Jeopardy Template By Carl Lyman September 2001 Jeopardy

Jeopardy Template By Carl Lyman © September 2001

Jeopardy Bonding Kinetics/Equilibri um Solutions Acid/Base 100 100 200 200 300 300 400 400 500 500

Credits Ü That’s all folks. Exit

Given the balanced equation: I + I → I 2 Which statement describes the process represented by this equation? A. ) A bond is formed as energy is absorbed. B. ) A bond is formed and energy is released. C. ) A bond is broken as energy is absorbed. D. ) A bond is broken and energy is released.

Given the balanced equation: I + I → I 2 Which statement describes the process represented by this equation? A. ) A bond is formed as energy is absorbed. B. ) A bond is formed and energy is released. C. ) A bond is broken as energy is absorbed. D. ) A bond is broken and energy is released.

Which atom will form an ionic bond with a Br atom? A. ) N B. ) Li C. ) O D. ) C

Which atom will form an ionic bond with a Br atom? Ionic Bond: Metal and a Nonmetal Bromine is a nonmetal A. ) N B. ) Li C. ) O D. ) C

In which compound do atoms form bonds by sharing electrons? A. ) H 2 O B. ) Na 2 O C. ) Ca. O D. ) Mg. O

In which compound do atoms form bonds by sharing electrons? Sharing = Covalent = 2 nonmetals A. ) H 2 O B. ) Na 2 O C. ) Ca. O D. ) Mg. O

Conductivity in a metal results from the metal atoms having A. ) high electronegativity B. ) high ionization energy C. ) highly mobile protons in the nucleus D. ) highly mobile electrons in the valence shell

Conductivity in a metal results from the metal atoms having “Sea of Mobile Electrons” A. ) high electronegativity B. ) high ionization energy C. ) highly mobile protons in the nucleus D. ) highly mobile electrons in the valence shell

Which formula represents a polar molecule? A. ) Cl 2 C. ) CH 4 B. ) CO 2 D. ) NH 3
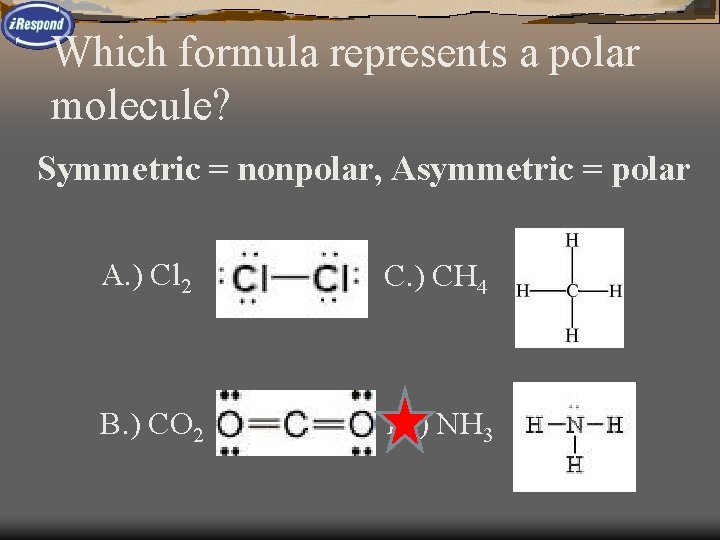
Which formula represents a polar molecule? Symmetric = nonpolar, Asymmetric = polar A. ) Cl 2 C. ) CH 4 B. ) CO 2 D. ) NH 3
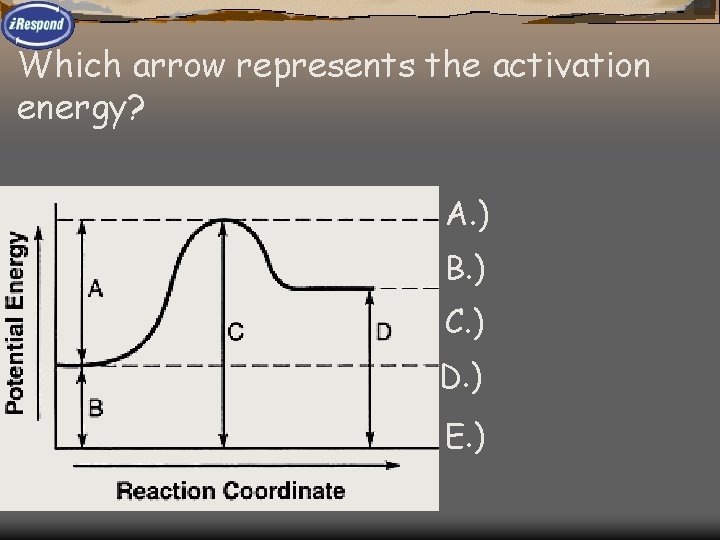
Which arrow represents the activation energy? A. ) 1 B. ) 2 E C. ) 3 D. ) 4 E. ) 5
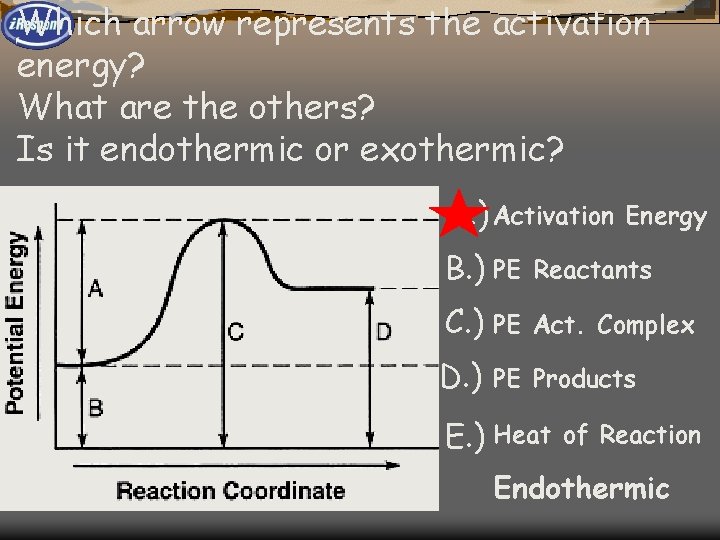
Which arrow represents the activation energy? What are the others? Is it endothermic or exothermic? A. ) Activation 1 E Energy B. ) PE 2 Reactants C. ) PE 3 Act. Complex D. ) 4 PE Products E. ) Heat 5 of Reaction Endothermic

A chemical reaction between iron atoms and oxygen molecules can only occur if A. ) the particles are heated B. ) the atmospheric pressure decreases C. ) there is a catalyst present D. ) there are effective collisions between the particles

A chemical reaction between iron atoms and oxygen molecules can only occur if A. ) the particles are heated B. ) the atmospheric pressure decreases C. ) there is a catalyst present D. ) there are effective collisions between the particles Proper orientation and sufficient kinetic energy

At equilibrium, what is the rate of production of reactants compared with the rate of production of products? A. ) much higher B. ) higher C. ) the same D. ) lower

At equilibrium, what is the rate of production of reactants compared with the rate of production of products? A. ) much higher B. ) higher C. ) the same D. ) lower Dynamic Equilibrium: Rates equal, Concentrations constant

What is the effect of adding more water to the following equilibrium reaction? CO 2 + H 2 O ↔ H 2 CO 3 A. ) More H 2 CO 3 is produced. B. ) CO 2 concentration increases. C. ) The equilibrium is pushed in the direction of reactants. D. ) There is no effect.

What is the effect of adding more water to the following equilibrium reaction? Shift right, causing an CO 2 + H 2 O ↔ H 2 CO 3 increase in A. ) More H 2 CO 3 is produced. product concentration B. ) CO 2 concentration increases. C. ) The equilibrium is pushed in the direction of reactants. D. ) There is no effect.

If a system is left to change spontaneously, in what state will it end? A. ) the same state in which it began B. ) the state with lowest possible energy C. ) the state with the maximum disorder D. ) the state with the lowest possible energy consistent with the state of maximum disorder

If a system is left to change spontaneously, in what state will it end? A. ) the same state in which it began B. ) the state with lowest possible energy C. ) the state with the maximum disorder D. ) the state with the lowest possible energy consistent with the state of maximum disorder

At STP, which of these substances is most soluble in H 2 O? A. ) CCl 4 C. ) HCl B. ) CO 2 D. ) N 2
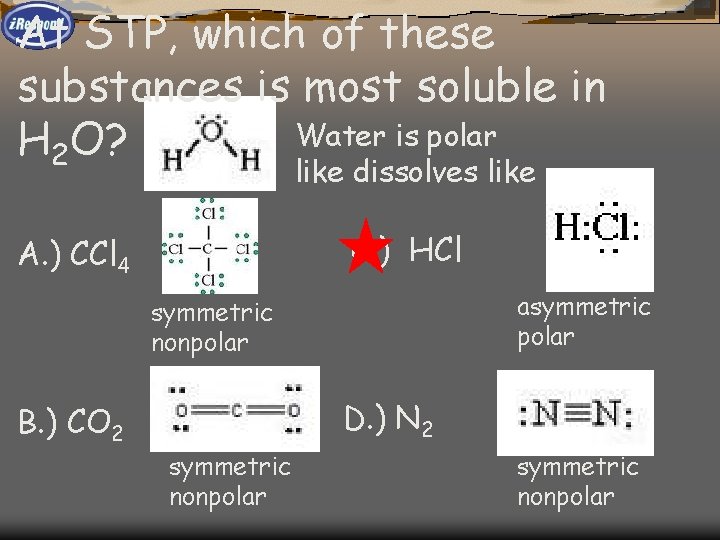
At STP, which of these substances is most soluble in Water is polar H 2 O? like dissolves like C. ) HCl A. ) CCl 4 asymmetric polar symmetric nonpolar D. ) N 2 B. ) CO 2 symmetric nonpolar
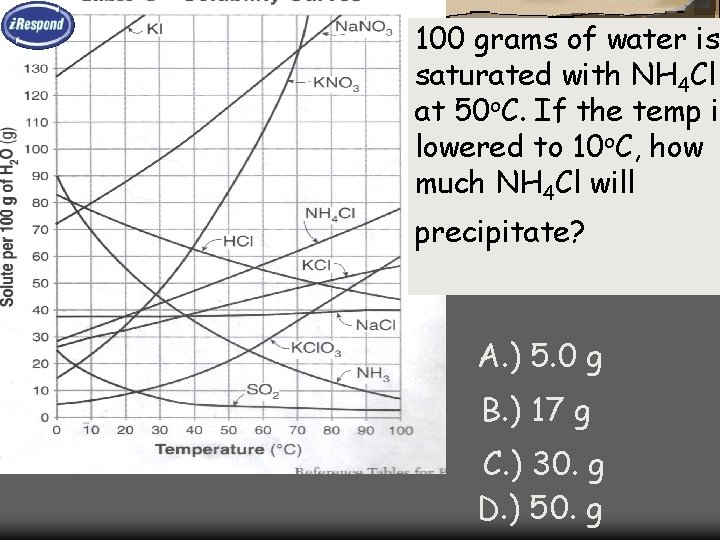
100 grams of water is saturated with NH 4 Cl at 50 o. C. If the temp is lowered to 10 o. C, how much NH 4 Cl will precipitate? A. ) 5. 0 g B. ) 17 g C. ) 30. g D. ) 50. g
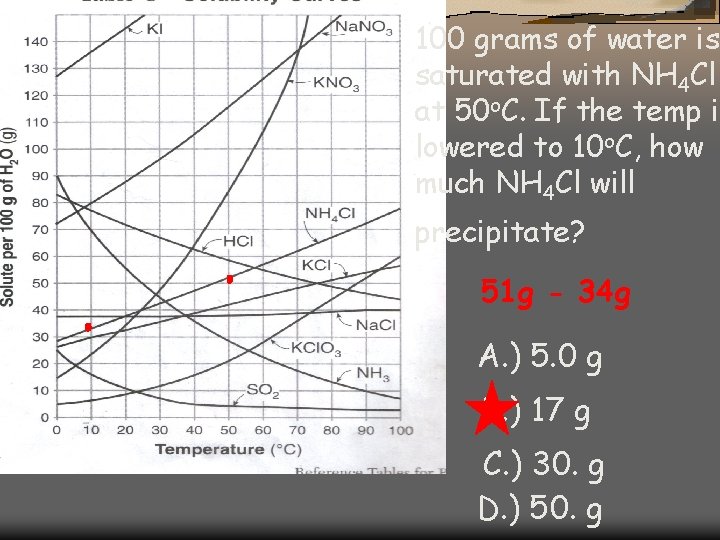
100 grams of water is saturated with NH 4 Cl at 50 o. C. If the temp is lowered to 10 o. C, how much NH 4 Cl will precipitate? 51 g - 34 g A. ) 5. 0 g B. ) 17 g C. ) 30. g D. ) 50. g

What is the total number of grams of Na. I(s) needed to make 1. 0 liter of a 0. 010 M solution? A. ) 0. 015 B. ) 0. 15 C. ) 1. 5 D. ) 15

What is the total number of grams of Na. I(s) needed to make 1. 0 liter of a 0. 010 M solution? A. ) 0. 015 B. ) 0. 15 C. ) 1. 5 D. ) 15 Molarity = moles solute Liters solution 0. 010 mol x 150 g = 1 mol 0. 010 M = X 1 L

How many milliliters of 12. 0 M HCl(aq) must be diluted with water to make exactly 500. m. L of 3. 00 M hydrochloric acid? A. ) 100. m. L B. ) 125. m. L C. ) 200. m. L D. ) 250. m. L

How many milliliters of 12. 0 M HCl(aq) must be diluted with water to make exactly 500. m. L of 3. 00 M hydrochloric acid? A. ) 100. m. L B. ) 125. m. L C. ) 200. m. L D. ) 250. m. L MV = MV (12. 0 M)V = (3. 00 M)(500 m. L)

At standard pressure when Na. Cl is added to water, the solution will have a A. ) higher freezing point and a lower boiling point than water B. ) higher freezing point and a higher boiling point than water C. ) lower freezing point and higher boiling point than water D. ) lower freezing point and a lower boiling point than water

At standard pressure when Na. Cl is added to water, the solution will have a A. ) higher freezing point and a lower boiling point than water B. ) higher freezing point and a higher boiling point than water C. ) lower freezing point and higher boiling point than water D. ) lower freezing point and a lower boiling point than water

An aqueous solution of an ionic compound turns red litmus blue, conducts electricity, and reacts with an acid to form a salt and water. This compound could be A. ) HCl B. ) Na. I C. ) KNO 3 D. ) Li. OH

D) Li. OH Ü Ü Ü Ü Statement describes a base and they end with OH ACIDS BASES Start with H* Example HCl(aq) Taste sour Turn blue litmus red Clear in phenolphthalein React with active metals to form H 2 gas React with carbonates to produce CO 2 gas p. H < 7 Ü Ü Ü End in OH* Example Na. OH(aq) Taste bitter Turn red litmus blue Pink in phenolphthalein Feel slippery p. H > 7 BOTH: Conduct electricity (are electrolytes)

H 2 O + CO 3 -2 OH- + HCO-3 The two Brönsted-Lowry acids are A. ) H 2 O and OHB. ) H 2 O and HCO-3 C. ) CO 3 -2 and OHD. ) CO 3 -2 and HCO-3

H 2 O + CO 3 -2 OH- + HCO-3 The two Brönsted-Lowry acids are A. ) H 2 O and OHB. ) H 2 O and HCO-3 C. ) CO 3 -2 and OHD. ) CO 3 -2 and HCO-3 Acids donate H+

Which p. H indicates a basic solution? A. ) 1 B. ) 5 C. ) 7 D. ) 12

Which p. H indicates a basic solution? A. ) 1 B. ) 5 C. ) 7 D. ) 12 p. H < 7, acidic p. H = 7, neutral p. H > 7, basic

What is the p. H of a 0. 01 M solution of HNO 3? A. ) 1 B. ) 2 C. ) 13 D. ) 14

What is the p. H of a 0. 01 M solution of HNO 3? A. ) 1 HNO 3 dissociates to produce H+(H 3 O+) ions B. ) 2 This problem is saying [H 3 O+] = 0. 01 M C. ) 13 p. H = -log[H 3 O+] D. ) 14 p. H = -log[0. 01]

How many milliliters of 1 M Na. OH are needed to exactly neutralize 100 milliliters of 1 M H 2 SO 4? A. ) 50 m. L B. ) 100 m. L C. ) 200 m. L D. ) 400 m. L

How many milliliters of 1 M Na. OH are needed to exactly neutralize 100 milliliters of 1 M H 2 SO 4? A. ) 50 m. L B. ) 100 m. L C. ) 200 m. L D. ) 400 m. L Moles H+ = Moles OHMV = MV 2(1 M)(100 m. L) = (1 M)V
- Slides: 43