Jessica Merrill and Eleanor Malone A Scrip Webinar

Jessica Merrill and Eleanor Malone A Scrip Webinar presented by biosimilar challenge How originator companies face up to the Battlegrounds Biosimilar

Logistics • If you have a question during the webinar enter it into the chat box below and click SUBMIT. We will respond at the end of the presentation. • A recording of this webinar and the slide deck will be sent to you. 2 Pharma intelligence | informa

About the Presenters Eleanor Malone eleanor. malone@informa. com | @Scrip. Eleanor Editor in Chief of Informa Pharma Insights Europe, overseeing Scrip, Pink Sheet, In Vivo and Generics Bulletin Jessica Merrill jessica. merrill@informa. com | @Jessicaemerrill US-based Senior Editor for Scrip, Pink Sheet, In Vivo 3 Pharma intelligence | informa

Overview • Originator firms on the front line of biosimilar competition • Key biologic brands facing competition • Extent of exposure for key firms • Defense tactics • US biosimilars landscape • European biosimilars landscape • Q&A 4 Pharma intelligence | informa

Those On The Front Line Key Originator Companies with blockbuster biologicals whose market exclusivity has expired or will soon expire 5 Pharma intelligence | informa
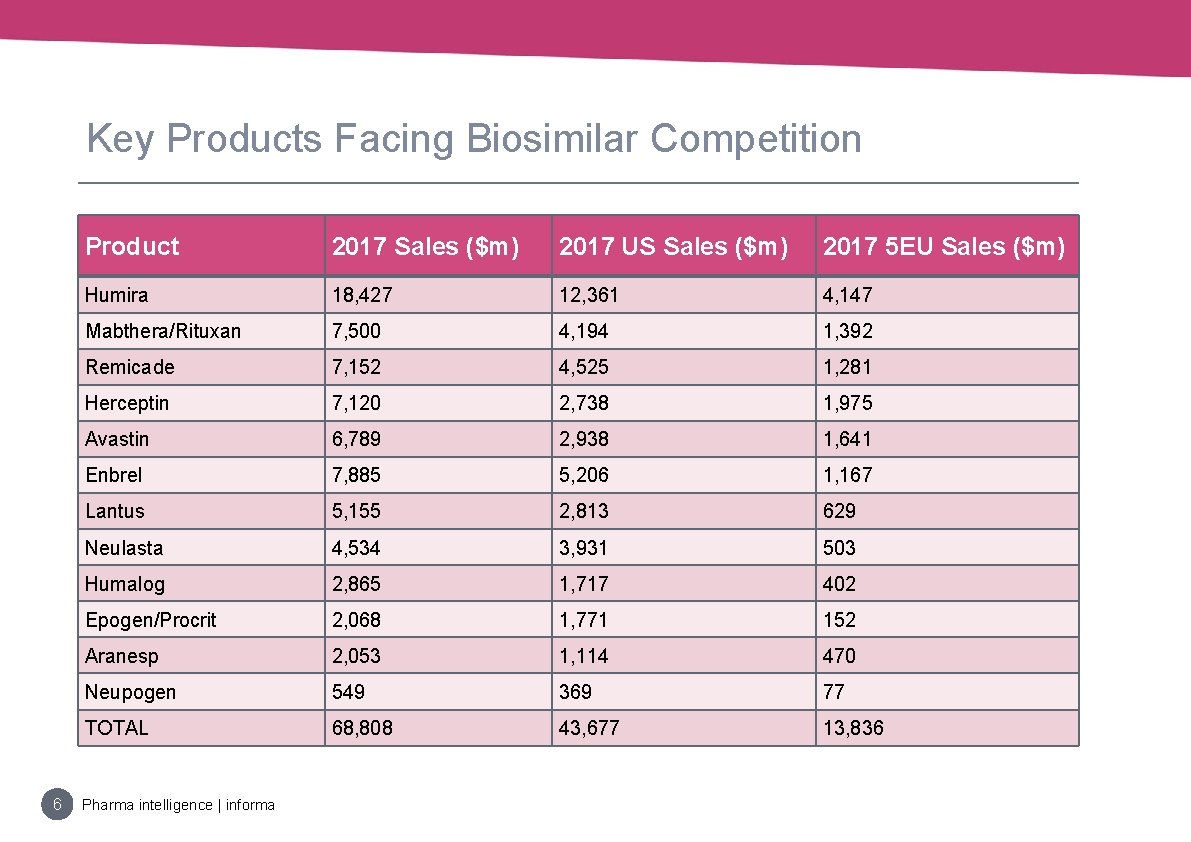
Key Products Facing Biosimilar Competition 6 Product 2017 Sales ($m) 2017 US Sales ($m) 2017 5 EU Sales ($m) Humira 18, 427 12, 361 4, 147 Mabthera/Rituxan 7, 500 4, 194 1, 392 Remicade 7, 152 4, 525 1, 281 Herceptin 7, 120 2, 738 1, 975 Avastin 6, 789 2, 938 1, 641 Enbrel 7, 885 5, 206 1, 167 Lantus 5, 155 2, 813 629 Neulasta 4, 534 3, 931 503 Humalog 2, 865 1, 717 402 Epogen/Procrit 2, 068 1, 771 152 Aranesp 2, 053 1, 114 470 Neupogen 549 369 77 TOTAL 68, 808 43, 677 13, 836 Pharma intelligence | informa

Extent of exposure for key firms • Amgen • Johnson & Johnson • Abb. Vie • Roche 7 Pharma intelligence | informa
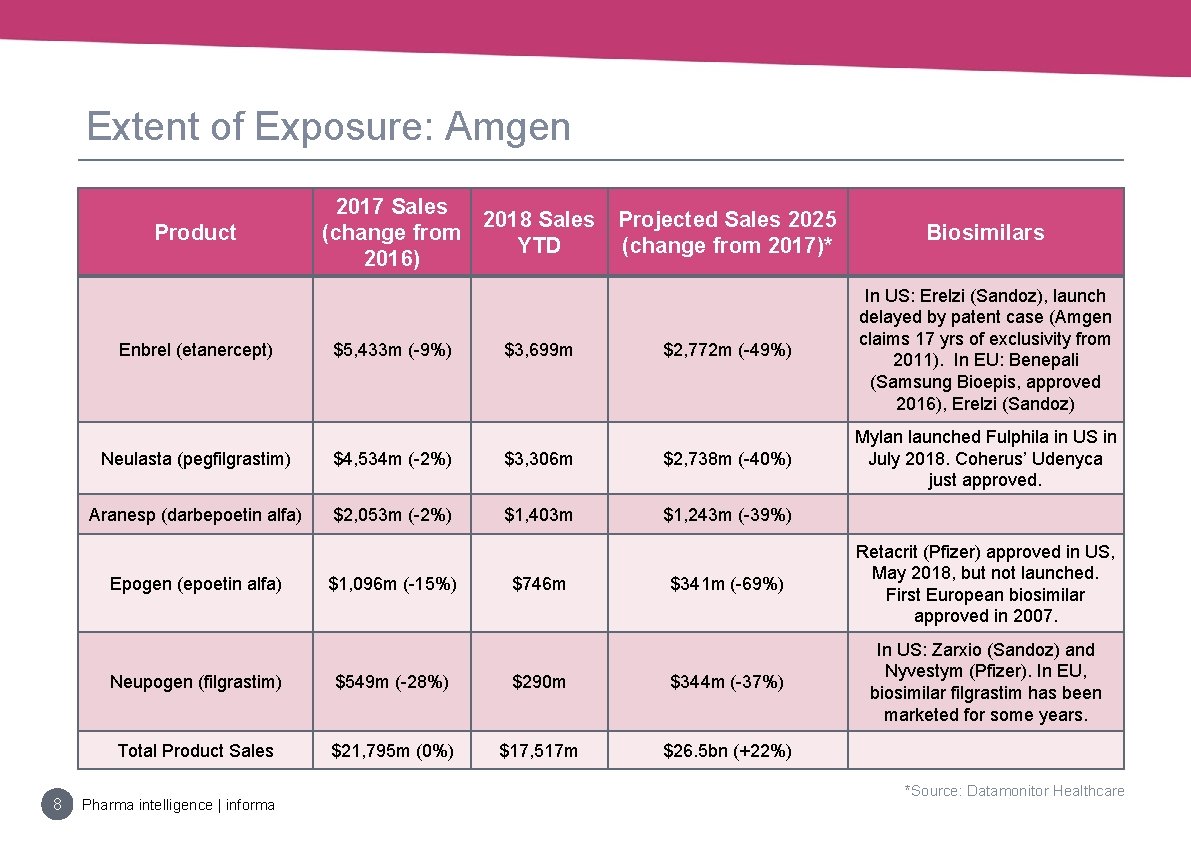
Extent of Exposure: Amgen Product Enbrel (etanercept) $5, 433 m (-9%) $3, 699 m Projected Sales 2025 (change from 2017)* Biosimilars $2, 772 m (-49%) In US: Erelzi (Sandoz), launch delayed by patent case (Amgen claims 17 yrs of exclusivity from 2011). In EU: Benepali (Samsung Bioepis, approved 2016), Erelzi (Sandoz) Mylan launched Fulphila in US in July 2018. Coherus’ Udenyca just approved. Neulasta (pegfilgrastim) $4, 534 m (-2%) $3, 306 m $2, 738 m (-40%) Aranesp (darbepoetin alfa) $2, 053 m (-2%) $1, 403 m $1, 243 m (-39%) Epogen (epoetin alfa) 8 2017 Sales 2018 Sales (change from YTD 2016) $1, 096 m (-15%) $746 m $341 m (-69%) Retacrit (Pfizer) approved in US, May 2018, but not launched. First European biosimilar approved in 2007. In US: Zarxio (Sandoz) and Nyvestym (Pfizer). In EU, biosimilar filgrastim has been marketed for some years. Neupogen (filgrastim) $549 m (-28%) $290 m $344 m (-37%) Total Product Sales $21, 795 m (0%) $17, 517 m $26. 5 bn (+22%) Pharma intelligence | informa *Source: Datamonitor Healthcare

Extent Of Exposure: Amgen • Enbrel, Neulasta, Neupogen, Epogen are all facing biosimilar competition • Amgen is hedging by building its own portfolio of biosimilars, which is expected to be a key growth driver for the company in the coming years • Defending its brands with price concessions and where possible, lifecycle management strategies, like Neulasta Onpro. 9 Pharma intelligence | informa
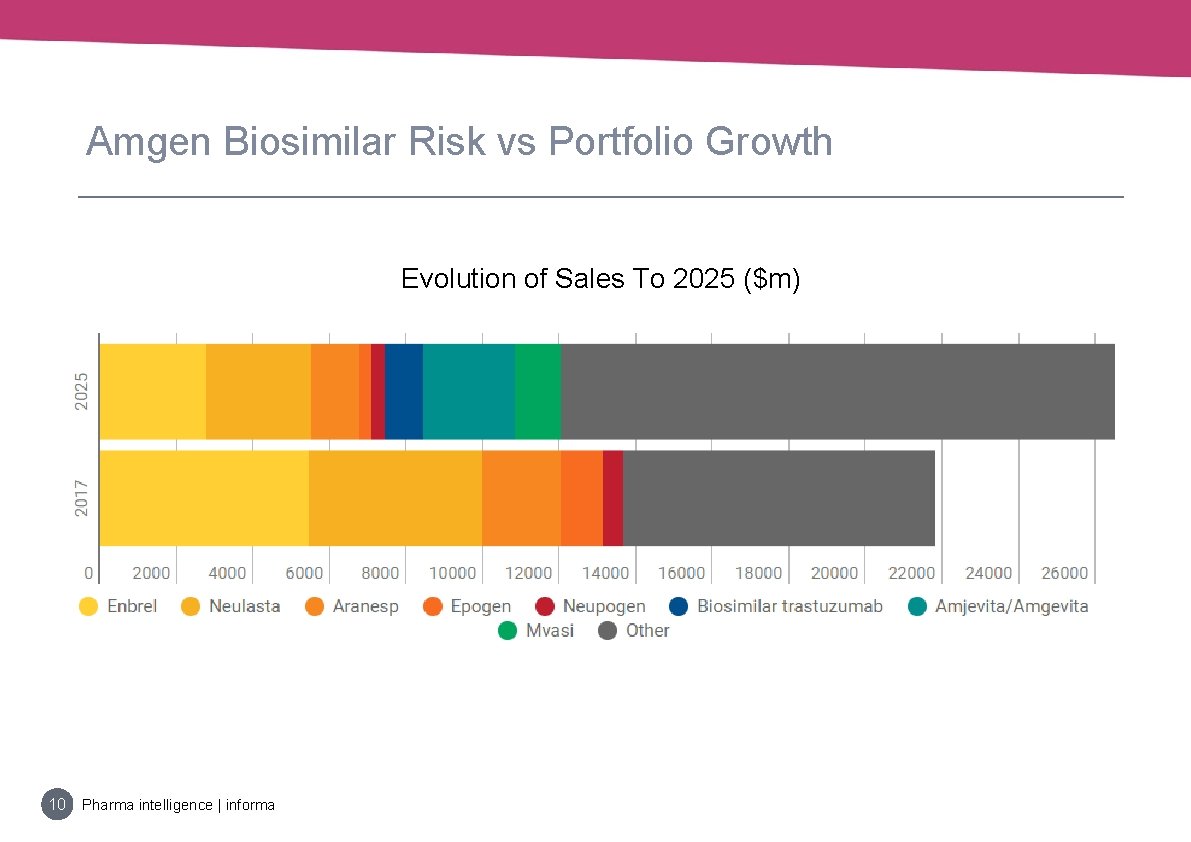
Amgen Biosimilar Risk vs Portfolio Growth Evolution of Sales To 2025 ($m) 10 Pharma intelligence | informa
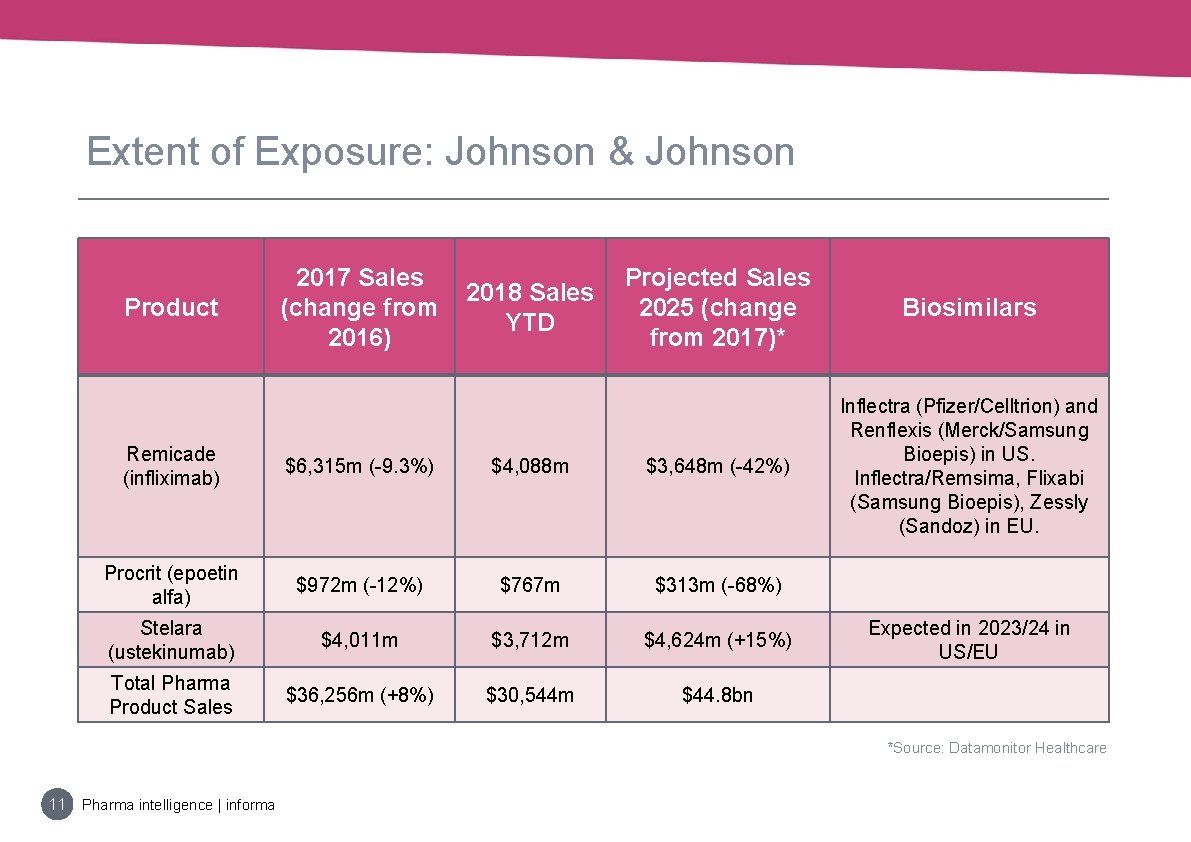
Extent of Exposure: Johnson & Johnson Product 2017 Sales (change from 2016) 2018 Sales YTD Projected Sales 2025 (change from 2017)* Biosimilars Inflectra (Pfizer/Celltrion) and Renflexis (Merck/Samsung Bioepis) in US. Inflectra/Remsima, Flixabi (Samsung Bioepis), Zessly (Sandoz) in EU. Remicade (infliximab) $6, 315 m (-9. 3%) $4, 088 m $3, 648 m (-42%) Procrit (epoetin alfa) $972 m (-12%) $767 m $313 m (-68%) Stelara (ustekinumab) $4, 011 m $3, 712 m $4, 624 m (+15%) Total Pharma Product Sales $36, 256 m (+8%) $30, 544 m $44. 8 bn Expected in 2023/24 in US/EU *Source: Datamonitor Healthcare 11 Pharma intelligence | informa
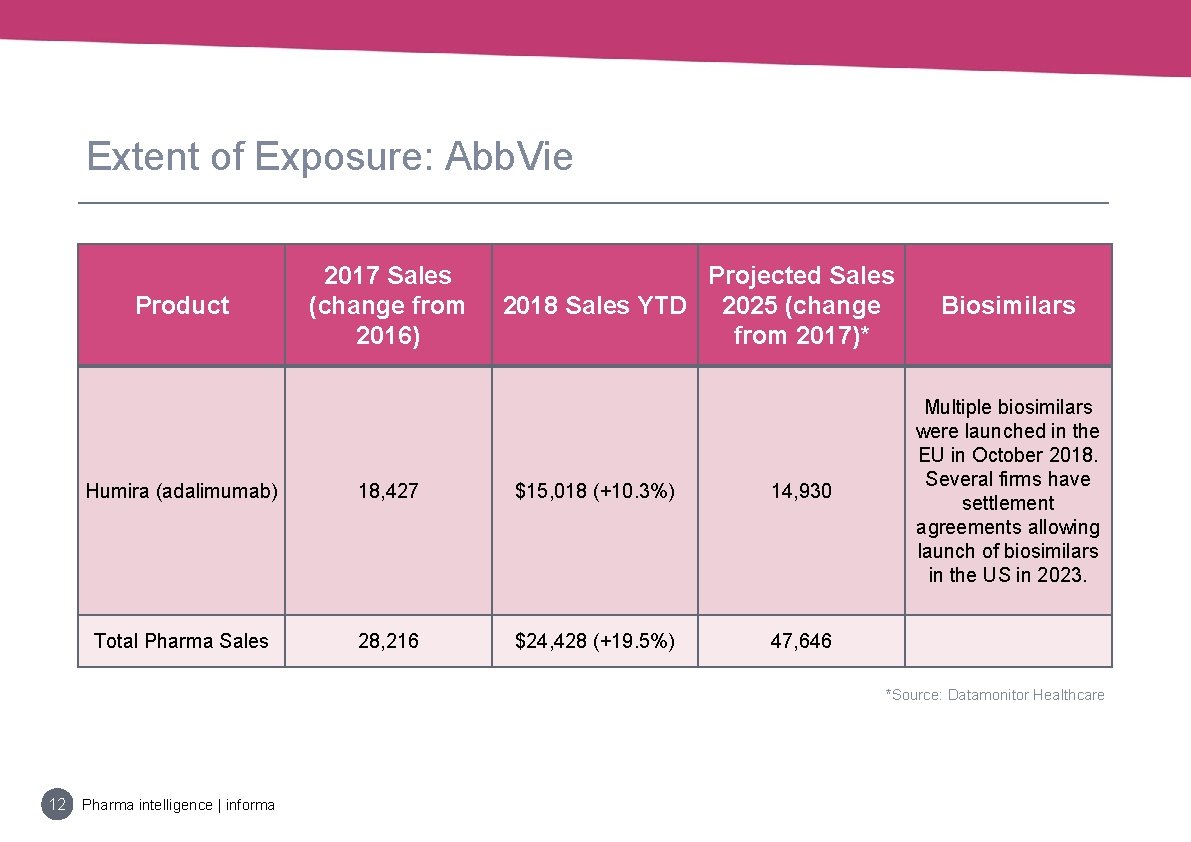
Extent of Exposure: Abb. Vie Product 2017 Sales (change from 2016) Projected Sales 2018 Sales YTD 2025 (change from 2017)* Humira (adalimumab) 18, 427 $15, 018 (+10. 3%) 14, 930 Total Pharma Sales 28, 216 $24, 428 (+19. 5%) 47, 646 Biosimilars Multiple biosimilars were launched in the EU in October 2018. Several firms have settlement agreements allowing launch of biosimilars in the US in 2023. *Source: Datamonitor Healthcare 12 Pharma intelligence | informa
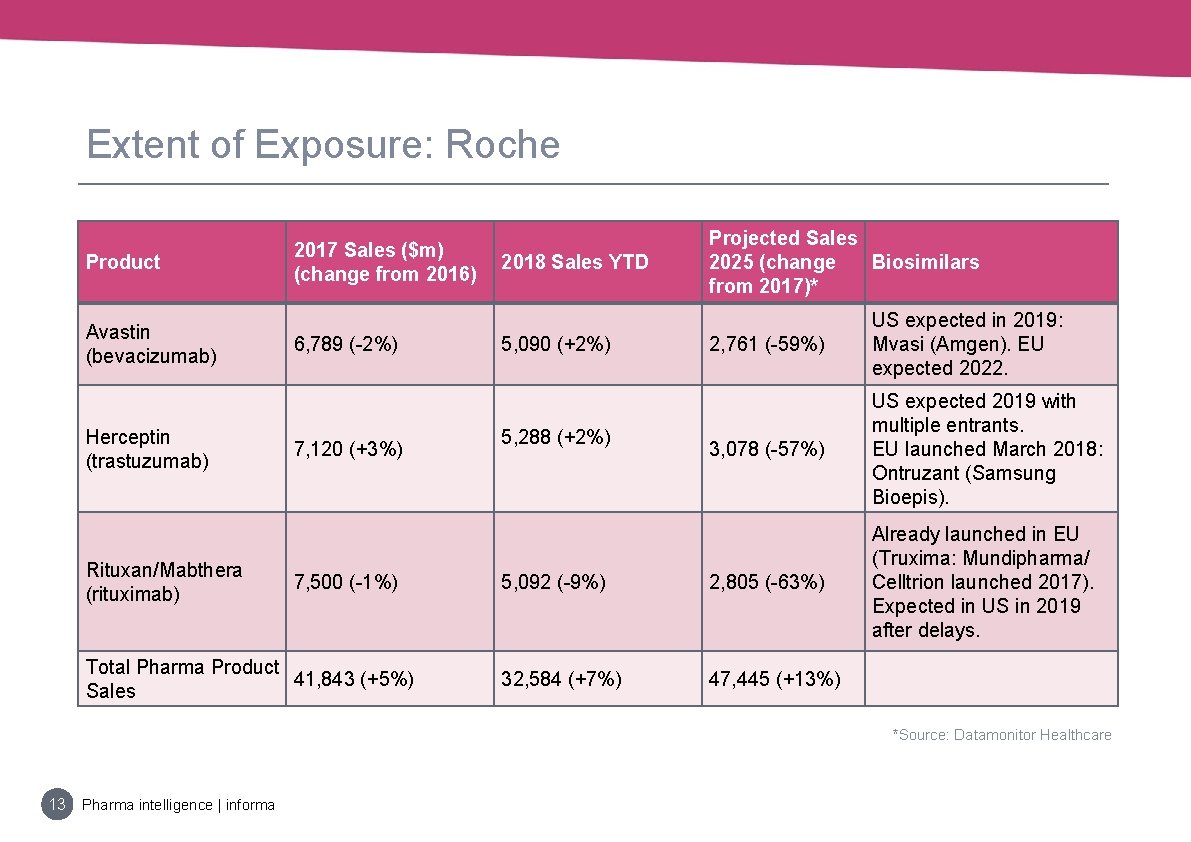
Extent of Exposure: Roche Product Avastin (bevacizumab) Herceptin (trastuzumab) Rituxan/Mabthera (rituximab) 2017 Sales ($m) (change from 2016) 6, 789 (-2%) 7, 120 (+3%) 7, 500 (-1%) Total Pharma Product 41, 843 (+5%) Sales 2018 Sales YTD Projected Sales 2025 (change Biosimilars from 2017)* 2, 761 (-59%) US expected in 2019: Mvasi (Amgen). EU expected 2022. 3, 078 (-57%) US expected 2019 with multiple entrants. EU launched March 2018: Ontruzant (Samsung Bioepis). 5, 092 (-9%) 2, 805 (-63%) Already launched in EU (Truxima: Mundipharma/ Celltrion launched 2017). Expected in US in 2019 after delays. 32, 584 (+7%) 47, 445 (+13%) 5, 090 (+2%) 5, 288 (+2%) *Source: Datamonitor Healthcare 13 Pharma intelligence | informa

Defense Tactics What can originator firms do to protect their top and bottom lines? 14 Pharma intelligence | informa

The Patent Thicket

Defense Tactics - Bundling, Rebates 16 Pharma intelligence | informa

Image source: Amgen Neulasta Onpro promotional website Defense Tactics: Life Cycle Management

Defense Tactics If you can’t beat ‘em, join ‘em – part 1 18 Pharma intelligence | informa

Defense Tactics If you can’t beat ‘em, join ‘em - part 2: * When they go low, you go lower 19 Pharma intelligence | informa

Defense Tactics: Weaponize Patients • DTC campaigns in US • Europe less susceptible as no DTC advertising 20 Pharma intelligence | informa

Defense Tactics- Innovate • Optimise your R&D pipeline to replace expiring products • Next-generation product development • New technologies • Partnerships 21 Pharma intelligence | informa

Defense Tactics Round-up • Grow a Patent Thicket • Tie in customers with discounts, bundles, rebates • Life cycle management • Make/sell biosimilars • Compete on price • Target patients • INNOVATE!! 22 Pharma intelligence | informa

US and Europe 23 Pharma intelligence | informa

US and Europe: Two Very Different Biosimilar Markets Europe US Around 50 biosimilars launched for drugs including: Sandoz Zarxio (filgrastim)/ Amgen's Neupogen Sanofi's Lantus (insulin glargine) Amgen's Epogen (epoetin alfa) Amgen/Pfizer's Enbrel (etanercept) Amgen's Neupogen (filgrastim) Amgen's Neulasta (pegfilgrastim) Johnson & Johnson/Merck's Remicade (infliximab) Roche's Rituxan (rituximab) Roche's Herceptin (trastuzumab) Abb. Vie's Humira (adalimumab) Pfizer Inflectra (infliximab)/ J&J's Remicade Merck Renflexis (infliximab)/ J&J's Remicade Mylan Fulphila(pegfilgrastim)/ Amgen's Neulasta Pfizer Nyvestym (filgrastim)/ Amgen's Neupogen ~50 biosimilars launched in Europe and 5 launched in the US 24 Pharma intelligence | informa

US Biosimilar Landscape 25 Pharma intelligence | informa

US and Europe: Two Very Different Biosimilar Markets Payer Strategy US Payers Expect To See Biosimilars Usurp Biologics Pharmacy Benefit Managers Able & Prepared To Switch Patients to Biosimilars ”While they may not be interchangeable, biosimilars will be delivered to patients who today are on the branded product” Timothy C Wentworth, CEO, Express Scripts Holding Co 2017 JP Morgan Healthcare Conference 26 Pharma intelligence | informa ”Biosimilars are being included as a component of our 2017 standard formulary strategy replacing higher-costs drugs” David M Denton, CFO & EVP, CVS Health Corp Company Analyst Day, 15 December, 2016

In The US, Mixed Biosimilar Success In the US, biosimilar uptake has remained limited, but varies by category and is impacted by category-specific issues and general challenges like multiple payers, rebates, lack of interchangeability and physician/patient education. Branded Remicade holds onto 93% share of infliximab market two years post biosimilar entry -chronic medication, which leaves biosimilar market to new scripts -J&J defensive contracting has blocked biosimilar infliximab from the market -Pfizer lawsuit against J&J could impact contracting for biosimilars Branded Neupogen only holds 37% share of filgrastim market three years post biosimilar entry -acute treatment, so a steady flow of new patients -administered in a cancer care setting in which patients often don’t know what brand they are receiving 27 Pharma intelligence | informa

Insulin: A category in need of cheaper options • Sanofi’s Lantus (insulin glargine) has been facing competition in the US from Eli Lilly/Boehringer’s Basaglar since 2015. • Sales of Lantus fell 19% in the first 9 months of 2018 to € 2. 7 bn, with some of the decline offset by Sanofi’s newer glargine formula Toujeo. • In the US, Basaglar has performed well, though it was not approved as a biosimilar under US regulatory guidelines, but rather a complex small molecule. Basaglar generated $569 m in the first nine months. • But the market could be restricted to just two products for now. Merck said it won’t launch its insulin glargine version, Lusdana. 28 Pharma intelligence | informa

Is Enthusiasm For Biosimilars Diminishing in the US? Lackluster uptake of some early biosimilars is raising concerns drug developers might hit the pause on biosimilar development if momentum doesn't pick up. -Mylan, early to biosimilars development, announced it is exploring strategic alternatives in August. Slow uptake of complex generics like Copaxone and biosimilars has been one challenge. -Momenta said it is exiting biosimilar development to focus on novel drug development in October, pointing to the slow path to value creation and uncertainty in the market. -Merck, in October, said it will not commercialize biosimilar version of Sanofi's insulin Lantus, after market assessment. Merck said it remains committed to biosimilars, however. -Sandoz has abandoned its rituximab biosimilar in the US after the FDA called for another pivotal study. -CRLS for biosimilars approved in Europe and elsewhere. 29 Pharma intelligence | informa
![Payers Sound the Alarm in US • "If [the US market is] not able Payers Sound the Alarm in US • "If [the US market is] not able](http://slidetodoc.com/presentation_image/12808eb5d7f5db1bcad78bd08e56ceec/image-30.jpg)
Payers Sound the Alarm in US • "If [the US market is] not able to produce movement … to the biosimilars and generate a return for those manufacturers that have gone down this biosimilar pathway and invested in those molecules I think they will just realize over time that they have to close up shop and move on. America would have missed a very significant opportunity. " • Kaiser Permanente's national pharmaceutical contracting leader Ambrose Carrejo, 2017 30 Pharma intelligence | informa

Biosimilar Market: commentary on outlook "Do they want to continue to benefit from monopoly rents today, or help generate a vibrant biosimilar market that can help reset biologic pricing – and drug pricing more generally – through competition. These are binary choices. You can't have your cake – or in this case, your rebates – and a vibrant market for biosimilar competition too. " Scott Gottlieb speaking to US insurers at America's Health Insurance Plans (AHIP) National Health Policy Conference, March 2018

Signs of Life “Some of our competitors and public officials have made statements that the US biosimilar market is not working. We have a different point of view … The fact that a biosimilar now holds the majority share of [filgrastim] segment less than three years postlaunch proves that biosimilars can find a meaningful place in the US market just as they have in Europe. " Tony Hooper Amgen Former Exec VP-Global Commercial Operations

Give it time… • “We’re still in the infancy in the US” – Chrys Kokino, head of biologics for North America at Mylan • “If we look at what’s happened in Europe, what’s happened in other regulated markets, it’s a really bright future for the US. ” – Mike Woolcock, senior VP at Apobiologix • “It’s an exciting time for biosimilars in the US today. ” – Gary Deeb, senior VP global licensing and business development, Lupin Ltd. 33 Pharma intelligence | informa

Next up: Neulasta the next bellwether for US biosimilar uptake • Mylan launched Fulphila in July • Mylan priced aggressively lower versus the brand, a WAC of $4, 175 per syringe, 33% discount to Neulasta WAC • Acute treatment administered in cancer care setting could result in more encouraging uptake • Still early days 34 Pharma intelligence | informa

European Biosimilar Landscape 35 Pharma intelligence | informa

More Products, Longer History • >50 launched biosimilars • Multiple molecules and competing products • Diversity of market type across Europe 36 Pharma intelligence | informa
Europe: Varied, But Tougher To Defend 37 Pharma intelligence | informa

Prospects For Originator Firms In Europe • Compete on price to share the market • Play to strengths in other areas: • • Education and support Logistics and inventory management Delivery device and administration mode Environmental impact of production • Focus on use of savings from biosimilars 38 Pharma intelligence | informa

A Turning Point For Humira • The first adalimumab biosimilars launched in Europe in October, all competing for a piece of Abb. Vie's $4. 4 bn in European revenues • It's the first time four biosimilars have launched at the same time, including Sandoz' Hyrimoz, Samsung Bioepsis/Biogen's Imraldi, Amgen's Amgevita and Mylan/Fujifilm Kyowa Kirin's Hulio and more on the way in 2019. • How will Abb. Vie defend its brand? Early signs suggest the company will be competing on price. • Abb. Vie has won one undisclosed tender for an 80% discount off the price of Humira 39 Pharma intelligence | informa

Eventually… • Biosimilars will take their place, even in the US, though it will be driven by European markets. • Some at the starting line may not stay the course, but big pharma’s presence in biosimilars could bring credibility to the market. • The speed and magnitude of price and market share erosion may be less painful for originators than some originally thought. • But eventually, biosimilar uptake is needed if global health care systems are going to be able to afford pharma’s next wave of innovative launches. 40 Pharma intelligence | informa

Questions?

Thanks for Attending! Next steps: We will address additional questions off line. All participants will be emailed a recording of the Webinar with slides. For more information visit pharmaintelligence. informa. com or email pharma@informa. com

Informa House 30 -32 Mortimer Street London W 1 W 7 RE T T Phone (US): +1 212 -520 -2765 Phone (UK & Europe): +44 (0) 20 701 76180 pharma@informa. com 43 Pharma intelligence | informa
- Slides: 43