Japanese SOF RBV Study SOF RBV in genotype
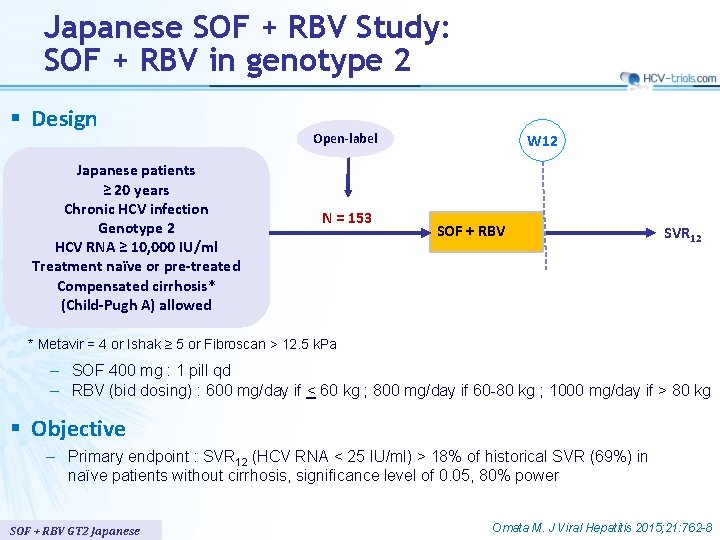
Japanese SOF + RBV Study: SOF + RBV in genotype 2 § Design Japanese patients ≥ 20 years Chronic HCV infection Genotype 2 HCV RNA ≥ 10, 000 IU/ml Treatment naïve or pre-treated Compensated cirrhosis* (Child-Pugh A) allowed Open-label N = 153 W 12 SOF + RBV SVR 12 * Metavir = 4 or Ishak ≥ 5 or Fibroscan > 12. 5 k. Pa – SOF 400 mg : 1 pill qd – RBV (bid dosing) : 600 mg/day if < 60 kg ; 800 mg/day if 60 -80 kg ; 1000 mg/day if > 80 kg § Objective – Primary endpoint : SVR 12 (HCV RNA < 25 IU/ml) > 18% of historical SVR (69%) in naïve patients without cirrhosis, significance level of 0. 05, 80% power SOF + RBV GT 2 Japanese Omata M. J Viral Hepatitis 2015; 21: 762 -8
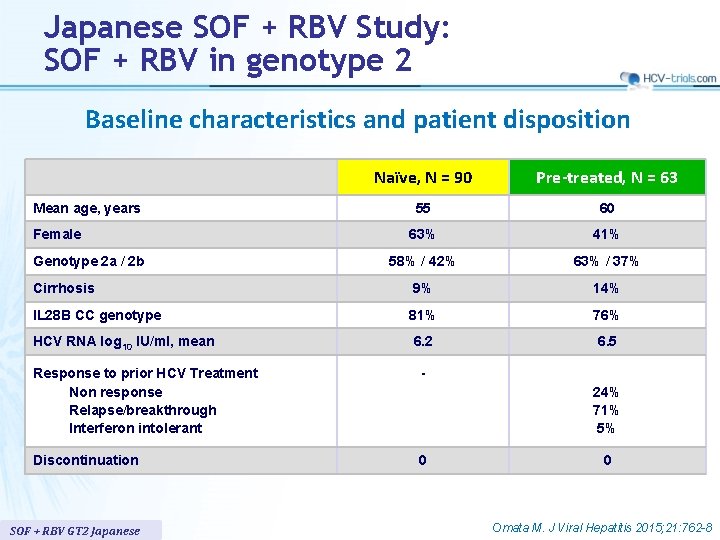
Japanese SOF + RBV Study: SOF + RBV in genotype 2 Baseline characteristics and patient disposition Naïve, N = 90 Pre-treated, N = 63 55 60 63% 41% 58% / 42% 63% / 37% Cirrhosis 9% 14% IL 28 B CC genotype 81% 76% 6. 2 6. 5 Mean age, years Female Genotype 2 a / 2 b HCV RNA log 10 IU/ml, mean Response to prior HCV Treatment Non response Relapse/breakthrough Interferon intolerant - Discontinuation 0 SOF + RBV GT 2 Japanese 24% 71% 5% 0 Omata M. J Viral Hepatitis 2015; 21: 762 -8
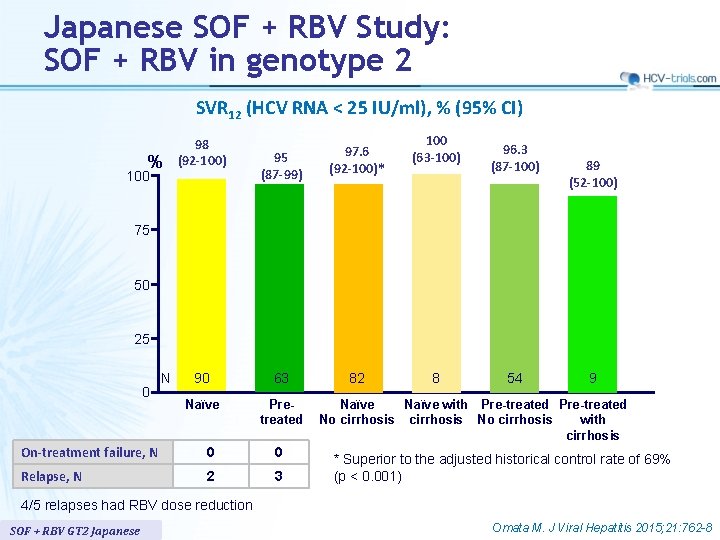
Japanese SOF + RBV Study: SOF + RBV in genotype 2 SVR 12 (HCV RNA < 25 IU/ml), % (95% CI) % 100 98 (92 -100) 95 (87 -99) 97. 6 (92 -100)* 90 63 82 Naïve Pretreated 100 (63 -100) 96. 3 (87 -100) 89 (52 -100) 75 50 25 0 N On-treatment failure, N 0 0 Relapse, N 2 3 8 54 9 Naïve with Pre-treated No cirrhosis with cirrhosis * Superior to the adjusted historical control rate of 69% (p < 0. 001) 4/5 relapses had RBV dose reduction SOF + RBV GT 2 Japanese Omata M. J Viral Hepatitis 2015; 21: 762 -8
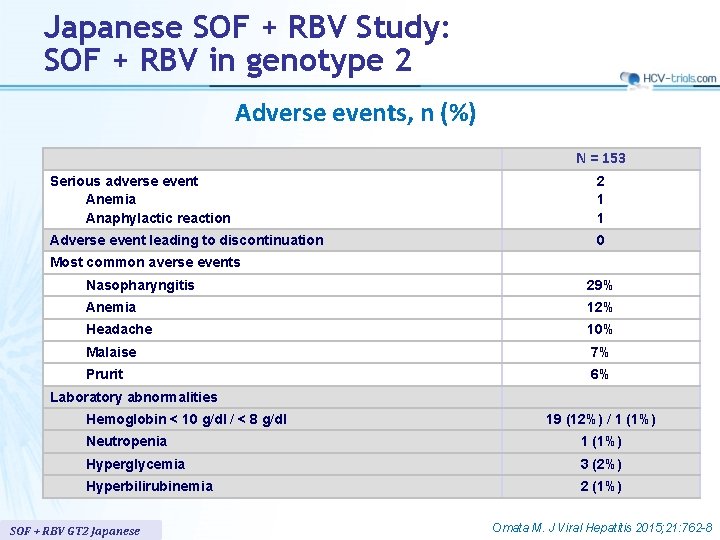
Japanese SOF + RBV Study: SOF + RBV in genotype 2 Adverse events, n (%) N = 153 Serious adverse event Anemia Anaphylactic reaction 2 1 1 Adverse event leading to discontinuation 0 Most common averse events Nasopharyngitis 29% Anemia 12% Headache 10% Malaise 7% Prurit 6% Laboratory abnormalities Hemoglobin < 10 g/dl / < 8 g/dl 19 (12%) / 1 (1%) Neutropenia 1 (1%) Hyperglycemia 3 (2%) Hyperbilirubinemia 2 (1%) SOF + RBV GT 2 Japanese Omata M. J Viral Hepatitis 2015; 21: 762 -8

Japanese SOF + RBV Study: SOF + RBV in genotype 2 § Summary – 12 weeks of treatment with SOF + RBV resulted in high rates of SVR 12 (> 95%) in treatment-naïve and previously treated Japanese patients with chronic genotype 2 HCV infection – In the present study, 22% of patients were aged 65 or older and 11% had cirrhosis • Efficacy similar • Increases in reported adverse events and laboratory abnormalities in patients ≥ 65 years, but these differences did not present a barrier to treatment as no premature discontinuation of study treatment occurred in any patient – Relapse rate was 3%, and none of the subjects who relapsed had S 282 T or other nucleoside inhibitor resistance-associated variants SOF + RBV GT 2 Japanese Omata M. J Viral Hepatitis 2015; 21: 762 -8
- Slides: 5