Jeopardy Mixed Topics Endothermic Exothermic Reaction Rates Energy
















































- Slides: 53

Jeopardy Mixed Topics Endothermic/ Exothermic Reaction Rates Energy Diagrams Systems Q $100 Q $100 Q $200 Q $200 Q $300 Q $300 Q $400 Q $400 Q $500 Q $500 Final Jeopardy

$100 Question from H 1 What is activation energy?

$100 Answer from H 1 Energy required to start reaction

$200 Question from H 1 If the particles of a sample are moving Faster, how has the temperature changed?

$200 Answer from H 1 increased

$300 Question from H 1 If a catalyzed reaction stops and you Add more of the reactants, do you Need to add more of the catalyst? Explain.

$300 Answer from H 1 No. Catalyst isn’t used.

$400 Question from H 1 How does the temperature of a Substance change when a phase Change is occurring?

$400 Answer from H 1 Stays the same

$500 Question from H 1 What three phase changes increase In energy? I want the names!

$500 Answer from H 1 Melting, vaporization, sublimation

$100 Question from H 2 What happens in endothermic and Exothermic reactions?

$100 Answer from H 2 Endothermic- gains energy Exothermic- loses energy

$200 Question from H 2 How will a flask feel if an exothermic Reaction is happening inside it?

$200 Answer from H 2 hot

$300 Question from H 2 How will a flask feel if an endothermic Reaction is going on inside it?

$300 Answer from H 2 cold

$400 Question from H 2 In an endothermic reaction, do the Products have more or less energy Than the reactants?

$400 Answer from H 2 more

$500 Question from H 2 In an exothermic reaction, do the Products have more or less energy Than the reactants?

$500 Answer from H 2 less

$100 Question from H 3 How does increasing the concentration Affect the reaction rate?

$100 Answer from H 3 increases

$200 Question from H 3 How does increasing particle size Affect the reaction rate?

$200 Answer from H 3 decreases

$300 Question from H 3 Why does increasing temperature Increase the reaction rate?

$300 Answer from H 3 Faster particles, more collisions and More particles have activation energy

$400 Question from H 3 Why does increasing concentration Increase the reaction rate?

$400 Answer from H 3 More particles, more collisions

$500 Question from H 3 How does a catalyst increase the Reaction rate?

$500 Answer from H 3 Decreasing activation energy

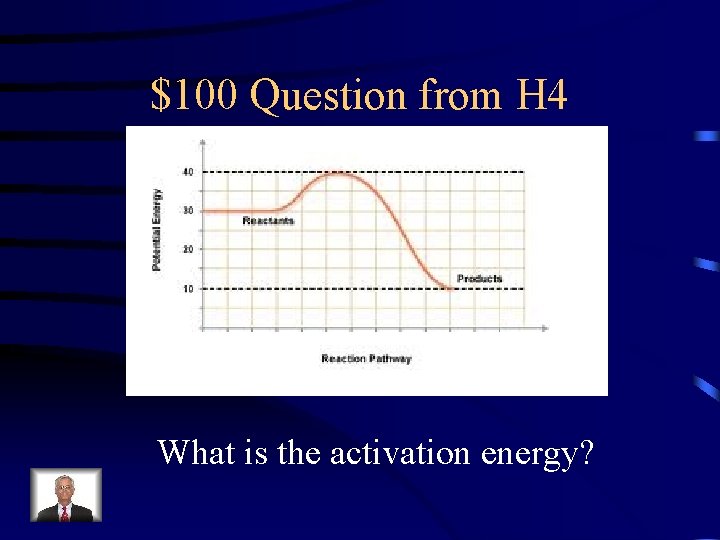
$100 Question from H 4 What is the activation energy?

$100 Answer from H 4 10 k. J

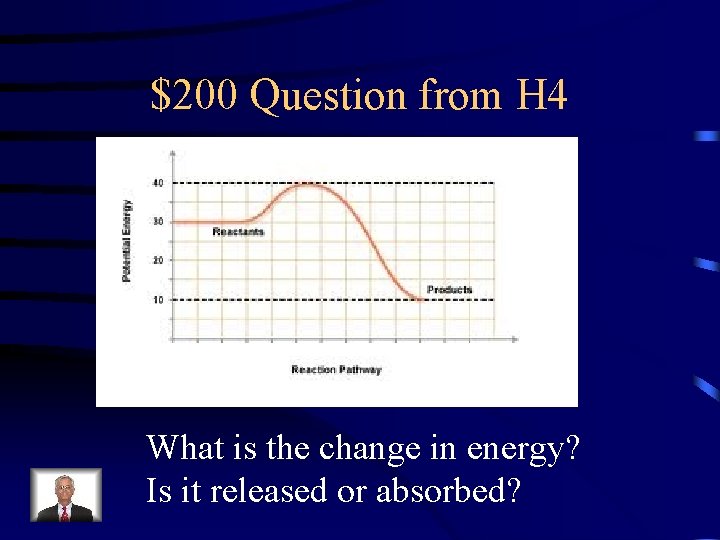
$200 Question from H 4 What is the change in energy? Is it released or absorbed?

$200 Answer from H 4 20 k. J released

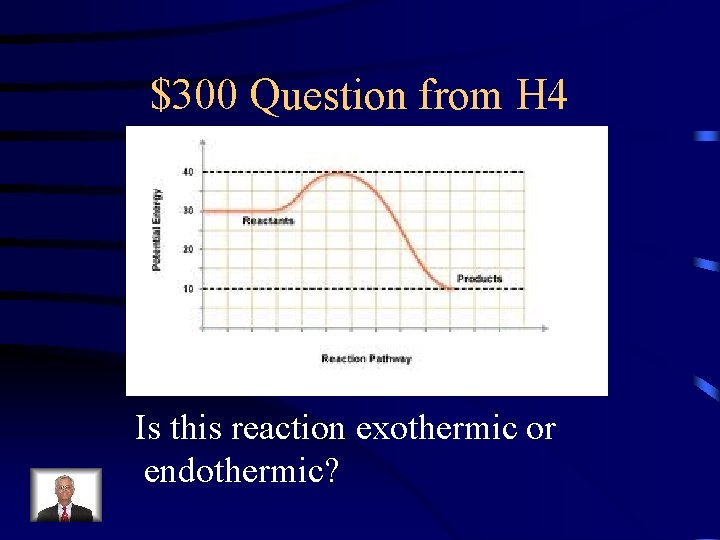
$300 Question from H 4 Is this reaction exothermic or endothermic?

$300 Answer from H 4 exothermic

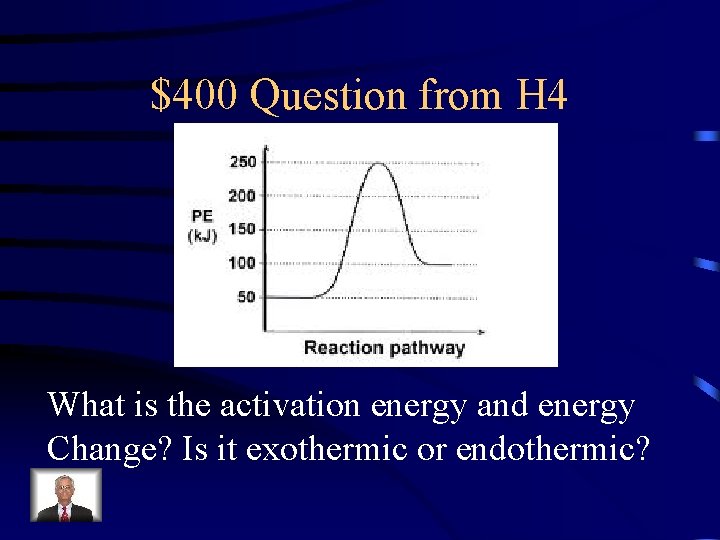
$400 Question from H 4 What is the activation energy and energy Change? Is it exothermic or endothermic?

$400 Answer from H 4 200 k. J, 50 k. J absorbed, endothermic

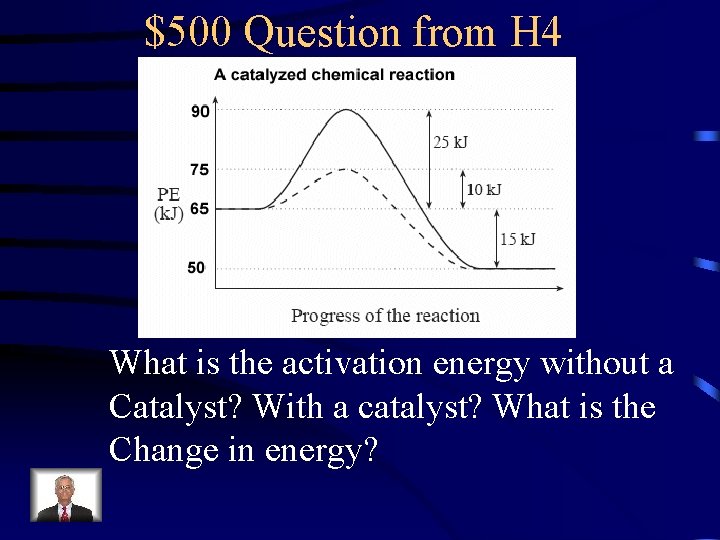
$500 Question from H 4 What is the activation energy without a Catalyst? With a catalyst? What is the Change in energy?

$500 Answer from H 4 25 k. J, 10 k. J, 15 k. J

$100 Question from H 5 What are three types of systems?

$100 Answer from H 5 Open, closed, isolated

$200 Question from H 5 What is a closed system?

$200 Answer from H 5 Energy exchange, not matter

$300 Question from H 5 What is an isolated system?

$300 Answer from H 5 Energy and matter don’t exchange

$400 Question from H 5 What is an open system?

$400 Answer from H 5 Energy and matter exchange

$500 Question from H 5 Give two examples for open, Closed, and isolated systems.

$500 Answer from H 5 Answers Vary

Final Jeopardy Draw an energy diagram where the reactants Have an energy of 25 k. J, the activation energy Is 60 k. J, and the reaction absorbs 30 k. J of Energy. Is the reaction endothermic or exothermic?

Final Jeopardy Answer endothermic