Jeopardy Decay Nuclear Stability Einsteins Famous Equation 100
Jeopardy Decay Nuclear Stability Einstein’s Famous Equation $100 $200 $200 $300 $300 $400 $400 $500 Final Jeopardy $500 Fundamental Forces Uses for Nuclear Physics $100

Fundamental Forces - $100 • This force pulls all mass together and is responsible for very large systems • What is Gravity?

Fundamental Forces - $200 n This fundamental force is the reason negative electrons surround a positive nucleous n What is electromagnetic force?

Fundamental Forces - $300 n This fundamental force is what allows stars to “burn” and allows neutrons to change into protons-Beta Decay n What is weak force?

Fundamental Forces - $400 n This fundamental force fights against the repelling force of electromagnetic force caused by proton-proton interactions inside the nucleus…it holds the nucleus together n What is strong force?

Fundamental Force - $500 n The fission of an atom occurs when this force is overpowered due to its limited effective distance n What is strong force?

Uses for Nuclear Physics - $100 n This device is used to detect radioactive particles n What is the Geiger counter?

Uses for Nuclear Physics - $200 n This device takes 3 -D “pictures” of internal body structures and acronym CAT scan n What is computerized axial tomography?

Uses for Nuclear Physics - $300 n High energy photons of this type of EM radiation are used to precisely destroy cancer cells n What are X-rays?

Uses for Nuclear Physics - $400 n Wilhelm Rӧntgen first discovered an unknown radiation that allowed him to see through tissue and examine the bones. A device was later created that is commonly used in the medical field and industrial fields. n What is an X-ray machine?

Uses for Nuclear Physics - $500 n This device uses radio frequencies to excite hydrogen atoms within a body to produce a magnetic field. The field is used to produce an image of the internal structures of the body including soft tissue. n What is magnetic resonance imaging or MRI?

Decay- $100 n This type of decay uses the weak force to change neutrons into protons. In the process an electron is emitted from the nucleus. n What is beta decay?

Decay- $200 n This type of decay leads to a lower atomic mass and number; a positive particle consisting of two protons and two neutrons…a helium nucleus n What is alpha decay?

Decay- $300 n This “decay” result from when an energetic nucleus releases the energy in the form of a high energy (high frequency) photon n What is gamma decay?

Decay- $400 n This is the order from least to greatest penetration power for the three types of decay studied n What is alpha, beta and gamma?

Decay- $500 n The correct order of decays for the first three reactions (arrows) n What is alpha, beta and beta

Nuclear Stability - $100 n This is the nuclear process by which lighter elements combine to form heavier elements n What is fusion?

Nuclear Stability - $200 n This is the process by which heavy elements divide to form lighter elements and is due to an imbalances between protons and neutrons…too many neutrons n What is fission?

Nuclear Stability - $300 n This is the most atomically stable element on periodic table of elements n What is iron?

Nuclear Stability - $400 n Nuclear reactors produce energy uses heavy elements that undergo this process n What is fission?

Nuclear Stability - $500 n Unstable isotopes of uranium are used in nuclear bombs and nuclear reactors. This subatomic particle allows more uranium to split creating a “chain reaction” n What is slow moving neutrons?
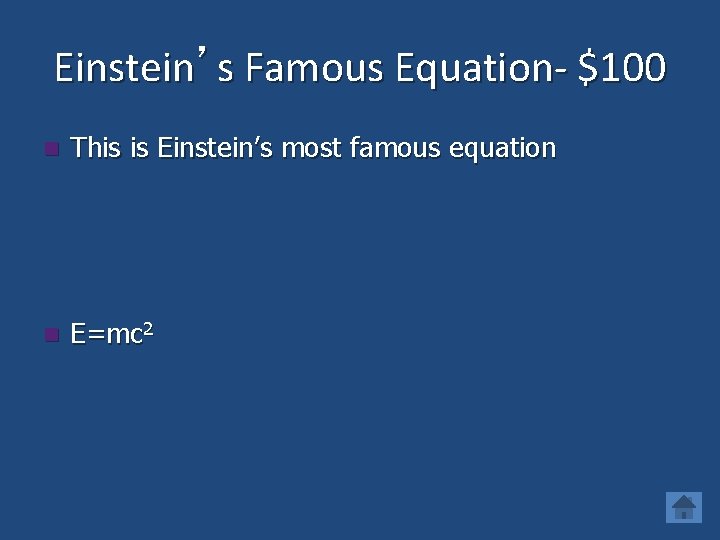
Einstein’s Famous Equation- $100 n This is Einstein’s most famous equation n E=mc 2
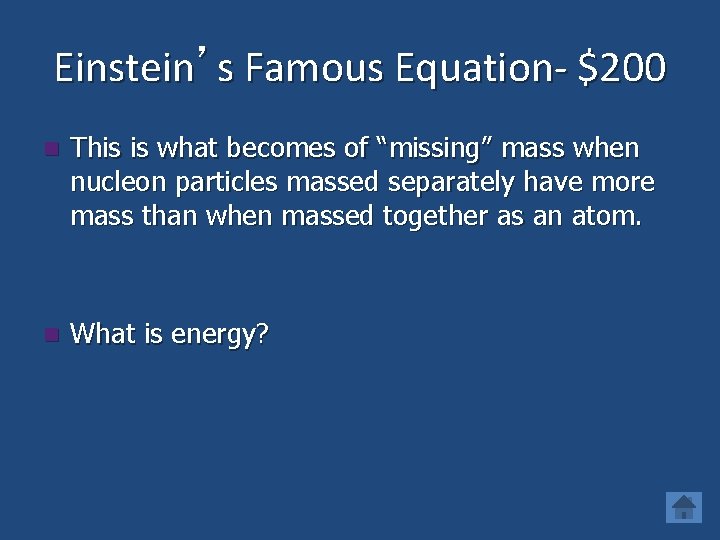
Einstein’s Famous Equation- $200 n This is what becomes of “missing” mass when nucleon particles massed separately have more mass than when massed together as an atom. n What is energy?

Einstein’s Famous Equation- $300 n This is the amount of energy that is produced when a nuclear reaction has a mass defect (missing mass) of 9 x 10 -16 kg n 81 joule

Einstein’s Famous Equation- $400 n This element has the lowest mass per nucleon of any element n Iron…the most stable

Einstein’s Famous Equation- $500 n The first atomic bomb, code name Trinity, exploded with an energy of 75 TJ or 7. 5 x 1013 Joules. A mass of this much had to be converted to energy. n 8. 33 x 10 -4 kg or. 833 grams

Final Jeopardy n Today’s modern nuclear warheads are this type. The first atomic bomb used against another country was this type. n Fusion bombs, Fission bombs
- Slides: 27