements into Triads Groups of 3 elements with similar chemical and physical properties

John Newlands (1864) Law of Octaves Every eighth is a repeat of properties

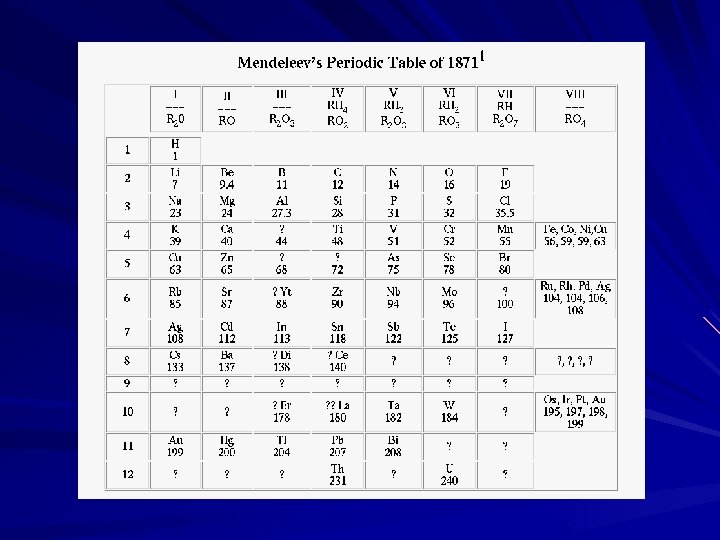
Dmitri Ivanovich Mendeleev (1869) Arranged based on atomic mass First working periodic table Predicted the existence of undiscovered elements

Henry Moseley (1913) Arranged based on atomic number Broken into metals and nonmetals Looks like periodic table of today – Just more elements now

Alkali Metals Group One (Not hydrogen) Soft metal Highest reactivity of all metals – Larger ones are more reactive – Due to having just 1 VE Form +1 ion Make one bond in most cases

Alkaline Earth Metals Group 2 Harder/stronger than alkali metals Lower reactivity than alkali, but still high – Have 2 VE Form +2 ion Make 2 bonds in most cases

Transition Metals Group 3 -12 Very strong in most cases Good conductors (heat/electricity) Pretty low reactivity (some almost non reactive) Form ions between +1 and +3 most times Will make 1 -3 bonds most times

Halogens Group 17 Most reactive of the nonmetals – Have 7 VE (1 short of being full) Exist as diatomic molecule Form -1 ion Form 1 bond

Inert / Noble Gases Group 18 Non reactive (larger ones can form bonds) Full valence shell – 8 for all except He No charge most times No bonds most times

Lanthanide f-block in period 6 2 VE

Actinide f-block period 7 2 VE Highly radioactive Many are synthetic Grouped with lanthanide series as rare earth metals

Metalloids Found on the separation between metals and nonmetals Metallic and non metallic properties
- Slides: 13