Jeff Taylor Mars Bulk Composition 1 Jeff Taylor

Jeff Taylor Mars Bulk Composition 1

Jeff Taylor Mars Bulk Composition 2

Bulk Composition of Silicate Mars • Ways used to determine Mars bulk composition • Cosmochemical components approach (Morgan and Anders) • Isotopically-based estimates (Lodders and Fegley, Sanloup et al. ) • Outline of Wänke and Driebus approach • Revised Martian bulk composition • Planetary Comparisions Jeff Taylor Mars Bulk Composition 3
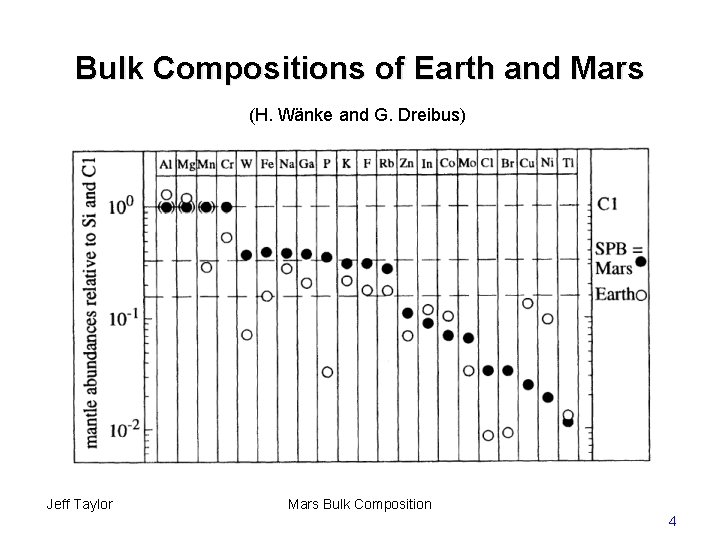
Bulk Compositions of Earth and Mars (H. Wänke and G. Dreibus) Jeff Taylor Mars Bulk Composition 4
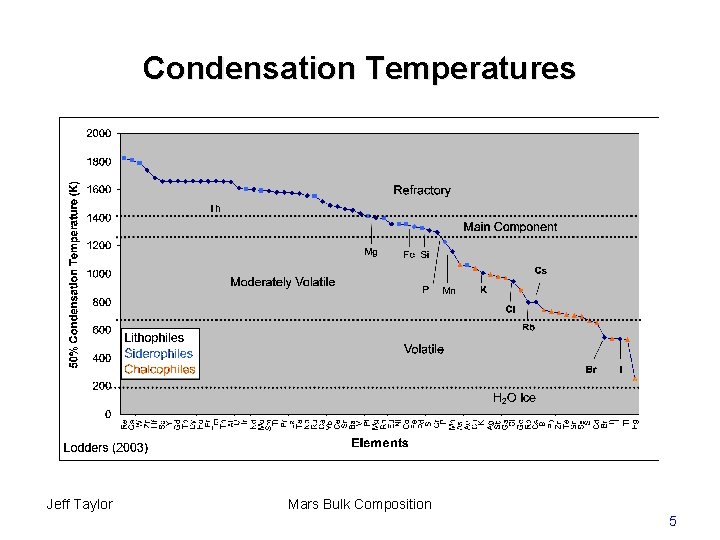
Condensation Temperatures Jeff Taylor Mars Bulk Composition 5

Morgan and Anders Approach • Mars a mixture of five primary condensates from nebula: – – High-T, refractory-rich condensate Fe-Ni metal Major silicates Fe. S and Fe. O (postulated to have formed by reaction with H 2 S and H 2 O, respectively) – Volatile-rich • Use “index” elements to calculate the abundances of all the elements in that group, assuming chondritic proportions – – – U, Th, REE, etc. Just need one of ‘em Fe K/U or K/Th Fe. O/(Fe. O+Mg. O) Tl or some other highly volatile element (Br? ) Jeff Taylor Mars Bulk Composition 6

Morgan and Anders Test • They estimate K/Th in Mars is 620. • It is really 5000. • But they did not know about Martian meteorites or have good elemental data from orbital missions. Jeff Taylor Mars Bulk Composition 7
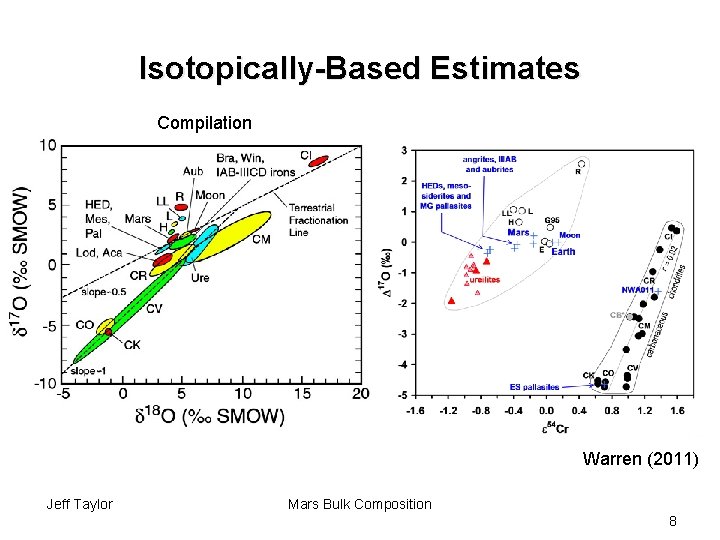
Isotopically-Based Estimates Compilation Warren (2011) Jeff Taylor Mars Bulk Composition 8

Isotopically-Based Estimates • Fit oxygen isotopic composition of Mars from known types of chondritic meteorites. • Lodders and Fegley (1997) estimate Mars made from 85% H-chondrites, 11% CV chondrites, and 4% CI condrites • Gives K/Th of 16, 000 for Mars, not the measured 5000. Jeff Taylor Mars Bulk Composition 9

Wänke and Dreibus Approach • Assumptions: – All elements less volatile than Na are in CI carbonaceous chondrite abundances (these elements are Al, Ca, Mg, REE, Th, U, Zr as common refractory elements, and Mn, Cr, W, and Fe) – In practice, they used elements less volatile (more refractory) than Mn – SNC meteorites reflect composition of Martian mantle Jeff Taylor Mars Bulk Composition 10

Wänke and Dreibus Approach • Inherent in model is concept of two components that combined to form Mars (and other terrestrial planets – Component A: Free of all elements equal or more volatile than Mn. Rest of the elements are in CI abundances. Fe and siderophile elements are metallic – Component B: All elements are oxidized, including volatiles, in CI abundances • Many elements were determined by ratios, e. g. , K/U or K/La, K vs Rb, etc. Jeff Taylor Mars Bulk Composition 11

Testing the Wänke and Dreibus Model • We have many more martian meteorites • We now have global data for some elements – K and Th to test K/refractory correlation – Fe on surface to test inferred high Fe. O in bulk silicate Mars • We will: – Test refractory element assumption – Show bunches of ratios of a refractory element to a volatile element (with similar geochemical behavior) – Compare the new composition to Earth Jeff Taylor Mars Bulk Composition 12

Jeff Taylor Mars Bulk Composition 13

Relation of Surface to Entire Crust • Ancient highlands megaregolith must have mixed the upper 10 -20 km; basin ejecta derived from depth • Sedimentary deposits (e. g. , in Arabia) may represent an average of upper crust, analogous to sediments on Earth • Abundant lava flows on surface probably accompanied by several times as much intrusive magmas • Magmas reflect mantle sources • Ancient enriched crust may preserve a geochemical record of the primitive mantle composition • Use of ratios of elements with similar geochemical behavior (e. g. , K/Th) avoids uncertainties in absolute concentrations Jeff Taylor Mars Bulk Composition 14
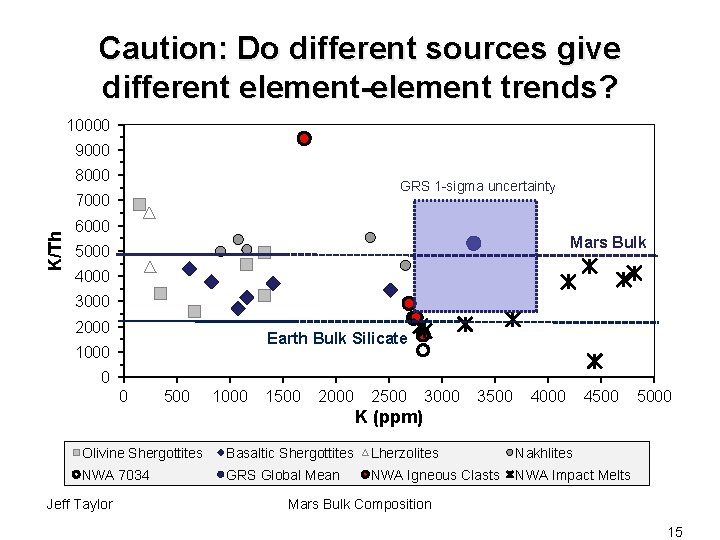
Caution: Do different sources give different element-element trends? 10000 9000 8000 GRS 1 -sigma uncertainty K/Th 7000 6000 Mars Bulk 5000 4000 3000 2000 Earth Bulk Silicate 1000 0 0 500 1000 1500 2000 2500 3000 3500 4000 4500 5000 K (ppm) Olivine Shergottites Basaltic Shergottites Lherzolites Nakhlites NWA 7034 GRS Global Mean NWA Igneous Clasts NWA Impact Melts Jeff Taylor Mars Bulk Composition 15
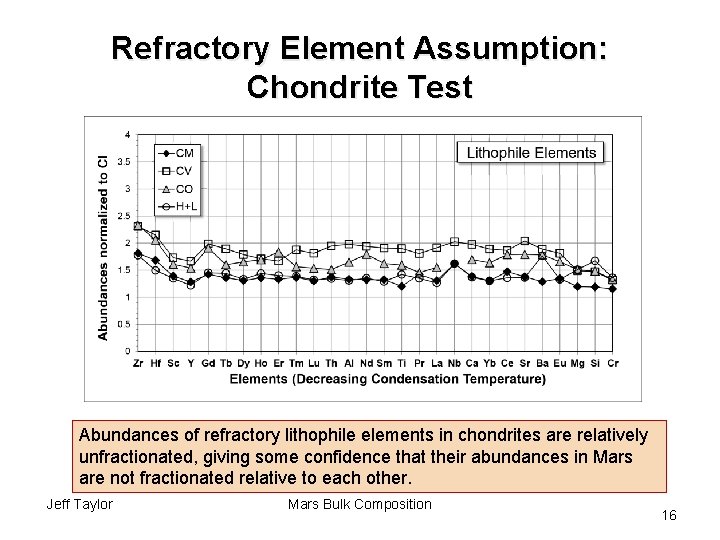
Refractory Element Assumption: Chondrite Test Abundances of refractory lithophile elements in chondrites are relatively unfractionated, giving some confidence that their abundances in Mars are not fractionated relative to each other. Jeff Taylor Mars Bulk Composition 16
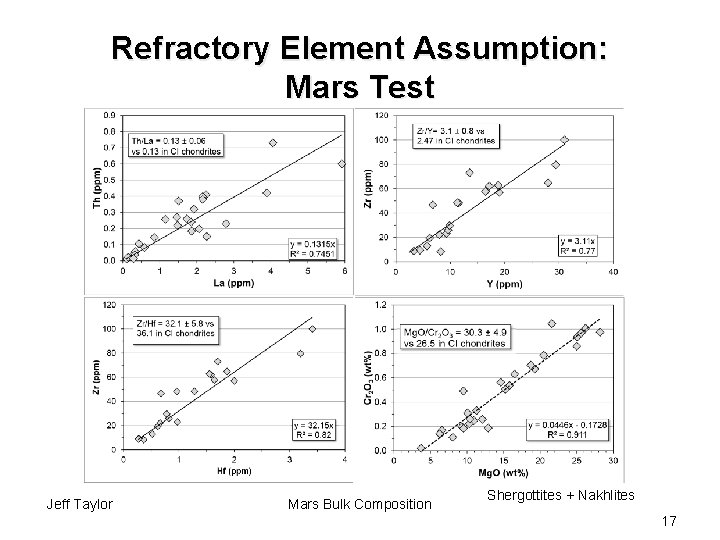
Refractory Element Assumption: Mars Test Jeff Taylor Mars Bulk Composition Shergottites + Nakhlites 17

Refractory Abundances • Assume elements as or more refractory than Cr have CI chondrite relative abundances • Exact procedures for this have never been explained. • Secret steps include: – Normalize CI abundances for Cr and more refractory to 100% – But do not include concentrations of Ni, Co, and Fe in metal (core). Ni. O, Co. O low, so no problem. Fe. O is high, so need to know that. – Figure out what to do about oxygen (volatile, but refractories are mostly oxides) – Do some secondary renormalizing to 100% – Don’t tell anyone how to do it. Jeff Taylor Mars Bulk Composition 18

Mn. O and Fe. O • Mn. O and Fe. O tend not to fractionate from each other if major phases are olivine or pyroxene (partition coefficients for both are about 1) • Mn. O in Martian meteorites are 0. 45 to 0. 55 wt% • If mantle is of CI chondrite composition, Mn. O would be expected to be 0. 46 wt% • Implies Mars not depleted in Mn • This means that Mn. O in Mars divided by Mn. O in CI is 1 Jeff Taylor Mars Bulk Composition 19

Mn. O and Fe. O: Wänke and Dreibus • Fe. O/Mn. O in Martian meteorites is 39. 1 • Fe. O/Mn. O in CI is 100. 6 • If Mn. O is at CI abundance (0. 46), then Fe. O in mantle is 39. 1*. 46 = 17. 9 wt Jeff Taylor Mars Bulk Composition 20
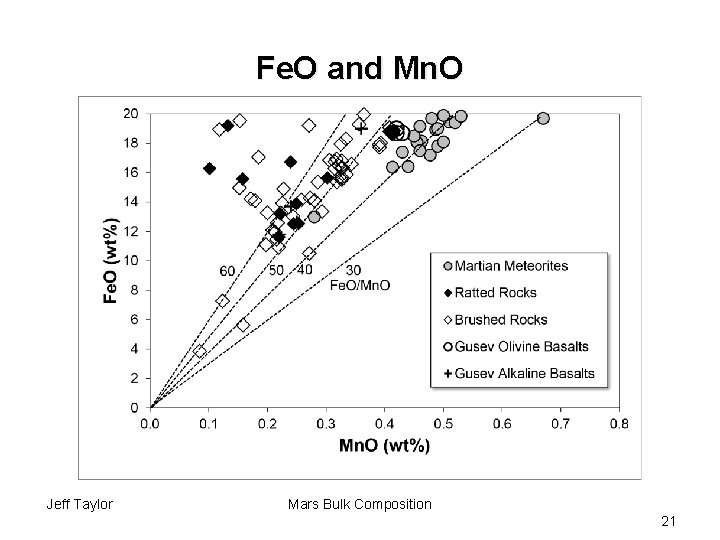
Fe. O and Mn. O Jeff Taylor Mars Bulk Composition 21
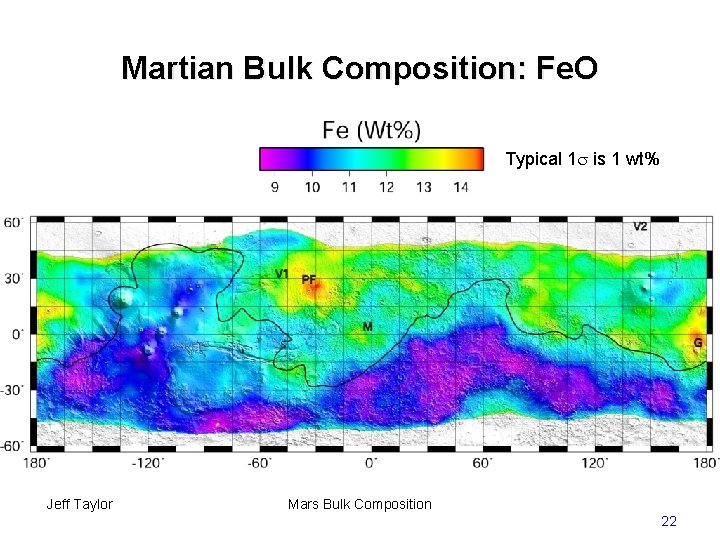
Martian Bulk Composition: Fe. O Typical 1 is 1 wt% Jeff Taylor Mars Bulk Composition 22
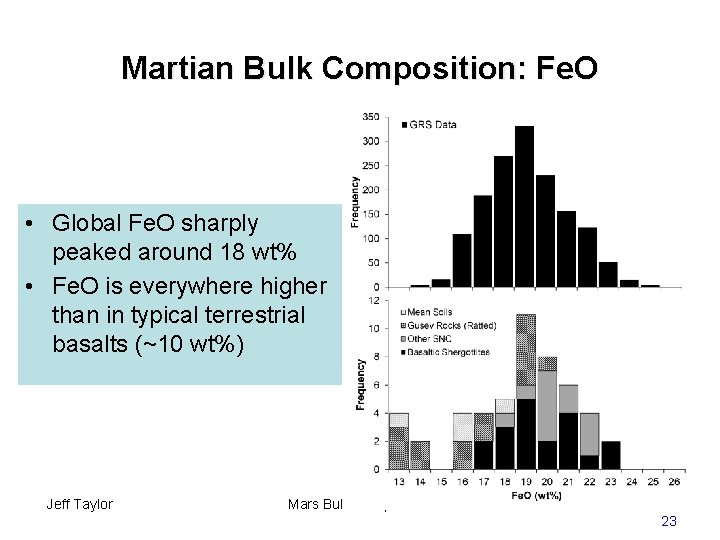
Martian Bulk Composition: Fe. O • Global Fe. O sharply peaked around 18 wt% • Fe. O is everywhere higher than in typical terrestrial basalts (~10 wt%) Jeff Taylor Mars Bulk Composition 23
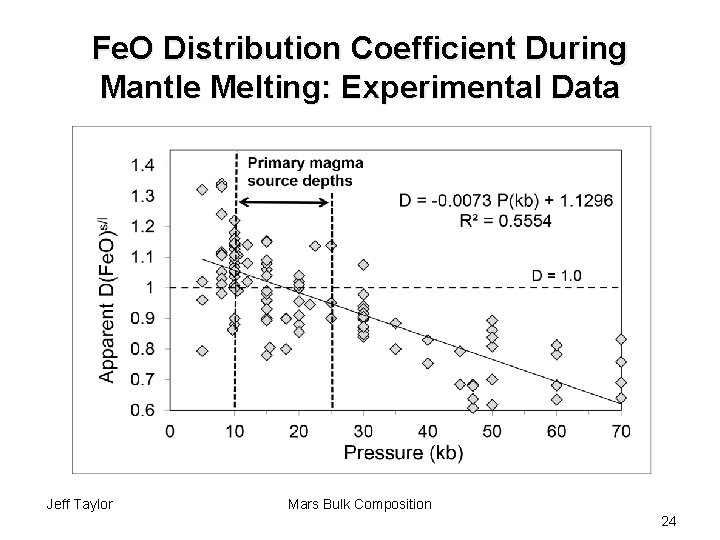
Fe. O Distribution Coefficient During Mantle Melting: Experimental Data Jeff Taylor Mars Bulk Composition 24

Fe. O cannot be much lower than surface composition implies • Moment of inertia constrains mg# of mantle to 60 -80 (though it depends on core radius and composition and crustal thickness) • Mars Fe. O (18. 1 wt%) and Mg. O (30. 5 wt%) imply an mg# of 75± 4, consistent with moment of inertia • Highest mg# possible (80) is still substantially less than in Earth (90), so Mars must have more Fe. O than Earth (> 8 wt%) • High Fe. O agrees with geophysical modeling and data. Jeff Taylor Mars Bulk Composition 25
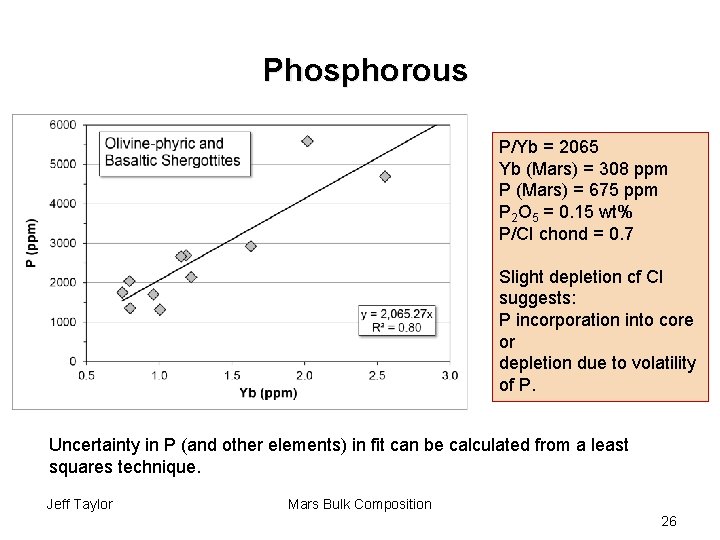
Phosphorous P/Yb = 2065 Yb (Mars) = 308 ppm P (Mars) = 675 ppm P 2 O 5 = 0. 15 wt% P/CI chond = 0. 7 Slight depletion cf CI suggests: P incorporation into core or depletion due to volatility of P. Uncertainty in P (and other elements) in fit can be calculated from a least squares technique. Jeff Taylor Mars Bulk Composition 26
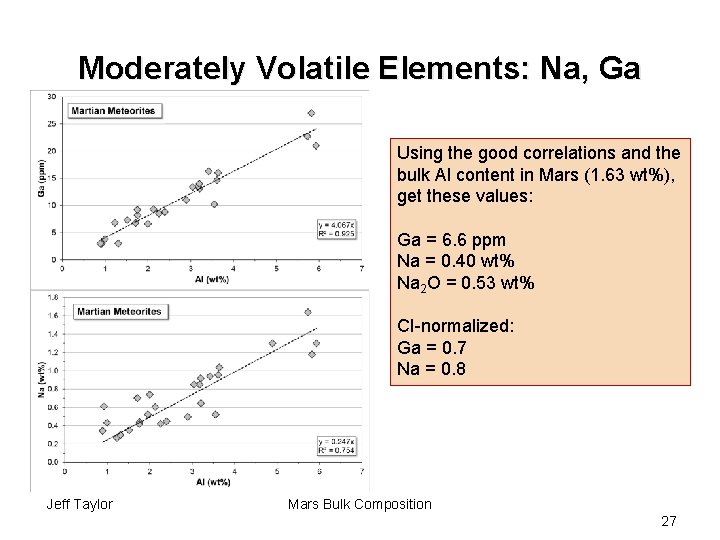
Moderately Volatile Elements: Na, Ga Using the good correlations and the bulk Al content in Mars (1. 63 wt%), get these values: Ga = 6. 6 ppm Na = 0. 40 wt% Na 2 O = 0. 53 wt% CI-normalized: Ga = 0. 7 Na = 0. 8 Jeff Taylor Mars Bulk Composition 27
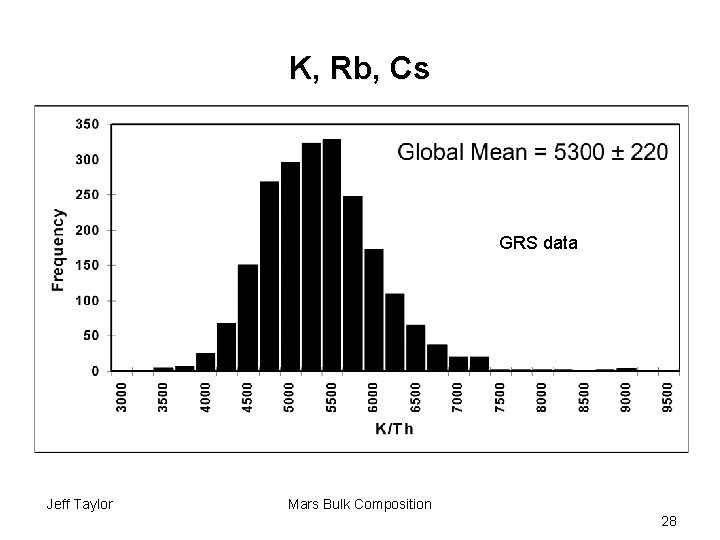
K, Rb, Cs GRS data Jeff Taylor Mars Bulk Composition 28
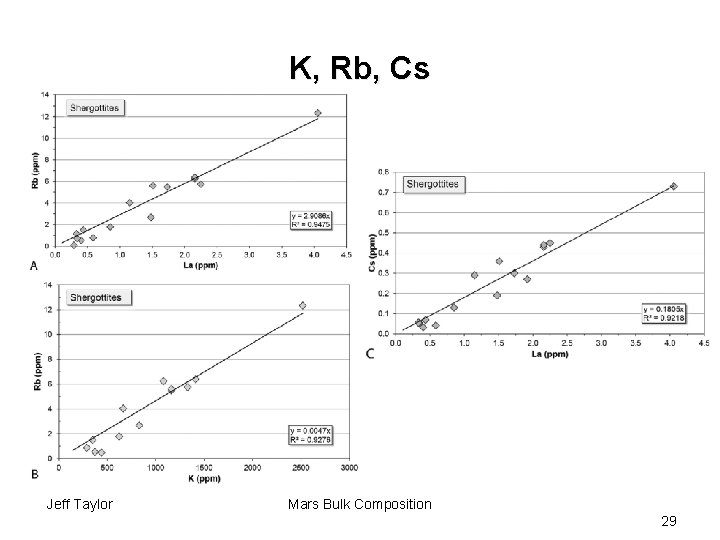
K, Rb, Cs Jeff Taylor Mars Bulk Composition 29
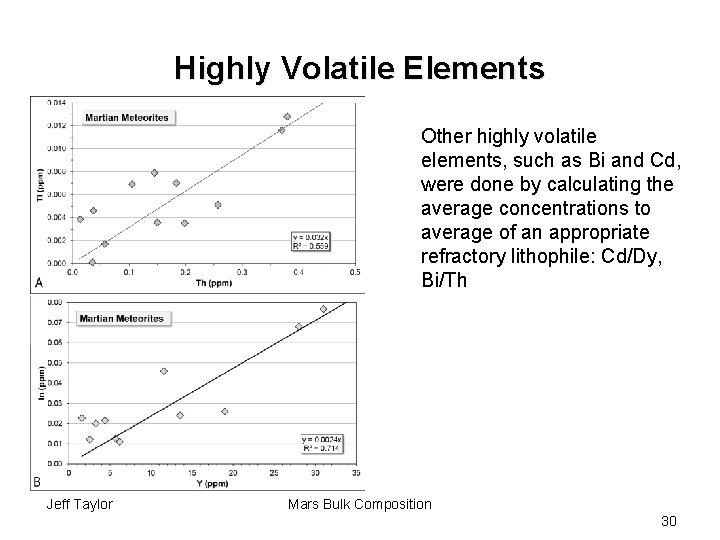
Highly Volatile Elements Other highly volatile elements, such as Bi and Cd, were done by calculating the average concentrations to average of an appropriate refractory lithophile: Cd/Dy, Bi/Th Jeff Taylor Mars Bulk Composition 30
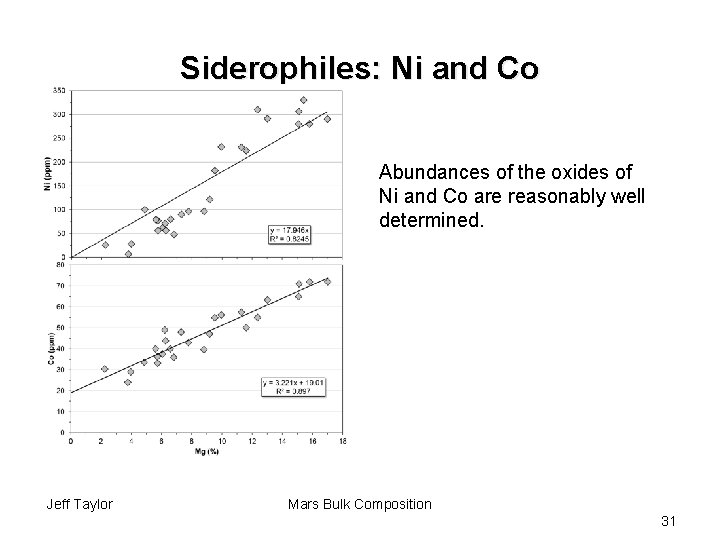
Siderophiles: Ni and Co Abundances of the oxides of Ni and Co are reasonably well determined. Jeff Taylor Mars Bulk Composition 31
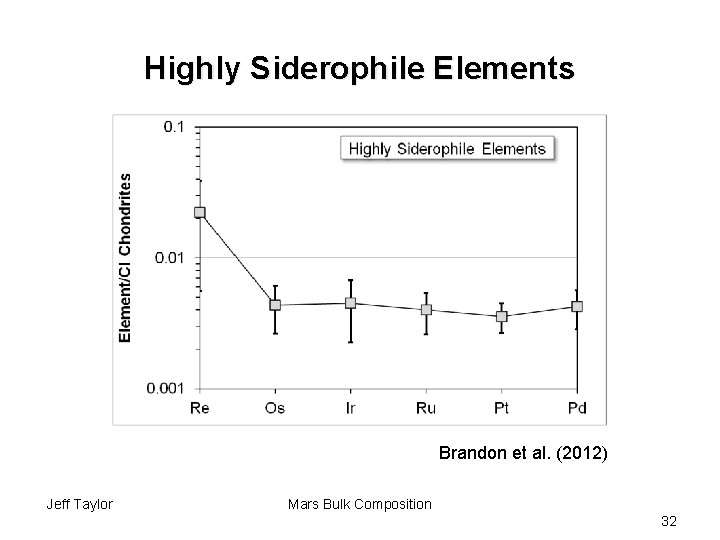
Highly Siderophile Elements Brandon et al. (2012) Jeff Taylor Mars Bulk Composition 32

The Core • Geophysical data suggest that the core has to have substantial S in it. • Refractory elements are 1. 9 x CI abundances – So Fe+Fe. O = CI abundance – Ni is essentially entirely in core • Assume S abundance is same as moderately volatile elements (0. 6 x CI) 5. 4 S in CI. • Partition total iron into mantle and core so the mantle ends up with 18 wt% Fe. O equivalent • Rest of iron, all the Ni, and all the S goes into core: 78. 6 wt% Fe-Ni, 21. 6 wt% S. Jeff Taylor Mars Bulk Composition 33
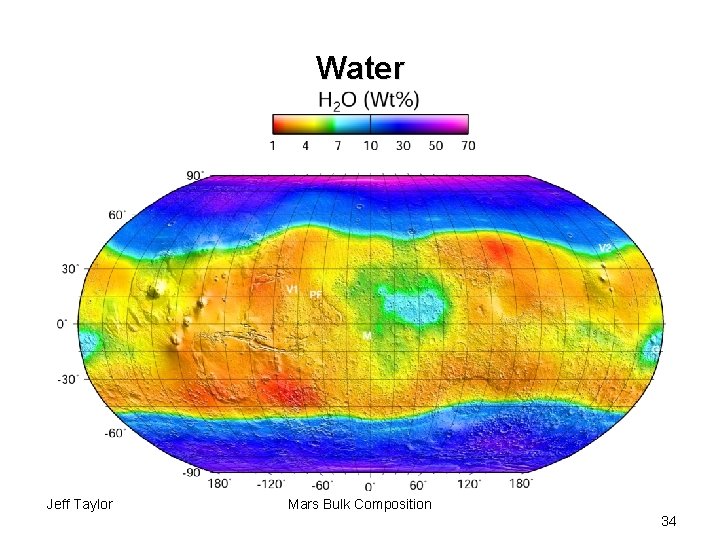
Water Jeff Taylor Mars Bulk Composition 34

Bulk Water Content Dreibus and Wänke: 30 ppm Estimates using data from: Mc. Cubbin (Chassigny): 130 -250 ppm Mc. Cubbin (shergottites): 63 -87 ppm Hallis (Nakhla): 150 -220 Mc. Cubbin (NWA 7034): 180 H/Cl on surface: 330 ppm Usui (Y 980459): 15 -47 ppm My best guess: 300 ± 150 ppm Earth: ≥ 500 ppm Jeff Taylor Mars Bulk Composition 35
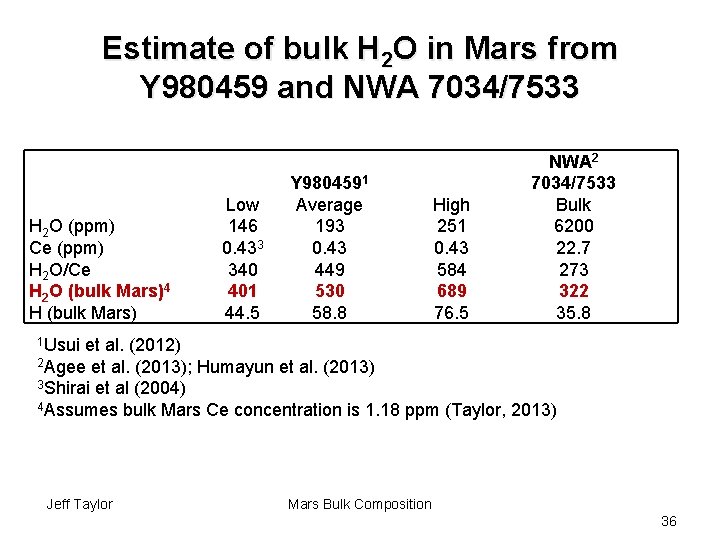
Estimate of bulk H 2 O in Mars from Y 980459 and NWA 7034/7533 H 2 O (ppm) Ce (ppm) H 2 O/Ce H 2 O (bulk Mars)4 H (bulk Mars) Low 146 0. 433 340 401 44. 5 Y 9804591 Average 193 0. 43 449 530 58. 8 High 251 0. 43 584 689 76. 5 NWA 2 7034/7533 Bulk 6200 22. 7 273 322 35. 8 1 Usui et al. (2012) 2 Agee et al. (2013); Humayun et al. (2013) 3 Shirai et al (2004) 4 Assumes bulk Mars Ce concentration is 1. 18 ppm (Taylor, 2013) Jeff Taylor Mars Bulk Composition 36
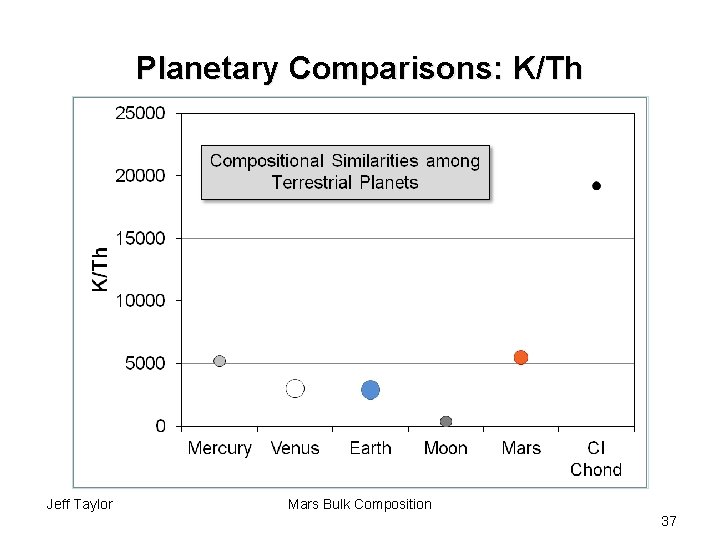
Planetary Comparisons: K/Th Jeff Taylor Mars Bulk Composition 37

Jeff Taylor Mars Bulk Composition 38
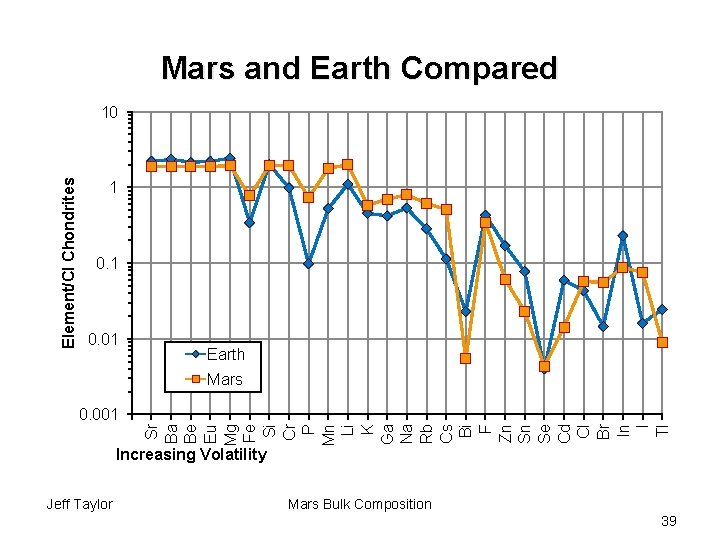
Mars and Earth Compared Element/CI Chondrites 10 1 0. 01 Earth Mars Sr Ba Be Eu Mg Fe Si Cr P Mn Li K Ga Na Rb Cs Bi F Zn Sn Se Cd Cl Br In I Tl 0. 001 Increasing Volatility Jeff Taylor Mars Bulk Composition 39
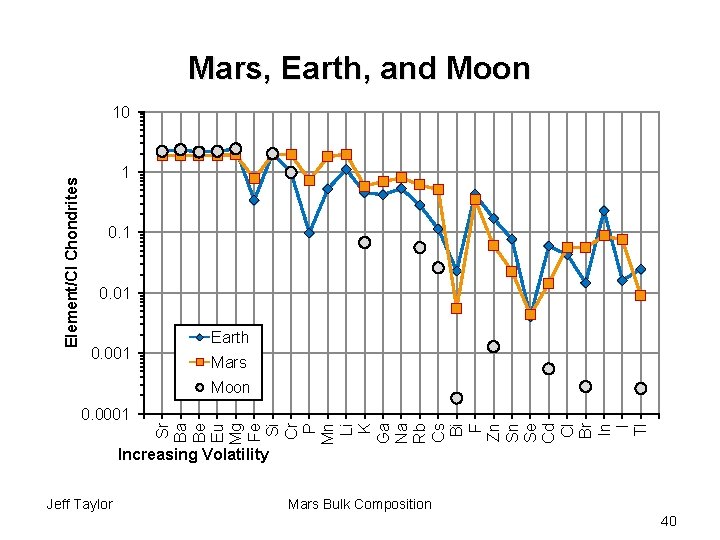
Mars, Earth, and Moon Element/CI Chondrites 10 1 0. 01 0. 001 Earth Mars Moon Sr Ba Be Eu Mg Fe Si Cr P Mn Li K Ga Na Rb Cs Bi F Zn Sn Se Cd Cl Br In I Tl 0. 0001 Increasing Volatility Jeff Taylor Mars Bulk Composition 40
- Slides: 40