JJ Thomsons Plum Pudding Model 8 December 1856

JJ Thomson’s “Plum Pudding” Model 8 December 1856 – 30 August 1940 http: //2011 period 6 group 4. wikispaces. com/2. +Thomson's+Model
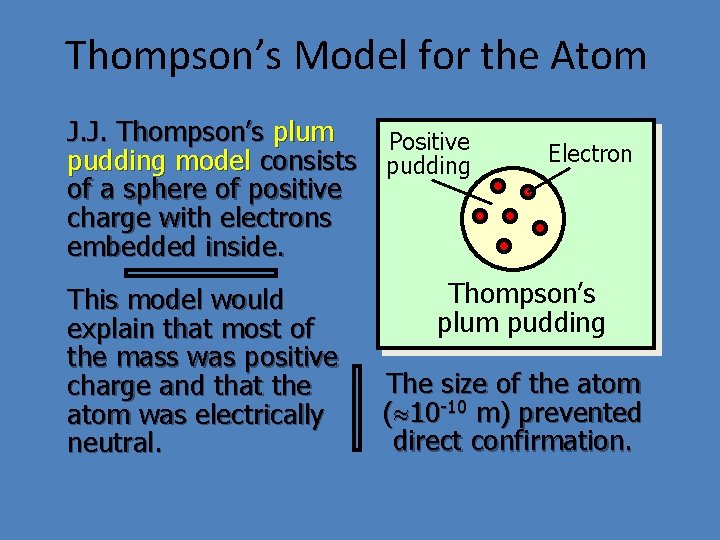
Thompson’s Model for the Atom J. J. Thompson’s plum pudding model consists of a sphere of positive charge with electrons embedded inside. This model would explain that most of the mass was positive charge and that the atom was electrically neutral. Positive pudding Electron Thompson’s plum pudding The size of the atom ( 10 -10 m) prevented direct confirmation.
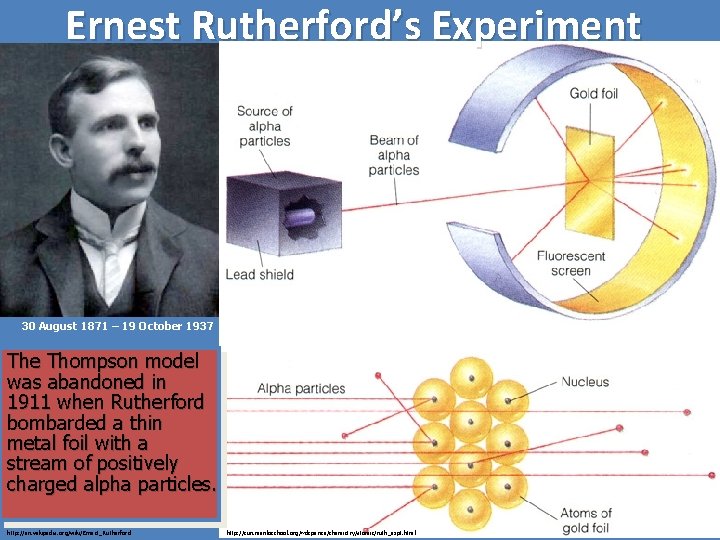
Ernest Rutherford’s Experiment 30 August 1871 – 19 October 1937 The Thompson model was abandoned in 1911 when Rutherford bombarded a thin metal foil with a stream of positively charged alpha particles. http: //en. wikipedia. org/wiki/Ernest_Rutherford http: //sun. menloschool. org/~dspence/chemistry/atomic/ruth_expt. html
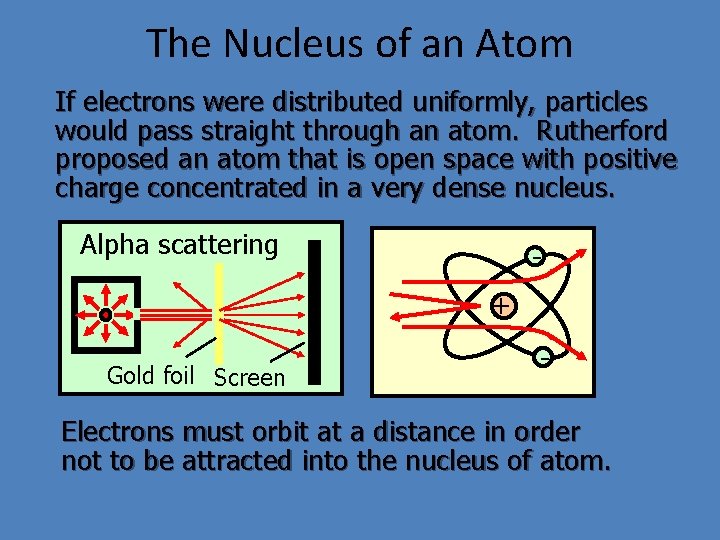
The Nucleus of an Atom If electrons were distributed uniformly, particles would pass straight through an atom. Rutherford proposed an atom that is open space with positive charge concentrated in a very dense nucleus. Alpha scattering + Gold foil Screen - Electrons must orbit at a distance in order not to be attracted into the nucleus of atom.
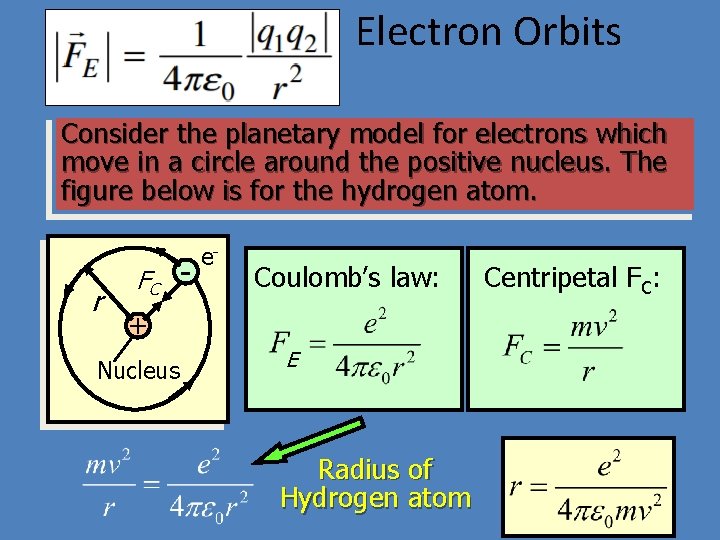
Electron Orbits Consider the planetary model for electrons which move in a circle around the positive nucleus. The figure below is for the hydrogen atom. r FC - e- Coulomb’s law: + Nucleus E Radius of Hydrogen atom Centripetal FC:
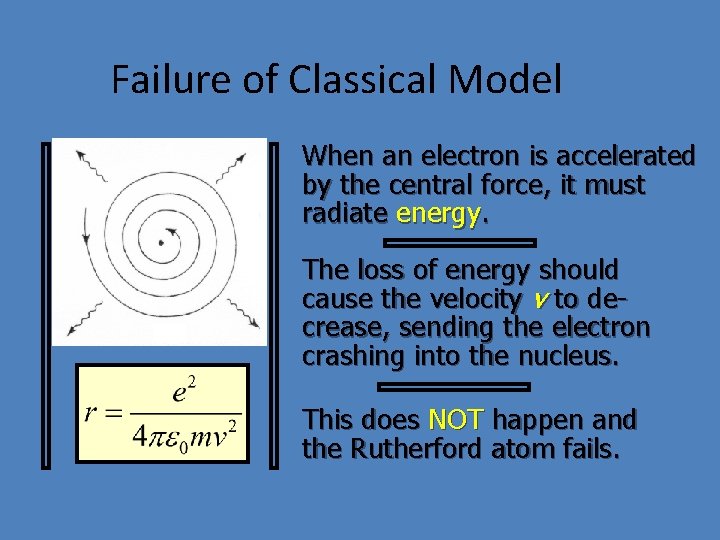
Failure of Classical Model v + Nucleus - e- When an electron is accelerated by the central force, it must radiate energy. The loss of energy should cause the velocity v to decrease, sending the electron crashing into the nucleus. This does NOT happen and the Rutherford atom fails.

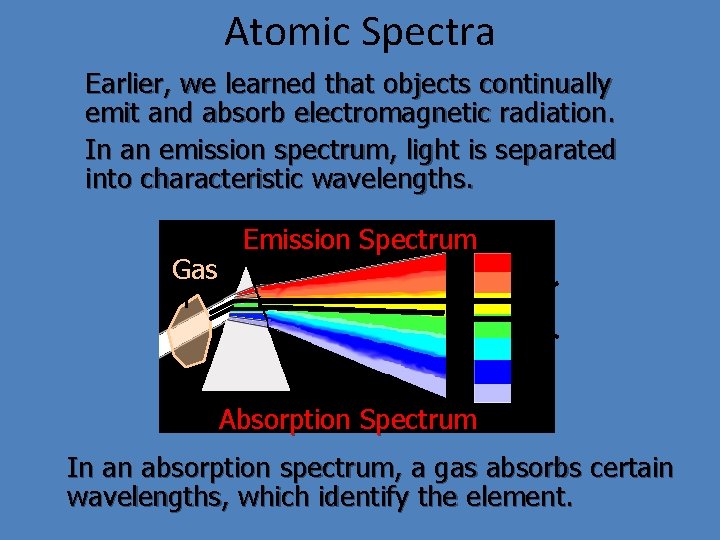
Atomic Spectra Earlier, we learned that objects continually emit and absorb electromagnetic radiation. In an emission spectrum, light is separated into characteristic wavelengths. Gas Emission Spectrum l 1 l 2 Absorption Spectrum In an absorption spectrum, a gas absorbs certain wavelengths, which identify the element.
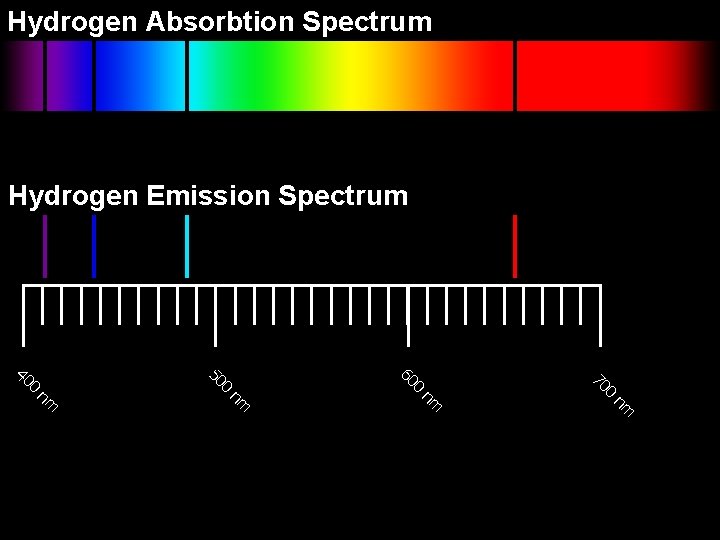
Hydrogen Absorbtion Spectrum Hydrogen Emission Spectrum nm nm 0 70 0 60 0 50 0 40

Sodium Mercury Neon http: //annwn. tempwebpage. com/cookpot/bits/neon. jpg

Bromine Deuterium Helium Hydrogen Krypton Mercury Neon Water Vapor Xenon http: //www. astrographics. com/Gallery. Prints. Index/GP 4259. html Image and text copyright © Ted Kinsman.
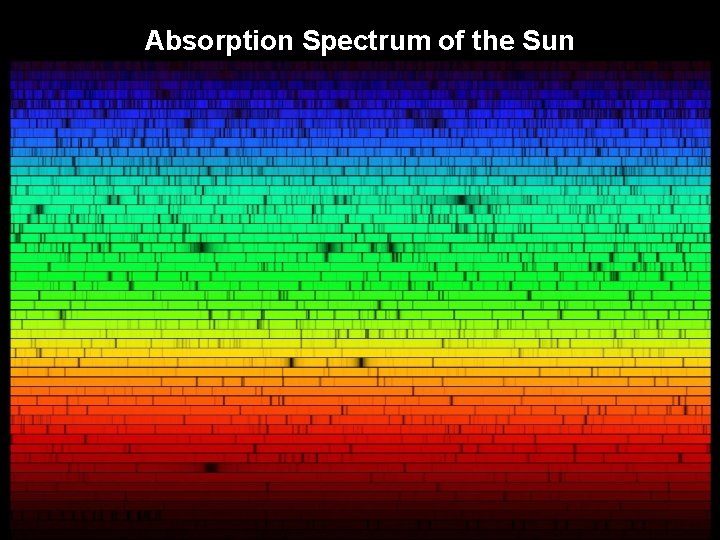
Absorption Spectrum of the Sun
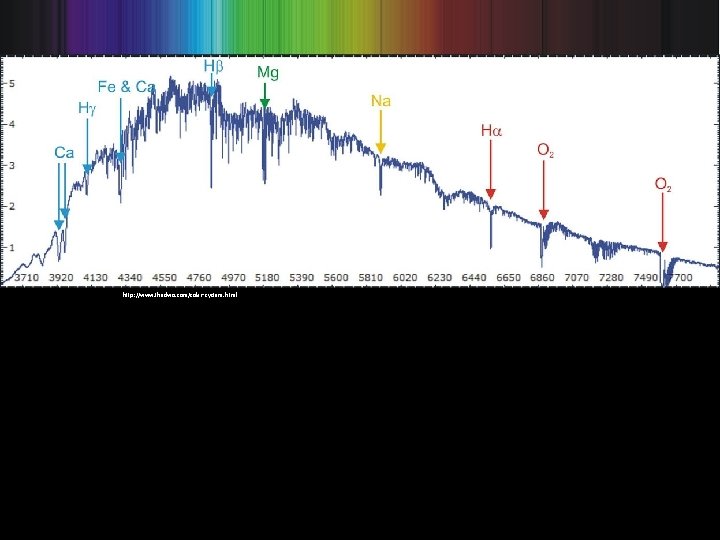
http: //www. thedwo. com/solar-system. html

1 2 3 4 7 8 15 16 31 32 63 64 127 128 255 256 Moves = 2(number of discs) - 1

Johann Jakob Balmer May 1, 1825 – March 12, 1898

Emission Spectrum for H Atom Characteristic wavelengths n n = = 6 5 4 3 0 41 4 43 6 3 48 65 nm nm Balmer worked out a mathematical formula, called the Balmer series for predicting the absorbed wavelengths from hydrogen gas. - R 1. 097 x 107 m-1

Example 1: Use the Balmer equation to find the wavelength of the first line (n = 3) in the Balmer series. How can you find the energy? - R = 1. 097 x 107 m-1 l = 656 nm The frequency and the energy are found from: c = fl and E = hf
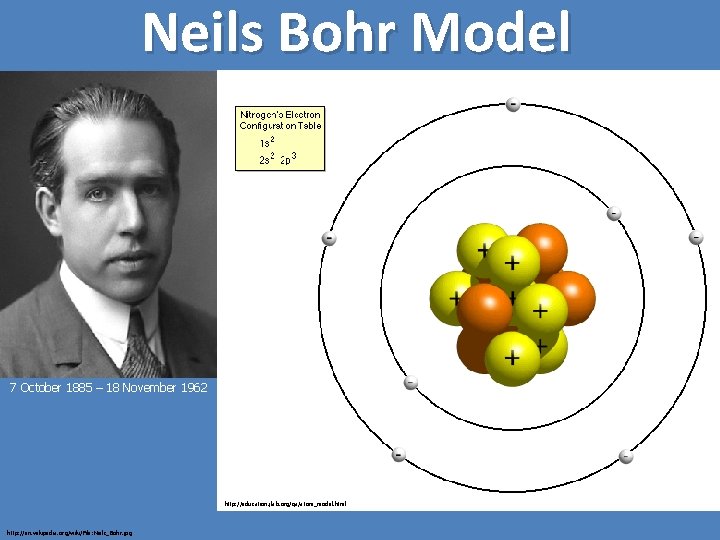
Neils Bohr Model 7 October 1885 – 18 November 1962 http: //education. jlab. org/qa/atom_model. html http: //en. wikipedia. org/wiki/File: Niels_Bohr. jpg

Bohr's starting point was to realize that classical mechanics by itself could never explain the atom's stability. A stable atom has a certain size so that any equation describing it must contain some fundamental constant or combination of constants with a dimension of length. The classical fundamental constants--namely, the charges and the masses of the electron and the nucleus--cannot be combined to make a length. Bohr noticed, however, that the quantum constant formulated by the German physicist Max Planck has dimensions which, when combined with the mass and charge of the electron, produce a measure of length. Numerically, the measure is close to the known size of atoms. This encouraged Bohr to use Planck's constant in searching for a theory of the atom
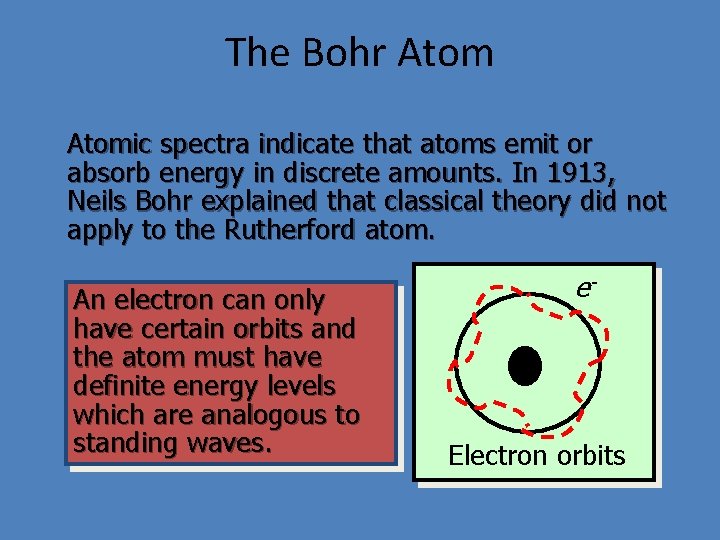
The Bohr Atomic spectra indicate that atoms emit or absorb energy in discrete amounts. In 1913, Neils Bohr explained that classical theory did not apply to the Rutherford atom. An electron can only have certain orbits and the atom must have definite energy levels which are analogous to standing waves. e+ Electron orbits
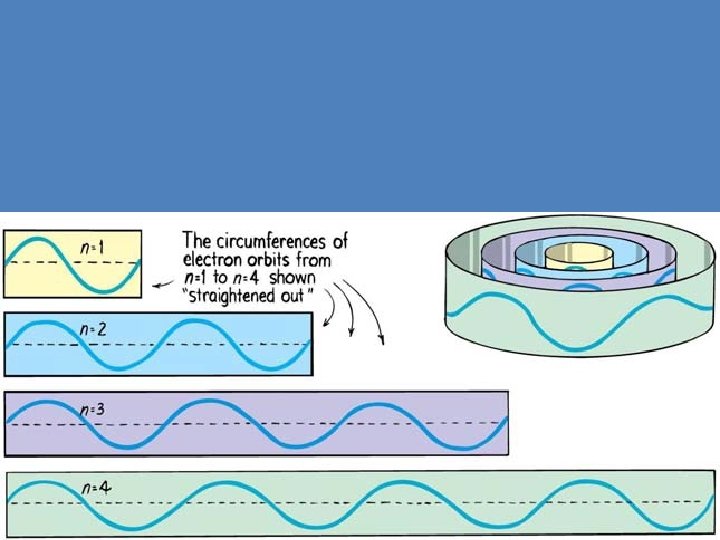
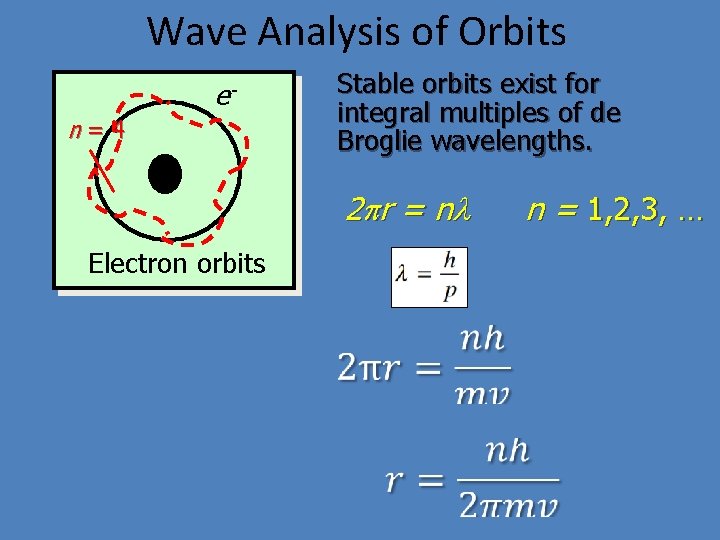
Wave Analysis of Orbits en=4 + Electron orbits Stable orbits exist for integral multiples of de Broglie wavelengths. 2 pr = n l n = 1, 2, 3, …
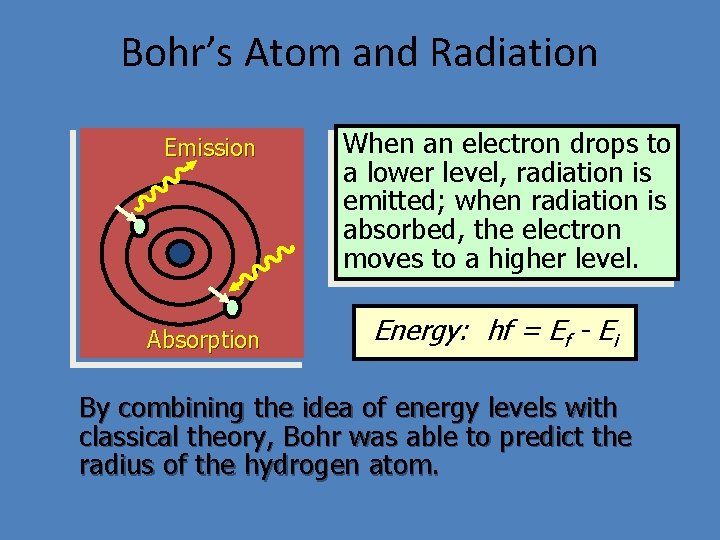
Bohr’s Atom and Radiation Emission Absorption When an electron drops to a lower level, radiation is emitted; when radiation is absorbed, the electron moves to a higher level. Energy: hf = Ef - Ei By combining the idea of energy levels with classical theory, Bohr was able to predict the radius of the hydrogen atom.

Radius of the Hydrogen Atom Radius as function of energy level: Bohr’s radius Classical radius By eliminating r from these equations, we find the velocity v; elimination of v gives possible radii rn:

Example 2: Find the radius of the Hydrogen atom in its most stable state (n = 1). m = 9. 1 x 10 -31 kg e = 1. 6 x 10 -19 C r = 5. 31 x 10 -11 m r = 53. 1 pm

Total Energy of an Atom The total energy at level n is the sum of the kinetic and potential energies at that level. But we recall that: Total energy of Hydrogen atom for level n. Substitution for v and r gives expression for total energy.

Energy for a Particular State It will be useful to simplify the energy formula for a particular state by substitution of constants. m = 9. 1 x 10 -31 e = 1. 6 x 10 -19 kg eo = 8. 85 x 10 --12 C 2/Nm 2 C h = 6. 63 x 10 -34 J s Or
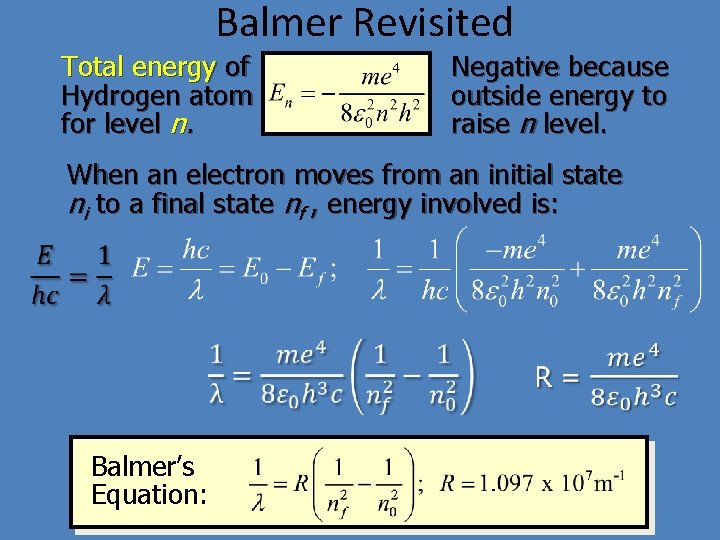
Balmer Revisited Total energy of Hydrogen atom for level n. Negative because outside energy to raise n level. When an electron moves from an initial state ni to a final state nf , energy involved is: Balmer’s Equation:
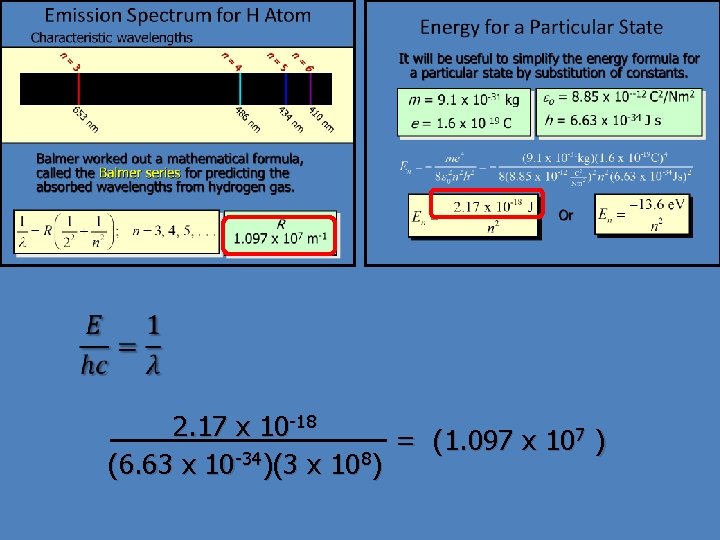
2. 17 x 10 -18 = (1. 097 x 107 ) (6. 63 x 10 -34)(3 x 108)

Energy Levels We can now visualize the hydrogen atom with an electron at many possible energy levels. Emission Absorption The energy of the atom increases on absorption (nf > ni) and decreases on emission (nf < ni). Energy of nth level: The change in energy of the atom can be given in terms of initial ni and final nf levels:

Fg = G m 1·m 2 r. A 2 Work = average force · distance m 1·m 2 = G r r · (r. B– r. A) A B r. A = G m 1·m 2 r. B r Ar B = G·m 1·m 2 1 r. A zero Fg = G r. B m 1·m 2 r. B 2 = G·m 1·m 2 Absolute Potential Energy 1 ∞ r. A r Ar B 1 r. B UG = - G·m 1·m 2 r. B Negative because this is how much energy the object lost coming from ∞ to rb.
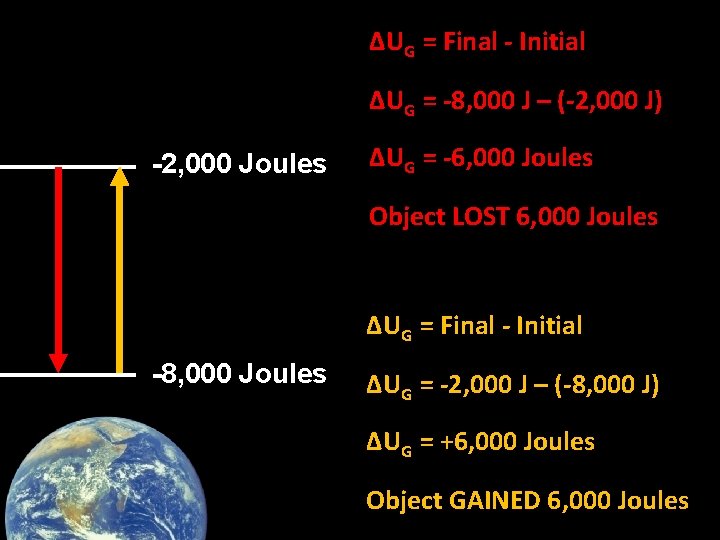
ΔUG = Final - Initial ΔUG = -8, 000 J – (-2, 000 J) -2, 000 Joules ΔUG = -6, 000 Joules Object LOST 6, 000 Joules ΔUG = Final - Initial -8, 000 Joules ΔUG = -2, 000 J – (-8, 000 J) ΔUG = +6, 000 Joules Object GAINED 6, 000 Joules
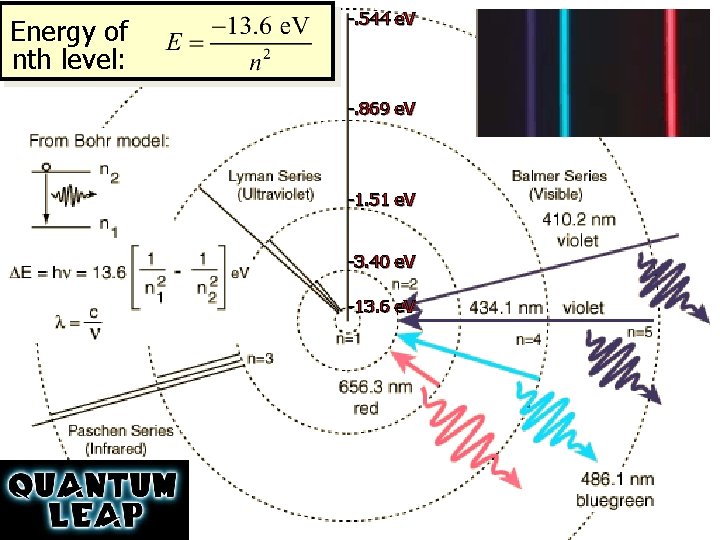
Energy of nth level: -. 544 e. V -. 869 e. V -1. 51 e. V -3. 40 e. V -13. 6 e. V http: //hyperphysics. phy-astr. gsu. edu/hbase/hyde. html
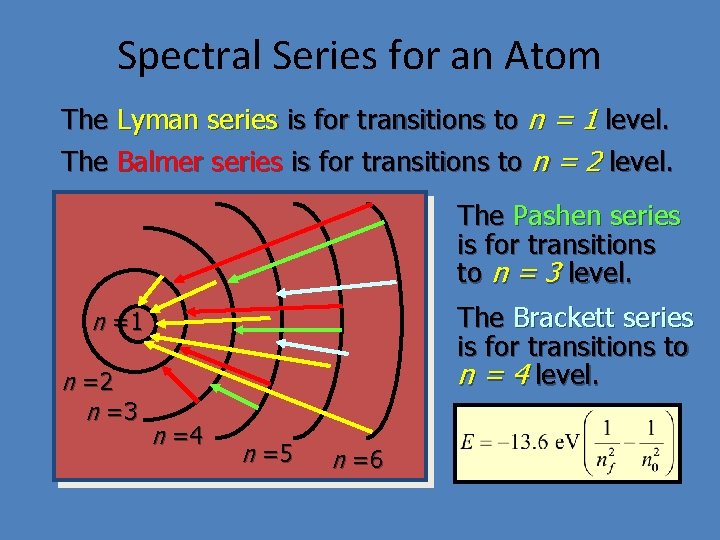
Spectral Series for an Atom The Lyman series is for transitions to n = 1 level. The Balmer series is for transitions to n = 2 level. The Pashen series is for transitions to n = 3 level. The Brackett series is for transitions to n = 4 level. n =1 n =2 n =3 n =4 n =5 n =6

Example 3: What is the energy of an emitted photon if an electron drops from the n = 3 level to the n = 1 level for the hydrogen atom? Change in energy of the atom. DE = -12. 1 e. V The energy of the atom decreases by 12. 1 e. V as a photon of that energy is emitted. You should show that 13. 6 e. V is required to move an electron from n = 1 to n = .

Modern Theory of the Atom The model of an electron as a point particle moving in a circular orbit has undergone significant change. • The quantum model now presents the location of an electron as a probability distribution - a cloud around the nucleus. • Additional quantum numbers have been added to describe such things as shape, orientation, and magnetic spin. • Pauli’s exclusion principle showed that no two electrons in an atom can exist in the exact same state.
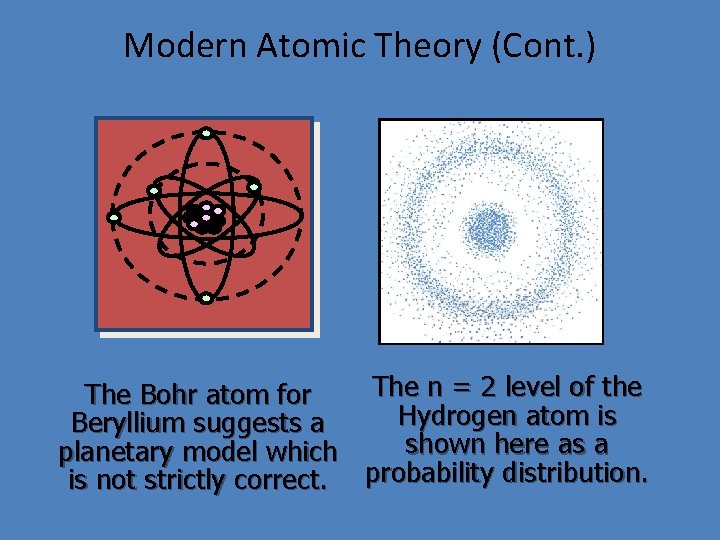
Modern Atomic Theory (Cont. ) The n = 2 level of the The Bohr atom for Hydrogen atom is Beryllium suggests a shown here as a planetary model which is not strictly correct. probability distribution.
- Slides: 40