JEOPARDY Click Once to Begin Chemistry Final Review

JEOPARDY! Click Once to Begin Chemistry Final Review Chapter 3: Matter and Gas Laws Template by Bill Arcuri, WCSD

JEOPARDY! States of Matter Properties of Matter Gas Behavior Gas Laws Calculations Misc. 100 100 100 200 200 200 300 300 300 400 400 400 500 500 500 Template by Bill Arcuri, WCSD

Name the five states of matter. Template by Bill Arcuri, WCSD

Solid, Liquid, Gas, Plasma, Bose-Einstein Condensate Template by Bill Arcuri, WCSD

What is it called when a solid enters a liquid state? Template by Bill Arcuri, WCSD

Melting Template by Bill Arcuri, WCSD

What is it called when a liquid enters the gas state? Template by Bill Arcuri, WCSD

Evaporation Template by Bill Arcuri, WCSD

What is it called when a gas enters the solid state? Template by Bill Arcuri, WCSD

Deposition Template by Bill Arcuri, WCSD

How fast does light travel through a Bose-Einstein Condensate? Template by Bill Arcuri, WCSD

The speed of a bicycle Template by Bill Arcuri, WCSD

What is the difference between physical and chemical properties? Template by Bill Arcuri, WCSD

Physical properties can be observed without changing the molecule, chemical properties cannot. Template by Bill Arcuri, WCSD

Explain the difference between intensive and extensive properties. Template by Bill Arcuri, WCSD

Intensive does not depend on the size of the sample and extensive does. Template by Bill Arcuri, WCSD

What is the equation for density? Template by Bill Arcuri, WCSD

Density= mass/volume Template by Bill Arcuri, WCSD

To what property of matter is floating due? Template by Bill Arcuri, WCSD

Density Template by Bill Arcuri, WCSD
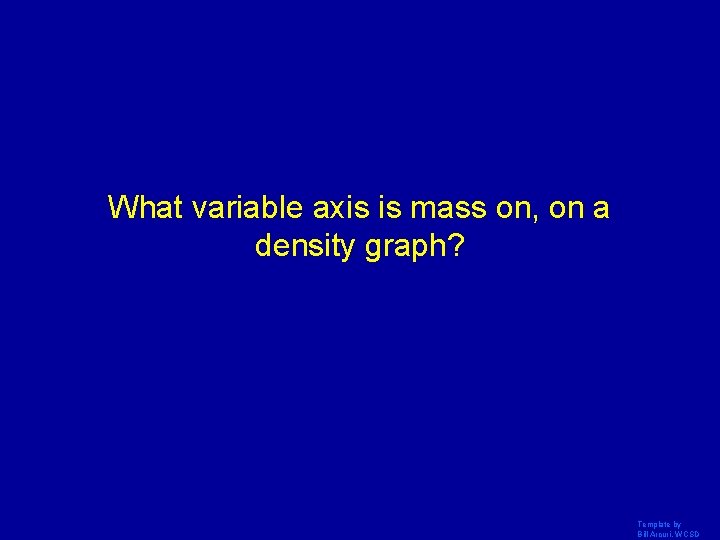
What variable axis is mass on, on a density graph? Template by Bill Arcuri, WCSD
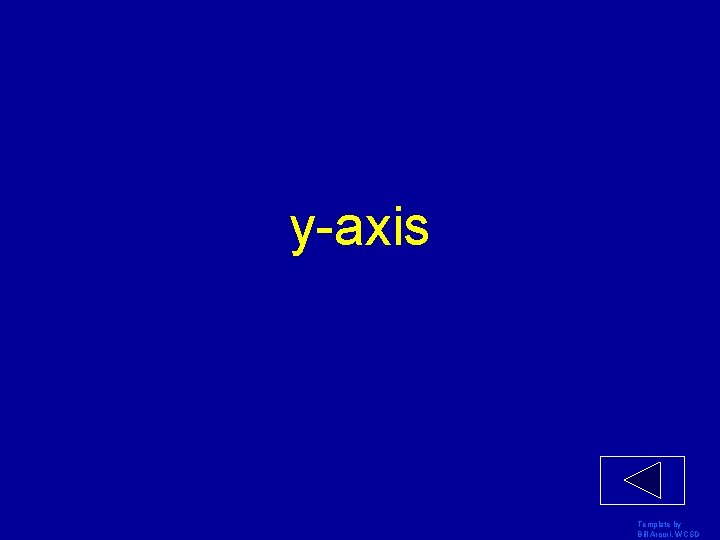
y-axis Template by Bill Arcuri, WCSD

Define pressure Template by Bill Arcuri, WCSD

The force of the gas atoms or molecules running into the wall of their container Template by Bill Arcuri, WCSD

What is the Kinetic Molecular Theory (not the assumptions)? Template by Bill Arcuri, WCSD

The theory that explains the over all behavior of gasses. Template by Bill Arcuri, WCSD

What are the main assumptions of the Kinetic Molecular Theory? Template by Bill Arcuri, WCSD

- Matter consists of small particles which have mass - The distances separating gas molecules is so large that the volume of the molecules themselves is insignificant - Gas particles move constantly, randomly, and rapidly - Collisions of gas particles are completely elastic (no energy loss). - The temperature of gas particles is directly related to their temperature - Gas particles exert no force on each other, and neither attract nor repel other gas particles Template by Bill Arcuri, WCSD

Describe the behavior of an ideal gas. Template by Bill Arcuri, WCSD

It exactly follows the KMT. Template by Bill Arcuri, WCSD

Do real gases always adhere to the KMT? Template by Bill Arcuri, WCSD

Template by Bill Arcuri, WCSD

What are the five units of pressure? Template by Bill Arcuri, WCSD

atm, mm Hg, psi, torr, k. Pa Template by Bill Arcuri, WCSD

1 atm is equal to (use another unit of pressure)… Template by Bill Arcuri, WCSD

760 mm Hg, 14. 7 psi, 760 torr, 101. 3 k. Pa (any are acceptable) Template by Bill Arcuri, WCSD
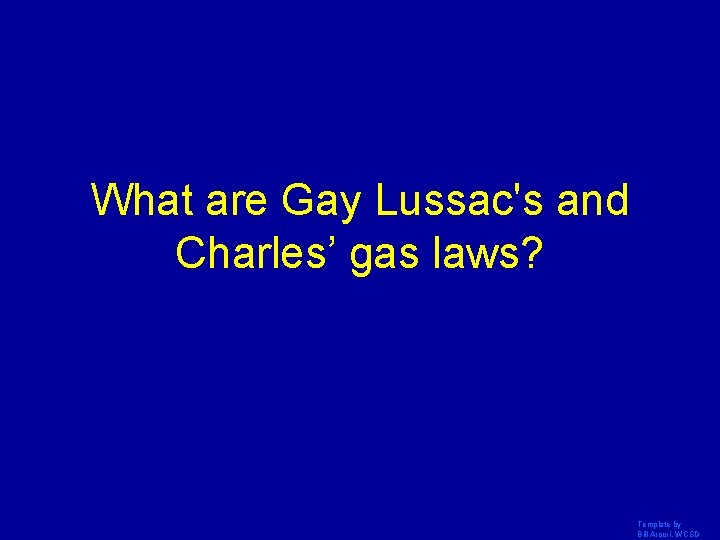
What are Gay Lussac's and Charles’ gas laws? Template by Bill Arcuri, WCSD
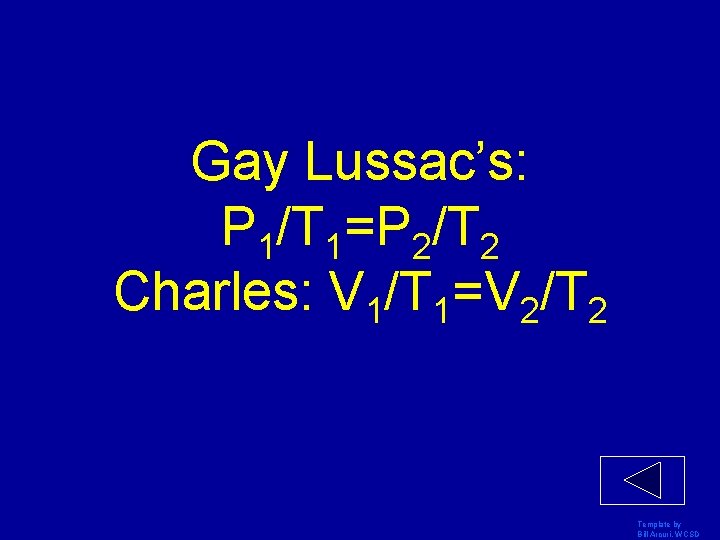
Gay Lussac’s: P 1/T 1=P 2/T 2 Charles: V 1/T 1=V 2/T 2 Template by Bill Arcuri, WCSD

What are Boyle’s and the Combined gas laws? Template by Bill Arcuri, WCSD

Boyle: P 1 V 1=P 2 V 2 Combined: P 1 V 1/n 1 T 1=P 2 V 2/n 2 T 2 Template by Bill Arcuri, WCSD

What is the ideal gas law? Template by Bill Arcuri, WCSD

PV=n. RT Template by Bill Arcuri, WCSD

What is 2+2? Template by Bill Arcuri, WCSD

Four Template by Bill Arcuri, WCSD

A gas occupies 15. 7 L at 1. 25 atm. What is the volume at standard pressure? Template by Bill Arcuri, WCSD

19. 6 L Template by Bill Arcuri, WCSD
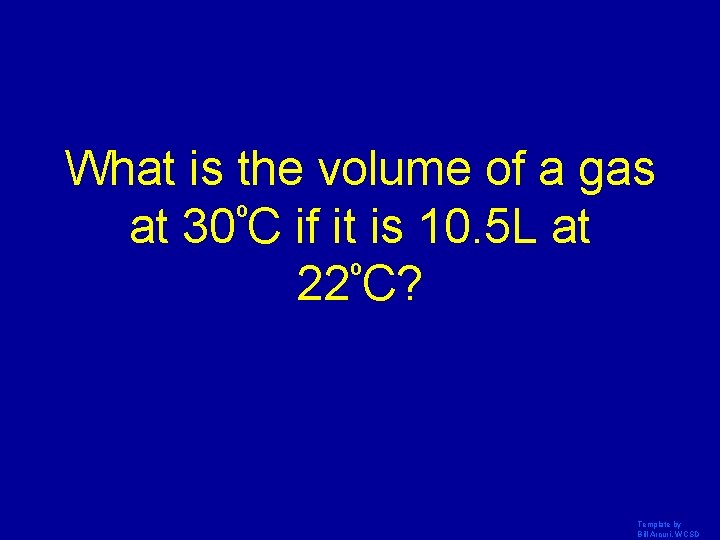
What is the volume of a gas at 30ºC if it is 10. 5 L at º 22 C? Template by Bill Arcuri, WCSD

10 L Template by Bill Arcuri, WCSD

How many moles are needed to create 25. 0 L at 105 k. Pa and 20ºC? Template by Bill Arcuri, WCSD

1 mol Template by Bill Arcuri, WCSD

Calculate Kp for each of the two reactions (happening in the same flask): 2 Fe. SO 4(s) <===> Fe 2 O 3(s) + SO 3(g) + SO 2(g)SO 3(g) <===> SO 2(g) + 1/2 O 2(g) After equilibrium is reached, total pressure is 0. 836 atm and partial pressure of oxygen is 0. 0275 atm. Template by Bill Arcuri, WCSD

First reaction: Kp=. 163 Second reaction: Kp=. 190 Template by Bill Arcuri, WCSD
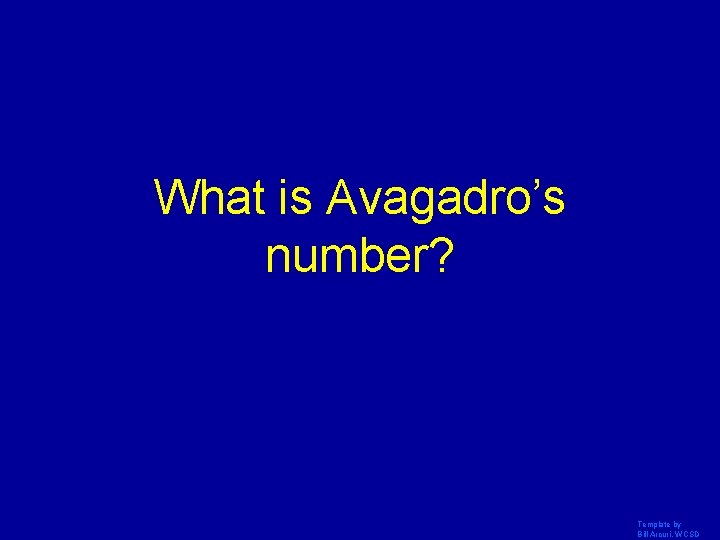
What is Avagadro’s number? Template by Bill Arcuri, WCSD

23 6. 02 x 10 Template by Bill Arcuri, WCSD

How many moles are in a mole of moles? Template by Bill Arcuri, WCSD

602, 000, 00 0, 000 moles Template by Bill Arcuri, WCSD

What are the units for atomic mass? Template by Bill Arcuri, WCSD

g/mol Template by Bill Arcuri, WCSD

What are the conditions of Standard Temperature and Pressure? Template by Bill Arcuri, WCSD

1 atm(760 mm Hg, 14. 7 psi, 760 torr, 101. 3 k. Pa), and 273 K (or 0ºC) Template by Bill Arcuri, WCSD

Why do balloons shrink when you put them in liquid nitrogen? Template by Bill Arcuri, WCSD

Liquid nitrogen is very cool and since gases become lesser in volume as they lose kinetic energy (temperature), the gases inside the balloons shrink bringing the container along with it. Template by Bill Arcuri, WCSD

Final Jeopardy: How will you do on the chemistry final? Template by Bill Arcuri, WCSD

Template by Bill Arcuri, WCSD
- Slides: 64