JEOPARDY Click Once to Begin Chemistry Final Review

JEOPARDY! Click Once to Begin Chemistry Final Review Template by Bill Arcuri, WCSD

JEOPARDY! Dissolving Concentration Parts of Solutions Solubility/ Precipitation Acids and Bases Real Life Applications 100 100 100 200 200 200 300 300 300 400 400 400 500 500 500 Template by Bill Arcuri, WCSD
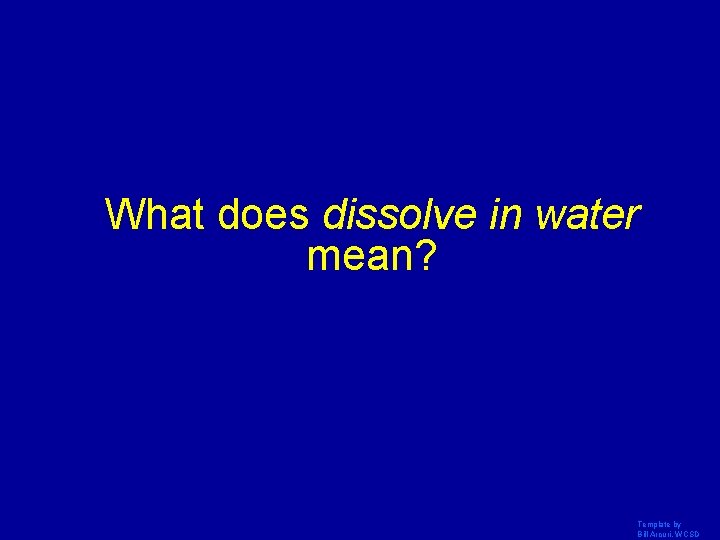
What does dissolve in water mean? Template by Bill Arcuri, WCSD

Dissolve in water means that the ions of the compound break up when put in water Template by Bill Arcuri, WCSD

What is an electrolyte? Template by Bill Arcuri, WCSD

An electrolyte is a compound that dissolves in water Template by Bill Arcuri, WCSD

What are unsaturated, and supersaturated solutions? Template by Bill Arcuri, WCSD

Unsaturated means that more solute can be added, saturated means that a solution has dissolved all the solute it can hold, supersaturated means that due to an increase in heat a solution has more solute dissolved than it can normally hold. Template by Bill Arcuri, WCSD

Break up (NH 4)3 PO 4 into ions Template by Bill Arcuri, WCSD
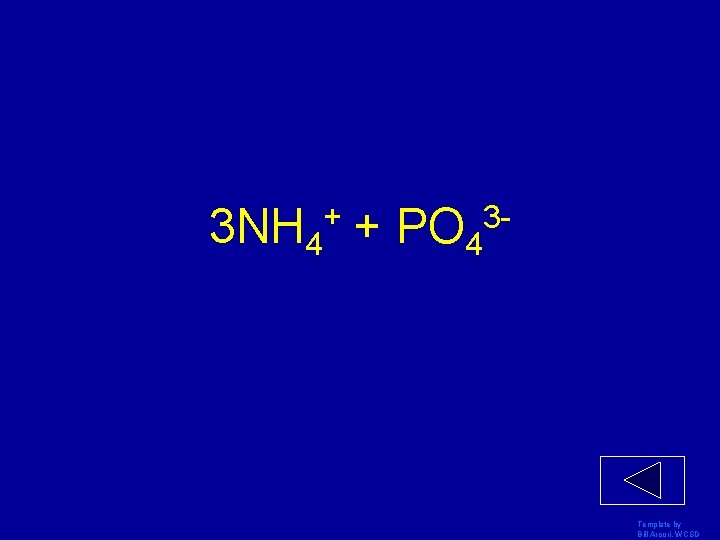
3 NH 4+ + PO 43 - Template by Bill Arcuri, WCSD

What is the formula for vinegar and what kind of electrolyte is it? Why? Template by Bill Arcuri, WCSD

The formula is HCH 3 COO and it is a weak electrolyte because the not many H+ ions dissociate. Template by Bill Arcuri, WCSD
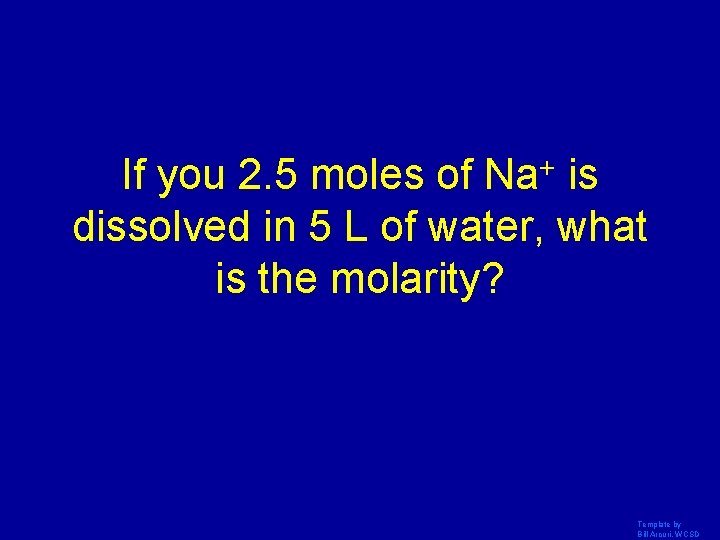
If you 2. 5 moles of Na+ is dissolved in 5 L of water, what is the molarity? Template by Bill Arcuri, WCSD

. 5 M + Na Template by Bill Arcuri, WCSD

What is the molarity of Cl- in 1. 2 M Ca. Cl 2? Template by Bill Arcuri, WCSD

2. 4 M Cl Template by Bill Arcuri, WCSD

Find the %(W/V) for 60 g of solute in. 6 L of a solution Template by Bill Arcuri, WCSD

10%(W/V) Don’t forget to convert to milliliters for %(W/V) Template by Bill Arcuri, WCSD

Find the molarity of 5. 6 g of lead nitrate in 400 m. L water Template by Bill Arcuri, WCSD
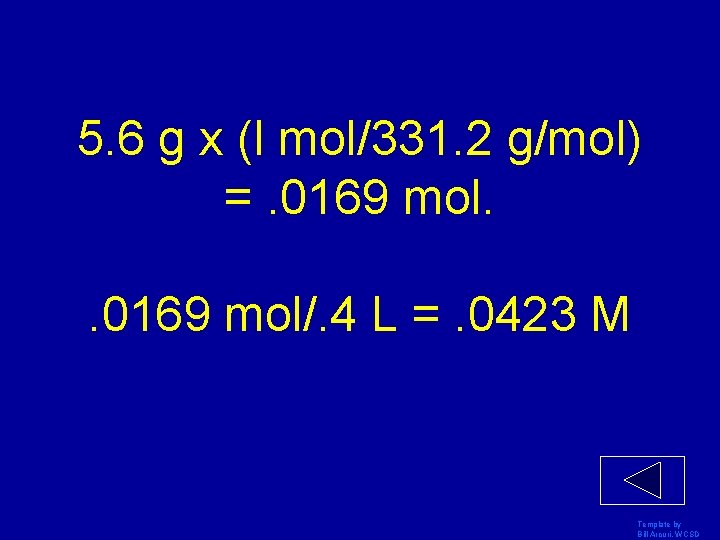
5. 6 g x (l mol/331. 2 g/mol) =. 0169 mol/. 4 L =. 0423 M Template by Bill Arcuri, WCSD

Daily Double!!! What is the total ion molarity for 6. 7 g of Ammonium Sulfide in 5 L of water? Template by Bill Arcuri, WCSD

6. 7 g x (1 mol Ammonium Sulfide/68. 154 g/mol) =. 0983 mol x 3 ions =. 295 mol Template by Bill Arcuri, WCSD

What is a solute? Template by Bill Arcuri, WCSD

A solute is the chemical that is dissolved Template by Bill Arcuri, WCSD
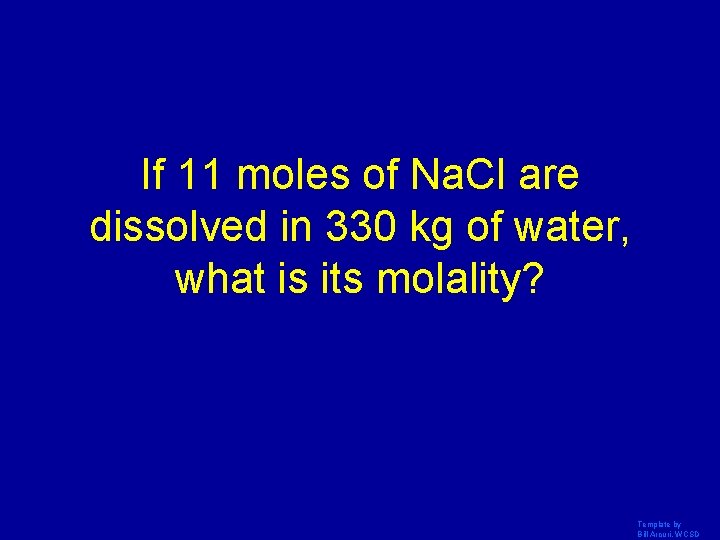
If 11 moles of Na. Cl are dissolved in 330 kg of water, what is its molality? Template by Bill Arcuri, WCSD

. 033 m Template by Bill Arcuri, WCSD

When magnesium chloride is placed into water, it seems to disappear. Does it disappear? If not, why does it seem this way? Template by Bill Arcuri, WCSD

Magnesium chloride does not disappear when put in water, the particles are just surrounded by water and too tiny to see. Template by Bill Arcuri, WCSD

What is hydration and ionization? Template by Bill Arcuri, WCSD

Hydration is what occurs when an ionic compound dissolves in water, ionization is the process of breaking up the compound into separate ions. Template by Bill Arcuri, WCSD

What kind of electrolytes conduct electricity well? Why? Template by Bill Arcuri, WCSD

Strong electrolytes conducted electricity better because they make freefloating ions in water. Template by Bill Arcuri, WCSD

What is a precipitate? Template by Bill Arcuri, WCSD

A precipitate is an insoluble compound that does not dissolve in water Template by Bill Arcuri, WCSD

If silver nitrate was mixed with sodium chloride, would a precipitate form? Template by Bill Arcuri, WCSD

Yes, Ag. Cl would precipitate Template by Bill Arcuri, WCSD

Indicate what precipitates forms, if any: Na. NO 3 + Li 2 CO 3 Template by Bill Arcuri, WCSD

No precipitate forms Template by Bill Arcuri, WCSD

Indicate what precipitates forms, if any: Ca(CH 3 COO)2 + Na 3 PO 4 Template by Bill Arcuri, WCSD

Ca 3(PO 4)2 would form Template by Bill Arcuri, WCSD

Indicate what precipitates forms, if any: Magnesium iodide and mercury (I) flouride Template by Bill Arcuri, WCSD

Hg 2 I 2 and Mg. F 2 would form Template by Bill Arcuri, WCSD

What is the difference between p. H and p. OH? Template by Bill Arcuri, WCSD

p. H is a measure of + the H concentration and p. OH is a measure of the OH concentration Template by Bill Arcuri, WCSD

If there are. 006 mol of H+ in 2 L of water, what is the H+ concentration? Template by Bill Arcuri, WCSD
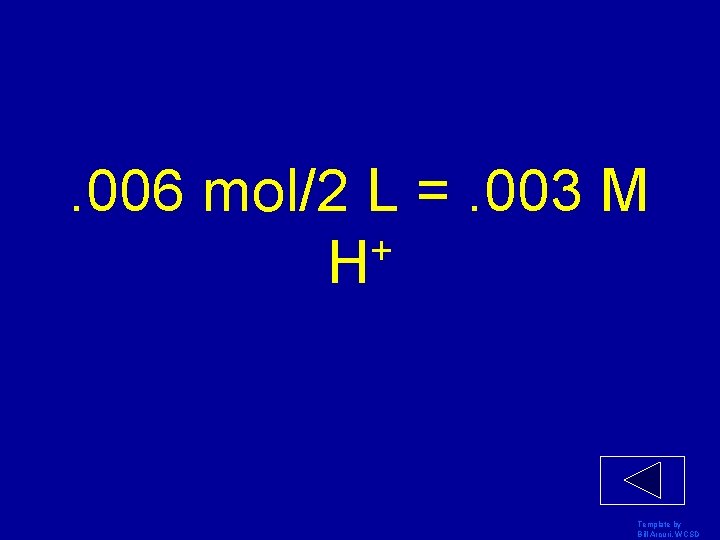
. 006 mol/2 L =. 003 M + H Template by Bill Arcuri, WCSD

If the p. H increase by 2, what happens to the H+ concentration? Template by Bill Arcuri, WCSD

It decreases by 100 Template by Bill Arcuri, WCSD

What is the H+ concentration if the p. OH is 8. 6? Template by Bill Arcuri, WCSD

14 – 8. 6 = 5. 4 p. H = 5. 4 -6 10 = 3. 98 x 10 Template by Bill Arcuri, WCSD

Daily Double!!! If. 05 moles of HCl are put in 60 L of water, what is the p. H? Template by Bill Arcuri, WCSD

. 05 moles / 60 L = -4 8. 33 x 10 M (-8. 33 x 10 -4) = 3. 08 10 Template by Bill Arcuri, WCSD

What are real life uses for an acid and a base? Template by Bill Arcuri, WCSD

Acid: Batteries, fertilizers, soaps, shampoo, detergents Base: Drain cleaner, laxatives, antacids Template by Bill Arcuri, WCSD

How could the sweetness of lemonade be maximized in accordance with saturation? Template by Bill Arcuri, WCSD

Super saturation of the lemonade with sugar Template by Bill Arcuri, WCSD

What effects from acid rain suggest that it is acidic? Template by Bill Arcuri, WCSD

Corrosion of metal/stone, lower the p. H of water bodies, damage to nature via resource ruination Template by Bill Arcuri, WCSD

Name one important food and the electrolyte in it Template by Bill Arcuri, WCSD

+ (K ), Banana Salty foods (Na+/Cl-), Milk (Ca 2+), Leafy 2+ Greens (Mg ) Template by Bill Arcuri, WCSD

If you frequently have heartburn , would putting ketchup on your French fries increase or decrease the odds of you getting heartburn? Why? Template by Bill Arcuri, WCSD

It would increase your odds of getting heartburn because ketchup has vinegar (acetic acid), which is acidic. Template by Bill Arcuri, WCSD

Final Jeopardy If. 5 mol H 2 SO 4 dissolved in 30 L of water is added to 2 mol HCl dissolved in 10 L of water, what is the p. OH? Template by Bill Arcuri, WCSD

. 5 mol / 40 L =. 0125 M x 2 H+ =. 025 M H+ in H 2 SO 4 2 mol / 40 L =. 5 M H+ in HCl. Click Once to Begin . 025 M H+ in H 2 SO 4 +. 05 M H+ in HCl =. 075 M H+ total. -log(. 075 M) = 1. 12 14 – 1. 12 = 12. 87 p. OH = 12. 87 Template by Bill Arcuri, WCSD
- Slides: 64